 |
 |
| Korean J Intern Med > Volume 40(2); 2025 > Article |
|
Abstract
Background/Aims
This study aimed to evaluate the impact of steatotic liver disease severity on the cumulative incidence of lung cancer utilizing data from the Korea National Health Insurance Service (NHIS).
Methods
This study examined the risk of lung cancer in the general population in conjunction with the incidence of steatotic liver disease. The study population consisted of 3,261,438 individuals aged 20 years or older who underwent a general health examination in 2009.
Results
Individuals with fatty liver index (FLI) of 30ŌĆō59 exhibited a 1.08-fold increased risk of lung cancer (95% CI: 1.04ŌĆō1.11), while FLI Ōēź 60 was associated with a 1.22-fold elevated risk of lung cancer (95% CI: 1.17ŌĆō1.28) compared to those with FLI < 30. The risk varied with smoking status; in current smokers, the adjusted HR for the FLI 30ŌĆō59 group was 1.05 (95% CI: 1.00ŌĆō1.10), while that in the FLI Ōēź 60 group was 1.11 (95% CI: 1.04ŌĆō1.18). In never- or past-smokers, the adjusted HR for the FLI 30ŌĆō59 group was 1.10, and that for the FLI Ōēź 60 group was 1.31. Subgroup analysis revealed an incidence rate of 1.06 per 1,000 person-years in the consistently high FLI group compared to 1.15 in those with improved FLI. Improving FLI over time was associated with a 0.93-fold decrease in lung cancer risk.
Steatotic liver disease viewed as a hepatic manifestation of metabolic dysfunction, has been reported to have an association with hepatocellular carcinoma (HCC). Steatotic liver disease prevalence, which is expected to increase, is approximately 30% in Korea and 30ŌĆō50% in Western countries [1]; clinical interest in diseases associated with steatotic liver disease is increasing. The carcinogenetic correlation with steatotic liver disease does not apply only to primary liver cancer. Excessive adipose tissue promotes the production of proinflammatory cytokines and suppresses cell death, creating an environment favorable to tumor development [2]. A meta-analysis of observational cohort studies showed an increased risk of extrahepatic cancers, e. g., lung, breast, gynecological, and urinary system cancers, in individuals with steatotic liver disease, suggesting that the carcinogenic correlation extends beyond HCC and includes extrahepatic solid cancers [3,4]. A study utilizing the Swedish National Patient Registry revealed that both the incidence of HCC and the risk for certain non-HCC solid cancer subtypes were significantly higher in the steatotic liver disease group compared to controls. However, the number of patients included in the extrahepatic subtype groups was small [5].
Lung cancer, the leading cause of cancer-related deaths worldwide, is primarily associated with environmental and lifestyle factors [6]. The most significant risk factors for its development include tobacco use, exposure to secondhand smoke, air pollution, and occupational or household exposure to carcinogenic materials [7ŌĆō10]. Despite the established links to these risk factors, the role of metabolic conditions, such as metabolic syndrome and fatty liver disease, in lung cancer development is an emerging area of interest. Recent studies suggest a potential association between metabolic dysfunctions and lung cancer risk [11,12]. Although the impact may not be as pronounced as that of smoking or air pollution, understanding this relationship is crucial because fatty liver disease represents a modifiable risk factor that could be targeted through lifestyle changes.
Therefore, while the primary causes of lung cancer have been identified, the implications of metabolic syndrome and fatty liver disease on lung cancer development is a significant task. To this end, we conducted a study comparing the cumulative incidence of lung cancer in cases of steatotic liver disease with that in the general population using data from the Korea National Health Insurance Service (NHIS). We also examined the pertinent risk factors of steatotic liver disease on lung cancer occurrence.
This study examined the risk of lung cancer in steatotic liver disease cases in the general population. A cohort was established using nationwide claims data from the Korea NHIS [13]. KoreaŌĆÖs universal health coverage system has provided healthcare coverage to the entire population since 1989. The claims data include a comprehensive overview of healthcare history, including demographic data; socioeconomic status determined by monthly household income; diagnoses following the International Classification of Diseases, 10th Revision (ICD-10) guidelines; and detailed treatment and prescription data. Additionally, the NHIS has been offering general health screenings once every two years to all Koreans aged 20 years or older. Participant rates in this examination program have ranged from 56.1ŌĆō67.7% during the recent decade [14]. The general health examination comprises body measurements, blood pressure assessment, blood tests, and a collection of questionnaires to acquire behavior information.
The study population consisted of adult individuals aged 20 years or older who underwent a general health examination in 2009 (n = 4,234,412). Heavy alcohol consumers (n = 354,376), those who had viral hepatitis infection (n = 4,045,888) or liver cirrhosis (n=14,835), those who had a history of any malignancies (n = 50,128), those had insufficient medical records (n = 140,677), and those diagnosed with lung cancer within the first year of enrollment (n = 8,370) were excluded from the analysis. After exclusions, our study included 3,261,438 individuals. Participants were followed from the index date until death, emigration, or the conclusion of the study in December 2020.
Our study was approved by the Institutional Review Board of Soongsil University (IRB No. SSU-202007-HR-236-01) and was conducted in compliance with both the Declarations of Helsinki and Istanbul. Informed consent was not required as only de-identified data were utilized.
We gathered data on a number of variables, including age; gender; socioeconomic status; comorbidities; body measurements of height, weight, and waist circumference; blood tests for ╬│-glutamyl transpeptidase and triglycerides; smoking and alcohol consumption habits; and physical activity levels from the NHIS claims data. Comorbidities claimed within one year before the enrollment were evaluated. Based on this information, we calculated the overall comorbidity burden using the Charlson Comorbidity Index (CCI).
We employed the fatty liver index (FLI) to identify steatotic liver disease. The FLI was calculated using the formula [15]:
Among various clinical approaches, the FLI has been validated as a reliable diagnostic tool for detecting fatty liver disease with a number of associated etiologies without imaging techniques [15ŌĆō17]. Participants with FLI scores less than 30 were classified as low risk for steatotic liver disease; those scoring between 30 and 59 were placed in the category of indeterminate risk, and individuals with FLI scores of 60 or higher were considered to be at high risk for steatotic liver disease.
The primary outcome was development of lung cancer during the study period. Lung cancer was defined as a claim with the ICD-10 code C34 in conjunction with cancer-specific rare and incurable disease (RID) codes, specifically V193. Cancer patients registered under the RID system are responsible for 5% of the total medical expenses incurred for both outpatient and inpatient care. This necessitates stringent documentation of biochemical, histological, and clinical evidence so the accuracy of RIDs in Korea is reliable at greater than 90% [18]. The secondary aim of the study was determination of the change in lung cancer risk in conjunction with decreases in risk of steatotic liver disease in patients initially at an intermediate or high risk of steatotic liver disease (FLI Ōēź 30).
For baseline characteristics, categorical variables are presented as numbers and percentages, and continuous variables are presented as means ┬▒ standard deviations. Continuous variables were analyzed using the ANOVA test, and categorical variables were assessed using the chi-square test. We calculated the incidence rate of lung cancer per 1,000 person-years (PYs). Utilizing the KaplanŌĆōMeier method, cumulative incidence curves for the risk of lung cancer were generated based on the FLI. Covariates adjusted in the model were age; sex; socioeconomic status; presence of diabetes, hypertension, or dyslipidemia; body mass index; smoking status; alcohol consumption; and weekly exercise level. Subgroup analysis based on smoking status was conducted to evaluate the impact of steatotic liver disease on the risk of lung cancer according to smoking status. Additionally, we investigated the change in lung cancer risk in individuals who underwent physical examinations in two consecutive years and experienced a decrease in steatotic liver disease from an intermediate or high risk (FLI Ōēź 30) status. p values < 0.05 were deemed to indicate statistical significance. All statistical operations were conducted using SAS Enterprise Guide Software Version 9.4 (SAS Institute, Inc., Cary, NC, USA).
The baseline characteristics of the study population are shown in Table 1. The mean follow-up duration was 10.1 ┬▒ 1.2 years. Of the 3,261,438 participants, 2,150,983 (66.0%) were classified low risk of steatotic liver disease with an FLI < 30; 737,515 (22.6%) were in the intermediate risk group with an FLI of 30ŌĆō59; and 372,940 (11.4%) were categorized as high risk with an FLI Ōēź 60. Individuals with FLI < 30, at low risk of steatotic liver disease, tended to be younger and were predominantly female. Additionally, this group exhibited fewer metabolic dysfunction-related comorbidities such as hypertension, diabetes mellitus, and dyslipidemia and had the lowest CCI score of 0.7 ┬▒ 1.2 vs. 0.9 ┬▒ 1.4 vs. 0.9 ┬▒ 1.4; p < 0.001 in the low-, intermediate-, and high-risk groups, respectively. The low-risk group had the highest proportion of individuals classified as underweight or within the normal weight range. Moreover, the prevalence of never-smokers (70.7% vs. 50.6% vs. 39.2%; p < 0.001) and non-drinkers (60.0% vs. 49.7% vs. 38.8%; p < 0.001) was also highest among the FLI < 30 group.
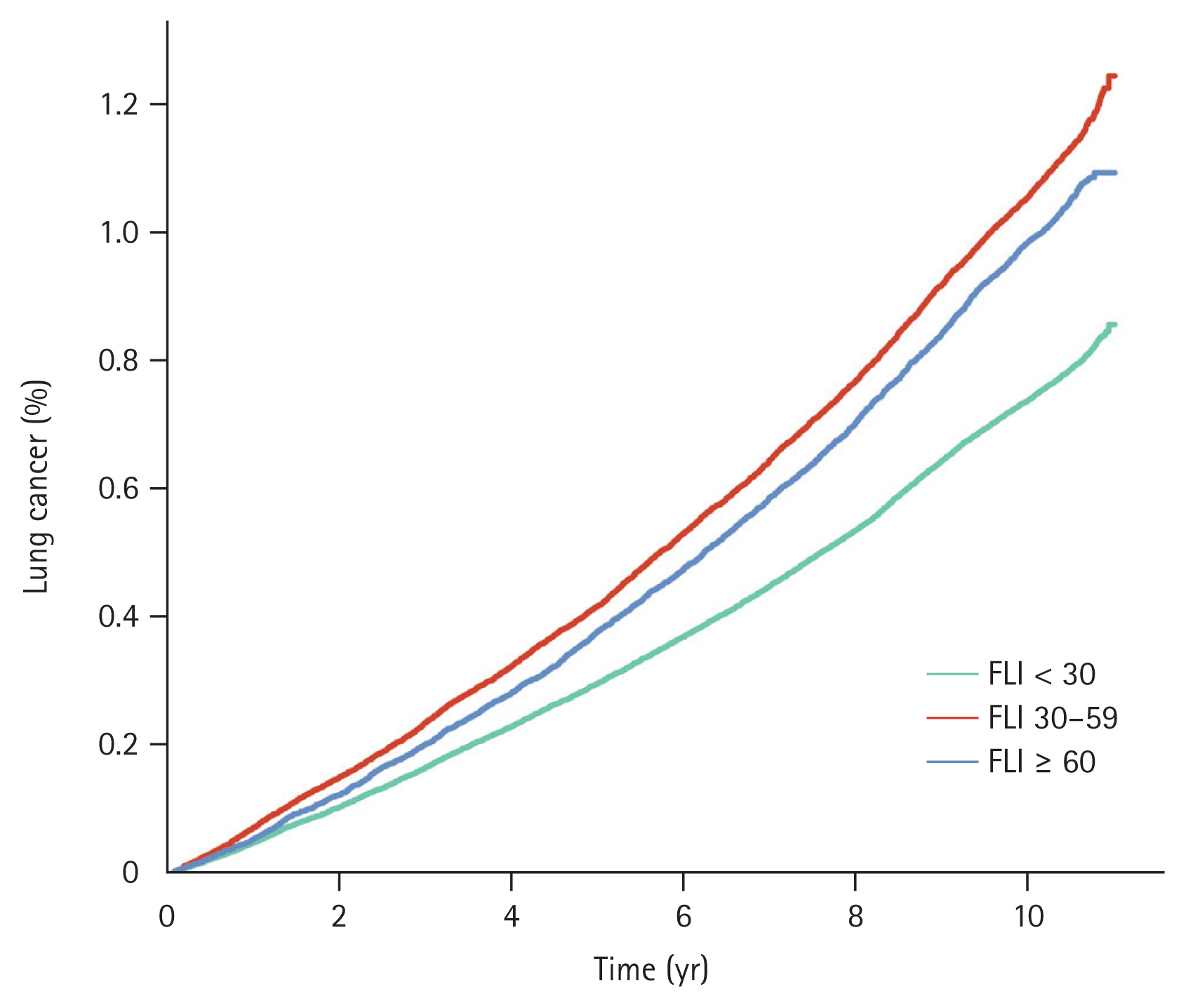
Throughout the observation period, 28,209 patients were diagnosed with lung cancer. Among these subjects, there were 16,415 cases of lung cancer in individuals with FLI < 30; the annual incidence was 0.75 per 1,000 PYs. There were 8,043 cases of lung cancer in the intermediate-risk, FLI 30ŌĆō59 group, with an annual incidence of 1.08 per 1,000 PYs. Additionally, there were 3,751 cases in the high-risk, FLI Ōēź 60 group, with an annual incidence of 1.00 per 1,000 PYs. The cumulative incidence rates of lung cancer for FLI < 30, FLI 30ŌĆō59, and FLI Ōēź 60 rates at one year were 0.05%, 0.07%, and 0.06%, respectively. These rates at five years were 0.30%, 0.42%, and 0.38%, respectively; and those at 10 years were 0.74%, 1.06%, and 0.99% (p < 0.001). These data are illustrated in Figure 1. Individuals with FLI < 30 demonstrated a significantly lower risk of lung cancer compared to the other groups; the crude hazard ratios (HRs) were 1.44 (95% confidence interval [CI]: 1.40ŌĆō1.48) in the FLI 30ŌĆō59 group and 1.33 in the FLI Ōēź 60 group (95% CI: 1.28ŌĆō1.38) before adjustment. Following adjustment for multiple variables, individuals with FLI 30ŌĆō59 exhibited a 1.08-fold increased risk of lung cancer (95% CI: 1.04ŌĆō1.11), and subjects with FLI Ōēź 60 had a 1.22-fold elevated risk of lung cancer (95% CI: 1.17ŌĆō1.28) compared to those with FLI < 30 (Table 2).
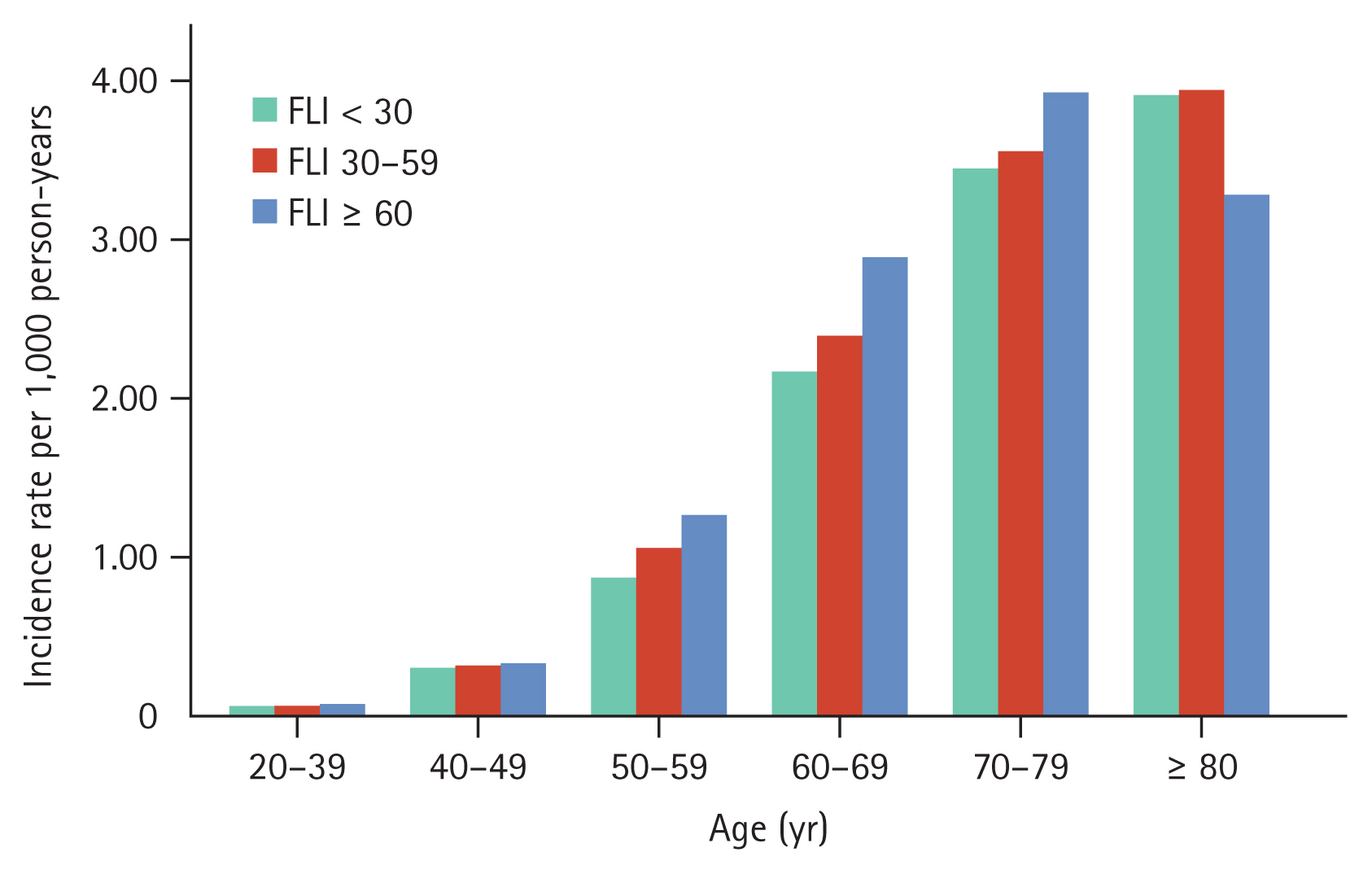
Lung cancer incidence rates increased with increasing age. The incidence rates for lung cancer across age groups were 0.08 for ages 20ŌĆō39 years, 0.32 per 1,000 PYs for ages 40ŌĆō49 years, 0.98 per 1,000 PYs for ages 50ŌĆō59 years, 2.32 per 1,000 PYs for ages 60ŌĆō69 years, 3.52 per 1,000 PYs for ages 70ŌĆō79 years, and 3.88 per 1,000 PYs for ages 80 years and above. In each age group, the incidence rate of lung cancer increased in conjunction with FLI increases except for those aged 80 years or above (Fig. 2). The adjusted HRs for lung cancer among individuals aged 50ŌĆō59 were 1.02 (95% CI: 0.96ŌĆō1.08) for FLI 30ŌĆō59 and 1.18 (95% CI: 1.10ŌĆō1.27) for FLI Ōēź 60. Similarly, in the 60ŌĆō69 age group, the adjusted HRs were 1.12 (95% CI: 1.07ŌĆō1.17) and 1.37 (95% CI: 1.29ŌĆō1.47), respectively. For those aged 70ŌĆō79, the adjusted HRs were 1.19 (95% CI: 1.13ŌĆō1.26) and 1.40 (95% CI: 1.29ŌĆō1.53), respectively; and in individuals aged 80 years or older, the adjusted HRs were 1.21 (95% CI: 1.04ŌĆō1.41) and 1.13 (95% CI: 0.85ŌĆō1.51; Supplementary Table 1).
We further stratified the study population into current smokers (n = 787,289), past smokers (n = 434,064), and never-smokers (n = 2,040,085). Regardless of smoking status, as FLI increased, the risk of lung cancer also increased. In current smokers, the adjusted HR for the FLI 30ŌĆō59 group was 1.05 (95% CI: 1.00ŌĆō1.10), while that of the FLI Ōēź 60 group was 1.11 (95% CI: 1.04ŌĆō1.18). Similarly, in past smokers, the adjusted HR for the FLI 30ŌĆō59 group was 1.18 (95% CI: 1.11ŌĆō1.25), while that of the FLI Ōēź 60 group was 1.46 (95% CI: 1.34ŌĆō1.58).
For the never-smoker group, the adjusted HR for lung cancer increased in those with FLI 30ŌĆō59 (1.06, 95% CI: 1.02ŌĆō1.11) and those with FLI Ōēź 60 (1.24, 95% CI: 1.16ŌĆō1.32; Table 3).
Of the 1,110,455 individuals with an FLI Ōēź 30 at enrollment, 665,801 underwent a general health examination two years later. We compared individuals with consistently intermediate or high FLI (n = 519,598) to those whose FLI improved from Ōēź 30 to < 30 (n = 146,203). The baseline characteristics are summarized in Supplementary Table 2. Individuals who experienced an improvement in FLI showed a lower prevalence of comorbidities of hypertension, diabetes mellitus, and dyslipidemia compared to their counterparts. During the observational period of the subgroup analysis, the incidence rate was 1.07 per 1,000 PYs in the consistently FLI Ōēź 30 group and was 1.14 per 1,000 PYs in the improved FLI group. Utilizing multivariable Cox regression analysis, the risk of lung cancer decreased by 0.93-fold (95% CI: 0.88ŌĆō0.99) in the groups with improved FLI (Table 4).
Our study showed that steatotic liver disease, manifested as high FLI, was associated with increased lung cancer development in various subgroups. steatotic liver disease was also an independent risk factor of lung cancer regardless of smoking status. Moreover, resolution of steatotic liver disease exhibited a possible protective influence against lung cancer among individuals with the condition.
Studies on the association of steatotic liver disease and lung cancer are relatively rare. The few studies on an association between lung cancer development and metabolic syndrome had inconsistent results. A meta-analysis that included five cohort studies with a total of 188,970 subjects did not find a meaningful association between metabolic syndrome and lung cancer (HR 0.95, 95% CI: 0.81ŌĆō1.10) [19]. A large Korean study that used a national insurance database of subjects showed a significant association between metabolic syndrome and lung cancer. Metabolic syndrome positively correlated with an increased risk of lung cancer in men (HR 1.15, 95% CI: 1.12ŌĆō1.18) [12]. A recently published UK study including 331,877 participants with a median follow-up duration of 10.9 years showed an increased risk of lung cancer with metabolic syndrome (HR 1.21, 95% CI: 1.09ŌĆō1.33); these data support our results [11].
In the present study, individuals in the low-risk steatotic liver disease category (FLI < 30) were younger, predominantly female, and showed lower prevalence of metabolic dysfunction-related comorbidities such as hypertension, diabetes mellitus, and dyslipidemia. This group also demonstrated the lowest CCI scores, suggesting a generally healthier baseline status compared to intermediate- and high-risk groups. These findings are consistent with those of previous studies on steatotic liver disease defined by FLI [20,21]. After adjusting for multiple variables, the persistence of increased risk of lung cancer (1.08-fold for the FLI 30ŌĆō59 group and 1.22-fold for the FLI Ōēź 60 group) compared to those with FLI < 30 supports the hypothesis that the relationship between FLI and lung cancer risk is not merely coincidental. The mechanism underlying the association between metabolic disorders and carcinogenesis is complex and has not been characterized fully. Possible pathways involve chronic inflammation caused by deregulation of cytokine production, insulin resistance, and overexpression of estrogen; these may be linked to the carcinogenesis process [22]. Lipid accumulation in hepatocytes and an associated lipotoxicity create a dynamic proinflammatory environment that promotes eventual fibrogenesis and subsequent carcinogenesis [23]. As for the potential impact of steatotic liver disease on lung cancer risk, we assume that mechanisms differ based on effects of liver diseases as the process of liver fibrogenesis does not directly lead to the development of lung cancer. We believe that the broader systemic inflammation associated with steatotic liver disease, driven by lipid accumulation, may increase the risk of lung cancer through pro-inflammatory pathways that support a tumor-friendly environment [24]. To further clarify the potential molecular and pathological connections between lung cancer and steatotic liver disease, extensive epidemiological data on the interplay between these two disease entities is needed. Furthermore, additional studies are needed to explore not only the isolated impact of steatotic liver disease on lung cancer risk, but also its synergistic effect when combined with known risk factors such as tobacco use and air pollution. Lipotoxicity associated with steatotic liver disease may act as a catalyst for these risk factors.
In current smokers, the adjusted HRs for lung cancer increased modestly but significantly as FLI increased. This is consistent with findings on metabolic syndrome and lung cancer risk; the risk of lung cancer resulting from metabolic syndrome was elevated mainly among patients who used tobacco [11]. Conversely, in the never- or past-smokers subgroup, the impact of FLI on lung cancer risk was more pronounced. These findings suggest that, in the absence of active smoking, metabolic dysfunction plays a more significant role in lung cancer risk, potentially acting through mechanisms independent of those associated with tobacco exposure. Furthermore, this finding supports interventions targeting metabolic health regardless of individual smoking status.
We separately analyzed data from individuals who initially presented with intermediate or high steatotic liver disease risk (FLI Ōēź 30) and underwent a general health examination two years later to evaluate the potential impact of longitudinal change in FLI on lung cancer risk. Notably, those who achieved an improvement in their FLI demonstrated a lower prevalence of comorbid conditions of hypertension, diabetes mellitus, and dyslipidemia and were more likely to be never-smokers, non-drinkers, and 65 years or older. In the crude analysis, contrary to expectations, the incidence rate of lung cancer was slightly higher in the group that improved their FLI compared to those with consistent FLI Ōēź 60. There is a possibility that this longitudinal analysis did not capture the beneficial effect of intentionally lowering the FLI index. Future studies need to determine if changes in the FLI result from lifestyle modifications [25ŌĆō27] or from the development of new systemic conditions such as chronic kidney diseases or autoimmune diseases that may affect body composition or BMI. These conditions ultimately influence the FLI [28ŌĆō31]. Furthermore, considering that the proportion of elderly patients was higher in the group that showed improved FLI, an age effect on cancer risk cannot be ignored [32]. In the multivariable analysis that included factors such as age and exercise frequency, however, the results showed that FLI improvement was associated with a 0.93-fold decrease in lung cancer. This significant reduction in multivariate analysis suggests a consistent potential beneficial effect from improvement in FLI. Interestingly, weight reduction, the first-line treatment for steatotic liver disease, tended to lower cancer risks in previous studies. Luo et al. [33] showed that postmenopausal women with weight loss significantly lowered the risk of endometrial cancer compared to those with stable weight (HR 0.71, 95% CI: 0.54ŌĆō0.95). Also, Chlebowski et al. [34] reported that postmenopausal women who experienced weight loss had a decreased risk of breast cancer compared to those who maintained a stable weight (HR 0.88, 95% CI: 0.78ŌĆō0.98). Moreover, rigorous lifestyle interventions among overweight or obese individuals and those with type 2 diabetes reduced the risk of obesity-related cancer by 0.84 times compared to the diabetes support and education group in a randomized clinical trial [35]. To accurately determine if lifestyle modifications that improve the FLI can reduce cancer risk, a well-designed, prospective, matched comparison study is necessary.
Our study that investigated the link between steatotic liver disease and lung cancer risk had some limitations. First, due to the innate limitation of claims data, we were not able to gather detailed medical information. Therefore, we could not assess the lung cancer stage or pathologic types and severity of other comorbidities. Also, steatotic liver disease and lung cancer share common risk factors such as chronic inflammation, insulin resistance, and oxidative stress, which may potentially be managed with medication. It highlights the need for further research to explore the mechanisms underlying the relationship between steatotic liver disease and lung cancer. Second, the incidence of lung cancer may be underestimated since it did not consider individuals with lung cancer who did not seek medical attention. Third, even if we defined steatotic liver disease by FLI, the lack of imaging techniques remains a limitation of our study. We defined change of hepatic steatosis with 2-year period variation, it might have an innate limitation of fully reflect individual variations of hepatic steatosis overtime. Last, detailed tobacco exposure or concurrent chronic lung disease information was not acquired. Despite these limitations, our study has strengths in its basis on a national population database with sufficient observation time.
In conclusion, our study utilizing nationwide population data demonstrated that the presence and severity of steatotic liver disease as assessed by the FLI have an incremental relationship with lung cancer risk. Future research that includes more detailed information on comorbidities and lifestyle modifications will further clarify this association.
1. In the analysis included over three million individuals who underwent a general health examination, higher FLI scores were associated with an increased risk of lung cancer, with variations in risk observed based on smoking status.
2. For the never-smoker group, the risk of lung cancer increased with higher FLI scores, with significant risk elevation observed in those with FLI 30ŌĆō59 and even more so in those with FLI Ōēź 60.
3. Individuals with consistently high FLI levels had a higher incidence of lung cancer compared to those who improved their FLI over time.
Notes
CRedit authorship contributions
Jihye Lim: conceptualization, methodology, resources, data curation, formal analysis, software, writing - original draft, visualization, funding acquisition; Bongseong Kim: methodology, resources, data curation, formal analysis, validation, software, visualization; Kyungdo Han: methodology, resources, investigation, data curation, formal analysis, validation, software, supervision, project administration, funding acquisition; Jeong Uk Lim: conceptualization, methodology, resources, investigation, validation, writing - original draft, writing - review & editing, supervision, project administration, funding acquisition
Data availability
Data used in this study are maintained by the Korea National Health Insurance Service (NHIS, https://nhiss.nhis.or.kr) and are available upon request to the NHIS.
Figure┬Ā1
Event frequencies and KaplanŌĆōMeier estimates for lung cancer according to metabolic disease-associated fatty liver disease risk. FLI, fatty liver index.
Figure┬Ā2
Incidence rate of lung cancer rates by metabolic-associated fatty liver disease risk across age groups. FLI, fatty liver index.
Table┬Ā1
Baseline characteristics of individuals according to the FLI
| Variable | Total (n = 3,261,438) | FLI | p value | ||
|---|---|---|---|---|---|
| < 30 (n = 2,150,983) | 30ŌĆō60 (n = 737,515) | Ōēź 60 (n = 372,940) | |||
| Follow-up duration (yr) | 10.1 ┬▒ 1.2 | 10.2 ┬▒ 1.2 | 10.1 ┬▒ 1.3 | 10.1 ┬▒ 1.3 | |
| Age (yr) | 46.8 ┬▒ 14.1 | 45.6 ┬▒ 14.4 | 50.1 ┬▒ 13.5 | 47.4 ┬▒ 12.8 | < 0.001 |
| ŌĆā20ŌĆō39 | 1,054,704 (32.3) | 759,579 (35.3) | 180,313 (24.5) | 114,812 (30.8) | |
| ŌĆā40ŌĆō64 | 1,788,068 (54.8) | 1,137,980 (52.9) | 434,618 (58.9) | 215,470 (57.8) | |
| ŌĆāŌēź 65 | 418,666 (12.8) | 253,424 (11.8) | 122,584 (16.6) | 42,658 (11.4) | |
| Male, sex | 1,704,307 (52.3) | 887,683 (41.3) | 512,839 (69.5) | 303,785 (81.5) | < 0.001 |
| Socioeconomic status | < 0.001 | ||||
| ŌĆāHousehold income < 25% and medicare | 714,481 (21.9) | 499,855 (23.2) | 143,869 (19.5) | 70,757 (19.0) | |
| ŌĆāHousehold income 25ŌĆō50% | 735,696 (22.6) | 513,074 (23.9) | 146,219 (19.8) | 76,403 (20.5) | |
| ŌĆāHousehold income 50ŌĆō75% | 863,072 (26.5) | 551,264 (25.6) | 202,947 (27.5) | 108,861 (29.2) | |
| ŌĆāHousehold income Ōēź 75% | 948,189 (29.1) | 586,790 (27.3) | 244,480 (33.2) | 116,919 (31.4) | |
| Comorbidities | |||||
| ŌĆāDiabetes | 260,443 (8.0) | 103,945 (4.8) | 89,528 (12.1) | 66,970 (18.0) | < 0.001 |
| ŌĆāHypertension | 790,721 (24.2) | 371,433 (17.3) | 257,613 (34.9) | 161,675 (43.4) | < 0.001 |
| ŌĆāDyslipidemia | 560,581 (17.2) | 255,861 (11.9) | 181,802 (24.7) | 122,918 (33.0) | < 0.001 |
| CCI score | 0.8 ┬▒ 1.2 | 0.7 ┬▒ 1.2 | 0.9 ┬▒ 1.4 | 0.9 ┬▒ 1.4 | < 0.001 |
| ŌĆā0 | 1,958,875 (60.1) | 1,335,914 (62.1) | 411,636 (55.8) | 211,325 (56.7) | |
| ŌĆā1 | 682,952 (20.9) | 444,362 (20.7) | 159,976 (21.7) | 78,614 (21.1) | |
| ŌĆā2 | 328,651 (10.1) | 207,506 (9.7) | 81,125 (11.0) | 40,020 (10.7) | |
| ŌĆāŌēź 3 | 290,960 (8.9) | 163,201 (7.6) | 84,778 (11.5) | 42,981 (11.5) | |
| Body mass index (kg/m2) | 23.6 ┬▒ 3.2 | 22.2 ┬▒ 2.4 | 25.5 ┬▒ 2.2 | 28.0 ┬▒ 3.1 | < 0.001 |
| ŌĆā< 18.5 | 125,951 (3.9) | 125,073 (5.8) | 735 (0.1) | 143 (0.0) | |
| ŌĆā18.5ŌĆō22.9 | 1,303,472 (40.0) | 1,210,638 (56.3) | 82,535 (11.2) | 10,299 (2.8) | |
| ŌĆā23.0ŌĆō24.9 | 800,562 (24.6) | 540,094 (25.1) | 219,609 (29.8) | 40,859 (11) | |
| ŌĆāŌēź 25 | 1,031,453 (31.6) | 275,178 (12.8) | 434,636 (58.9) | 321,639 (86.2) | |
| Smoking | < 0.001 | ||||
| ŌĆāNever | 2,040,085 (62.6) | 1,520,572 (70.7) | 373,486 (50.6) | 146,027 (39.2) | |
| ŌĆāPast | 434,064 (13.3) | 221,788 (10.3) | 138,079 (18.7) | 74,197 (19.9) | |
| ŌĆāCurrent | 787,289 (24.1) | 408,623 (19.0) | 225,950 (30.6) | 152,716 (41) | |
| Alcohol consumption | < 0.001 | ||||
| ŌĆāNon-drinker | 1,802,203 (55.3) | 1,290,835 (60.0) | 366,670 (49.7) | 144,698 (38.8) | |
| ŌĆāMild drinkera) | 1,459,235 (44.7) | 860,148 (40.0) | 370,845 (50.3) | 228,242 (61.2) | |
| Weekly exercise of one or more times | 577,550 (17.7) | 375,757 (17.5) | 138,236 (18.7) | 63,557 (17.0) | < 0.001 |
Table┬Ā2
Incidence rates and HRs of lung cancer according to the fatty liver index
| Fatty liver index | Number of individuals | Number of events | Duration (person-years) | Incidence ratea) | Model 0b) | Model 1c) | Model 2d) | |||
|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||
| HR (95% CI) | p value | HR (95% CI) | p value | HR (95% CI) | p value | |||||
| < 30 | 2,150,983 | 16,415 | 21,854,904.2 | 0.75 | 1 (ref.) | < 0.001 | 1 (ref.) | 0.002 | 1 (ref.) | < 0.001 |
|
|
||||||||||
| 30ŌĆō59 | 737,515 | 8,043 | 7,445,660.0 | 1.08 | 1.44 (1.40ŌĆō1.48) | 0.96 (0.94ŌĆō0.99) | 1.08 (1.04ŌĆō1.11) | |||
|
|
||||||||||
| Ōēź 60 | 372,940 | 3,751 | 3,759,974.0 | 1.00 | 1.33 (1.28ŌĆō1.38) | 1.03 (0.99ŌĆō1.07) | 1.22 (1.17ŌĆō1.28) | |||
Table┬Ā3
Incidence rates and HRs of lung cancer according to the smoking status and fatty liver index
| Smoking | Fatty liver index | Number of individuals | Number of events | Duration (person-years) | Incidence ratea) | Adjusted HRb) (95% CI) | p value |
|---|---|---|---|---|---|---|---|
| Current | < 30 | 408,623 | 5,287 | 4,124,552.3 | 1.28 | 1 (ref.) | < 0.001 |
| 30ŌĆō59 | 225,950 | 2,961 | 2,281,374.9 | 1.30 | 1.05 (1.00ŌĆō1.10) | ||
| Ōēź 60 | 152,716 | 1,585 | 1,543,349.8 | 1.03 | 1.11 (1.04ŌĆō1.18) | ||
| Past | < 30 | 221,788 | 2,402 | 2,235,925.0 | 1.07 | 1 (ref.) | < 0.001 |
| 30ŌĆō59 | 138,079 | 1,737 | 1,392,782.1 | 1.25 | 1.18 (1.11ŌĆō1.25) | ||
| Ōēź 60 | 74,197 | 902 | 748,024.0 | 1.21 | 1.46 (1.34ŌĆō1.58) | ||
| Never | < 30 | 1,520,572 | 8,726 | 15,494,426.9 | 0.56 | 1 (ref.) | < 0.001 |
| 30ŌĆō59 | 373,486 | 3,345 | 3,771,503.1 | 0.89 | 1.06 (1.02ŌĆō1.11) | ||
| Ōēź 60 | 146,027 | 1,264 | 1,468,600.3 | 0.86 | 1.24 (1.16ŌĆō1.32) |
Table┬Ā4
Subgroup analysis of incidence rates and HRs of lung cancer based on the 2-year change in FLI
| FLI change | Number of individuals | Number of events | Duration (person-years) | Incidence ratea) | Model 0b) | Model 1c) | Model 2d) | |||
|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||
| HR (95% CI) | p value | HR (95% CI) | p value | HR (95% CI) | p value | |||||
| Consistently FLI Ōēź 30 | 519,598 | 4,586 | 4,282,247.70 | 1.07 | 1 (ref.) | 0.051 | 1 (ref.) | 0.097 | 1 (ref.) | < 0.001 |
|
|
||||||||||
| Improvement FLI Ōēź 30 to < 30 | 146,203 | 1,370 | 1,203,778.18 | 1.14 | 1.06 (1.00ŌĆō1.13) | 0.95 (0.89ŌĆō1.01) | 0.89 (0.83ŌĆō0.95) | |||
REFERENCES
1. Teng ML, Ng CH, Huang DQ, et al. Global incidence and prevalence of nonalcoholic fatty liver disease. Clin Mol Hepatol 2023;29(Suppl):S32ŌĆōS42.




2. Chavez-Tapia NC, Mur├║a-Beltr├Īn Gall S, Ordo├▒ez-V├Īzquez AL, Nu├▒o-Lambarri N, Vidal-Cevallos P, Uribe M. Understanding the role of metabolic syndrome as a risk factor for hepatocellular carcinoma. J Hepatocell Carcinoma 2022;9:583ŌĆō593.




3. Mantovani A, Petracca G, Beatrice G, et al. Non-alcoholic fatty liver disease and increased risk of incident extrahepatic cancers: a meta-analysis of observational cohort studies. Gut 2022;71:778ŌĆō788.


4. Park JH, Choi IS, Han KD, Park H, Kim KH, Kim JS. Association between fatty liver index and risk of breast cancer: a nationwide population-based study. Clin Breast Cancer 2020;20:e450ŌĆōe457.


5. Bj├Črkstr├Čm K, Widman L, Hagstr├Čm H. Risk of hepatic and extrahepatic cancer in NAFLD: a population-based cohort study. Liver Int 2022;42:820ŌĆō828.


6. Leiter A, Veluswamy RR, Wisnivesky JP. The global burden of lung cancer: current status and future trends. Nat Rev Clin Oncol 2023;20:624ŌĆō639.



7. Shankar A, Dubey A, Saini D, et al. Environmental and occupational determinants of lung cancer. Transl Lung Cancer Res 2019;8(Suppl 1):S31ŌĆōS49.



8. Seow WJ, Hu W, Vermeulen R, et al. Household air pollution and lung cancer in China: a review of studies in Xuanwei. Chin J Cancer 2014;33:471ŌĆō5.



9. Mu L, Liu L, Niu R, et al. Indoor air pollution and risk of lung cancer among Chinese female non-smokers. Cancer Causes Control 2013;24:439ŌĆō450.




10. Lim JU, Yoon HK. Narrative review: association between lung cancer development and ambient particulate matter in never-smokers. J Thorac Dis 2022;14:553ŌĆō563.



11. Li M, Cao SM, Dimou N, Wu L, Li JB, Yang J. Association of metabolic syndrome with risk of lung cancer: a population-based prospective cohort study. Chest 2024;165:213ŌĆō223.



12. Sin S, Lee CH, Choi SM, Han KD, Lee J. Metabolic syndrome and risk of lung cancer: an analysis of Korean National Health Insurance corporation database. J Clin Endocrinol Metab 2020;105:dgaa596.



13. National Health Insurance Service. 2023 long-term care insurance act and enforcement decree. Wonju: National Health Insurance Service, 2023.
14. Korean Statistical Information Service. Status of general health examination targets and number of examinees [Internet] Daejeon: KOSIS, c2021. [cited 2024 March 20]. Available from: https://kosis.kr/statHtml/statHtml.do?orgId=350&tblId=DT_35007_N099&vw_cd=MT_ZTITLE&list_id=350_35007_A001&scrId=&seqNo=&lang_mode=ko&obj_var_id=&itm_id=&conn_path=MT_ZTITLE&path=%252FstatisticsList%252FstatisticsListIndex.do.
15. Bedogni G, Bellentani S, Miglioli L, et al. The fatty liver index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol 2006;6:33.




16. Khang AR, Lee HW, Yi D, Kang YH, Son SM. The fatty liver index, a simple and useful predictor of metabolic syndrome: analysis of the Korea National Health and Nutrition Examination Survey 2010ŌĆō2011. Diabetes Metab Syndr Obes 2019;12:181ŌĆō190.


17. Huang X, Xu M, Chen Y, et al. Validation of the fatty liver index for nonalcoholic fatty liver disease in middle-aged and elderly Chinese. Medicine (Baltimore) 2015;94:e1682.



18. Yang MS, Park M, Back JH, et al. Validation of cancer diagnosis based on the National Health Insurance Service Database versus the National Cancer Registry Database in Korea. Cancer Res Treat 2022;54:352ŌĆō361.




19. Qiao L, Ma D, Lv H, et al. Metabolic syndrome and the incidence of lung cancer: a meta-analysis of cohort studies. Diabetol Metab Syndr 2020;12:95.




20. Mahachai N, Washirasaksiri C, Ariyakunaphan P, et al. Clinical predictive score for identifying metabolic dysfunction-associated steatotic liver disease in individuals with prediabetes using transient elastography. J Clin Med 2023;12:7617.



21. Clayton-Chubb D, Kemp WW, Majeed A, et al. Metabolic dysfunction-associated steatotic liver disease in older adults is associated with frailty and social disadvantage. Liver Int 2024;44:39ŌĆō51.


22. Mendon├¦a FM, de Sousa FR, Barbosa AL, et al. Metabolic syndrome and risk of cancer: which link? Metabolism 2015;64:182ŌĆō189.


23. Margini C, Dufour JF. The story of HCC in NAFLD: from epidemiology, across pathogenesis, to prevention and treatment. Liver Int 2016;36:317ŌĆō324.

24. Greten FR, Grivennikov SI. Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 2019;51:27ŌĆō41.



25. LoConte NK, Gershenwald JE, Thomson CA, Crane TE, Harmon GE, Rechis R. Lifestyle modifications and policy implications for primary and secondary cancer prevention: diet, exercise, sun safety, and alcohol reduction. Am Soc Clin Oncol Educ Book 2018;38:88ŌĆō100.


26. Younossi ZM, Zelber-Sagi S, Henry L, Gerber LH. Lifestyle interventions in nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol 2023;20:708ŌĆō722.



27. Wong VW, Chan RS, Wong GL, et al. Community-based lifestyle modification programme for non-alcoholic fatty liver disease: a randomized controlled trial. J Hepatol 2013;59:536ŌĆō542.


28. Santos MJ, Vinagre F, Canas da Silva J, Gil V, Fonseca JE. Body composition phenotypes in systemic lupus erythematosus and rheumatoid arthritis: a comparative study of Caucasian female patients. Clin Exp Rheumatol 2011;29:470ŌĆō476.

29. Sabatino A, Cuppari L, Stenvinkel P, Lindholm B, Avesani CM. Sarcopenia in chronic kidney disease: what have we learned so far? J Nephrol 2021;34:1347ŌĆō1372.




30. Stenvinkel P, Carrero JJ, von Walden F, Ikizler TA, Nader GA. Muscle wasting in end-stage renal disease promulgates premature death: established, emerging and potential novel treatment strategies. Nephrol Dial Transplant 2016;31:1070ŌĆō1077.


31. An HJ, Tizaoui K, Terrazzino S, et al. Sarcopenia in autoimmune and rheumatic diseases: a comprehensive review. Int J Mol Sci 2020;21:5678.



32. White MC, Holman DM, Boehm JE, Peipins LA, Grossman M, Henley SJ. Age and cancer risk: a potentially modifiable relationship. Am J Prev Med 2014;46(3 Suppl 1):S7ŌĆōS15.


33. Luo J, Chlebowski RT, Hendryx M, et al. Intentional weight loss and endometrial cancer risk. J Clin Oncol 2017;35:1189ŌĆō1193.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement table 1
Supplement table 1 Print
Print



