 |
 |
|
|
|
Abstract
Background/Aims
This study aims to evaluate the incidence of malignancy in patients with rheumatoid arthritis (RA) and to investigate risk factors for such in a nationwide, population-based cohort.
Methods
In a large, prospective, observational cohort study, 5,077 patients with RA were enrolled from July 2009 to December 2011 and followed until February 2017. Standardized incidence ratios (SIRs) for malignancy were calculated using age- and sex-specific cancer rates in the Korean general population. Poisson regression was used to identify the risk of incident malignancy.
Results
The cohort included 5,023 participants with RA contributing 16,689 person-years of follow-up. A total of 148 malignancies were recorded. The risks of stomach cancer (SIR, 0.41; 95% confidence interval [CI], 0.21 to 0.74), colon cancer (SIR, 0.13; 95% CI, 0.03 to 0.37), and lung cancer (SIR, 0.35; 95% CI, 0.14 to 0.72) were lower in RA patients than in the general population. Poisson regression modeling demonstrated that the malignancy risk was more than two-fold greater in patients with thyroid disease than in those without thyroid disease. Hydroxychloroquine therapy was associated with a reduced risk (relative risk, 0.39; 95% CI, 0.189 to 0.801) of malignancy development.
Conclusions
The overall risk of malignancy in patients with RA is decreased relative to in the general population. In particular, stomach, colon, and lung cancers in Korean RA patients are less common, while brain and central nervous system cancers in male RA patients are more frequent. The patients with thyroid disease and longer RA disease duration were at increased risk for developing malignancy, while hydroxychloroquine users were at lower risk.
Rheumatoid arthritis (RA) is a chronic systemic disease characterized by joint inflammation resulting in articular destruction, requiring long-term treatment. RA is also associated with comorbidities and mortalities such as cardiovascular disease, infectious disease, osteoporosis, and malignancy.
The risk of malignancy is of considerable interest in RA patients because of its immunologic pathogenesis and immune therapeutic targeting in disease management using disease-modified antirheumatic drugs (DMARDs). A meta-analysis of the incidence of malignancy in RA as compared with in the general population [1] found that RA patients appear to be at increased risk for both lymphoma and lung cancer [2] but at lower risk for colorectal and breast cancers [3,4]. However, the findings of previous studies assessing the malignancy risk in RA patients are not consistent and there are disagreements demographically (e.g., age, sex), geographically, and nationally or according to variable definitions of cancer outcome. In China, the prevalence of malignancy within the Chinese Registry of Rheumatoid Arthritis (CREDIT) cohort was 0.6% and breast and lung cancers were the most common cancers [5]. The factors associated with malignancy included advanced age and methotrexate (MTX) treatment in the CREDIT cohort. However, CORRONA, a United States-based registry of RA, revealed that the overall incidence of malignancy was 0.56 per 100 person-years (PY) and non-melanoma skin cancer was the most common type of cancer [6]. In a comparative study of real-world RA populations [7], the age-/sex-standardized incidence rates of malignancy per 100 PY in SRR (a Swedish registry) and NOAR (a United Kingdom registry) were 0.87 and 0.77, respectively.
Meanwhile, the occurrence of malignancy among patients with RA in South Korea has not been thoroughly investigated in a nationwide-based population study, despite the importance of understanding the underlying malignancy risk among patients with RA.
Therefore, we aimed to investigate the incidence and risk factors of malignancy in patients with RA using a large observational cohort, the Korean Observational Study Network for Arthritis (KORONA), which was conducted in the Clinical Research Center for Rheumatoid Arthritis involving 23 centers across South Korea [8].
KORONA was established in July 2009 by the Clinical Research Center for Rheumatoid Arthritis, involving 23 centers across South Korea funded by the Ministry of Health and Affairs, South Korea. This cohort has been described in detail previously [8]. RA patients over the age of 18 years who satisfied the 1987 American Rheumatoid Arthritis Society criteria for RA [9] were enrolled into the KORONA cohort from 2009 to 2011 after informed consent was given and annually followed up with, thus representing the largest nationwide multicenter prospective observational RA cohort. Among 5,077 RA patients enrolled in the KORONA cohort, 5,023 patients were eligible for inclusion in this study, while 54 patients were excluded due to prior malignancy before the diagnosis of RA and one patient was excluded for missing data regarding the diagnosis date of malignancy (Fig. 1).
For the analysis of malignancy development, patients with fewer than two visits during the follow-up period were excluded. This study protocol was approved by the Institutional Review Board (IRB) of Uijeongbu St. Mary’s hospital (IRB No. UC20ECDI0051). The KORONA protocol was approved by IRB of all participating hospitals and informed consent was obtained from all participants prior to study enrollment.
Baseline data on demographic details, socioeconomic status, education level, income, disease-specific variables, self-reported past and current medical histories, and comorbidities were collected using an initial questionnaire provided to all enrolled patients. RA-related information such as age at RA diagnosis; disease duration; notable laboratory findings including rheumatoid factor (RF) status, anticitrullinated protein antibody, and inflammatory markers was gathered. Rheumatologists or well-trained health professionals performed a joint assessment and details of medication use were obtained by medical chart review. The Disease Activity Score 28 (DAS-28), the Korean version of the Health Assessment Questionnaire (HAQ), and the EuroQol five-dimensional questionnaire (EQ5D) questionnaire were deployed to ascertain study participant level of physical disability and disease activity at baseline. In a first analysis, we evaluated the gathered data in a cross-sectional manner to calculate the prevalence rate of malignancy at enrollment. A second analysis was then performed by investigating the data from a longitudinal perspective to obtain the incidence of malignancy during the follow-up period.
We observed patients from enrollment to their last annual follow-up visit or the occurrence of malignancy. Cases of newly developed malignancy in the KORONA cohort were identified by patient reported-questionnaires, which included questions on malignancy type and date of diagnosis annually during the follow-up period. We did not include all benign tumors or prior malignancy cases before RA diagnosis in this study.
The Shapiro-Wilk test was used to analyze variables with a normal distribution, and nonparametric tests were used for data that were not normally distributed. Continuous values were expressed as the median (interquartile range [IQR]) and categorical values as numbers (%). Demographic and clinical characteristics were compared between the malignancy group and non-malignancy group using the chi-square test or Fisher’s exact test and Student’s t test or Wilcoxon rank-sum test. PY for each patient were calculated by subtracting the dates from the date of enrollment in KORONA until the date of last annual follow-up or the date of malignancy reported. Standardized incidence ratios (SIRs) were used to compare the incidence of malignancy in the KORONA cohort with that in the general population using site, age, sex, and calendar year-specific incidence rates as obtained from Statistics Korea (http://kosis.kr) [10]. The SIR was calculated as follows: the number of observed malignancy cases in the KORONA cohort divided by the expected number of malignancy cases according to national age- and sex-specific malignancy rates. The 95% confidence interval (CI) was calculated by Poisson distribution. SIRs for each malignancy type were calculated separately for men and women as they differed in their malignancy risks. Multivariate Poisson regression modeling for the relative risk (RR) of malignancy with the following baseline characteristics was used in the KORONA cohort: RA disease duration; disease activity measured using DAS-28; physical function measured using HAQ; prior or current exposure to DMARDs, nonsteroidal anti-inflammatory drugs (NSAIDs), and/or corticosteroids; and smoking status. Our final model was adjusted for age, sex, disease duration, disease activity measures, smoking alcohol use, body mass index (BMI), and family history of malignancy. The variables used in the multivariable model were those with a p value of less than 0.05 in the crude model, as well as some others of clinical importance, and p values less than 0.05 were considered to indicate statistical significance. All analyses were carried out using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
The study cohort consisted of 5,023 patients with RA contributing 16,689 PY of follow-up to the analysis. The cohort was predominantly female (85.5%), with a median age at RA diagnosis of 46 years (IQR, 37 to 55) and at disease onset of 43 years (IQR, 34 to 52) (Table 1). The median disease duration was 6 years (IQR, 2 to 12) with moderate disease activity (median DAS-28 score, 3.67; IQR, 2.72 to 4.59) at baseline (Table 2). At the baseline of this study, the most frequent DMARD used by the cohort was MTX (n = 3,476; 83.9%) and the median dose was 12.5 mg per week (Table 2). The frequency of biologic DMARD usage was 6.4% (n = 263) at baseline, with prior biologic DMARD users composing 14.8% (n = 240) of the study population, and the median duration of biologic DMARDs was 3 months (IQR, 2 to 12) (Table 2). The proportion of patients with positive RF status was 75%, while the median EQ5D and median HAQ scores were 0.73 (IQR, 0.59 to 0.80) and 0.5 (IQR, 0.13 to 1.0), respectively.
When patients with malignancy were compared to those without, they were more often male (16.8% vs. 14.4%, p = 0.421) but not to a statistically significant degree (Table 1). However, they showed significant difference in RA disease duration at the time of diagnosis (median: 8 years vs. 6 years, p < 0.005) (Table 2) and lower BMI (21.4 vs. 22.5, p = 0.022) (Table 1). Malignancy patients showed less RF positivity (81.1% vs. 87%, p = 0.041) and were less often taking hydroxychloroquine (HCQ) (21% vs. 34.6%, p < 0.001), while greater corticosteroid usage was reported (81.1% vs. 73.7%, p = 0.046) (Table 2). Thyroid disease among the comorbidities identified at enrollment was more frequently reported in patients with malignancy (16.8% vs. 6.8%, p < 0.001). Comorbidity, medication use, and other demographic data are summarized in Tables 1 and 2.
Among the 5,077 RA patients enrolled in the KORONA cohort, we excluded 54 patients who were diagnosed with malignancy before their diagnosis of RA and one patient for missing data concerning the diagnosis date of malignancy. However, five patients among these 64 patients developed a second malignancy during the follow-up period. One of five patients was diagnosed with double primary cancer and the others were diagnosed with recurrent cancers. We identified 103 patients with malignancy (2.05%) among a total of 5,023 patients (prevalence rate: 2,050.6/100,000) during cross-sectional analysis. Most female patients had thyroid cancer (40.9%) and breast cancer (17.1%), while stomach cancer (47.1%) was the most prevalent cancer among male patients with RA. Fig. 2 summarizes the frequency of detected cancer subtypes by sex.
A total of 64 of the 4,054 patients (1.58%) at risk for new malignancy were diagnosed during the cumulative follow-up period of 16,688 PY (men: 2,317 PY; women: 14,371 PY). We estimated a crude incidence rate of 383.5/100,000 PY (men: 561.0/100,000 PY; women: 354.9/100,000 PY).
The 64 malignancies (13 men, 51 women) were found to equate to a crude incidence rate of 3.8/1,000 PY. Overall, the risk of malignancy was decreased in our RA cohort as compared with in the general population (SIR, 0.40; 95% CI, 0.31 to 0.51) (Table 3). The RRs were decreased in gastric, colon, and lung cancer (SIR, 0.41; 95% CI, 0.21 to 0.74; SIR, 0.13; 95% CI, 0.03 to 0.37, and SIR, 0.35; 95% CI, 0.14 to 0.72) but increased in brain and central nervous system cancers in male patients (SIR, 10.84; 95% CI, 1.22 to 39.14) (Table 3).
To identify the risk factors associated with the occurrence of malignancy, we performed a multivariate analysis using baseline data collected at first enrollment in the KORONA cohort. During multivariate analysis, thyroid disease and RA disease duration were associated with incident malignancy (RR, 2.82; 95% CI, 1.79 to 4.44 and RR, 1.64; 95% CI, 1.11 to 2.42), while the use of HCQ at enrollment was associated with a reduced risk (RR, 0.50; 95% CI, 0.32 to 0.78) of new-onset malignancy (Table 4). Age at RA diagnosis, sex, smoking, and disease activity were not identified as risk factors.
In this prospective cohort study of 5,023 patients with RA assessing the risk for developing malignancy, we provide several key insights into the current prevalence, incidence, and risk factors. First, we identified 103 malignancy patients, corresponding to a prevalence rate of 2.05%, during cross-sectional analysis. Second, we observed 64 newly developed malignancies during the follow-up period. Notably, the risks of stomach, colon, and lung cancers were decreased by 0.4−, 0.13−, and 0.35-fold, respectively. Third, we identified thyroid disease and RA disease duration as risk factors and HCQ use as a possible preventive factor in developing malignancy. This is the first study analyzing the prevalence, incidence, and risk factors of malignancy in a large observational prospective RA cohort in Korea.
In addition, we report that the SIR for overall malignancy in the RA cohort was decreased relative to that in the general population. Although, in one recent meta-analysis, the overall standardized incidence rate for overall malignancy was 10% increased in RA patients [4] as compared with in the general population, individual studies have offered more inconsistent results. Some studies reported that the overall risk of malignancy was similar to that of the general population [11–13], while others confirmed less overall malignancy risk among patients with RA than the general population [4,14,15]. From Korea, only two papers have been published concerning the malignancy risk in RA patients, with different results [12,16]. Lee [16] demonstrated that the overall cancer incidence among Korean RA patients was increased, while Kim et al. [12] showed that the incidence rate for cancer was not significantly increased from that of the general population. Both studies had limitations, having been performed in a single center with a limited information and short observational period. In contrast, the present study is the first large-scale nationwide investigation to estimate the risk of malignancy in a population of Korean patients with RA with no history of malignancy.
In previous studies from Western countries including meta-analyses [1,4], the overall risk for malignancy was not consistent in patients with RA, but the risk for site-specific malignancy was significantly varied from that in the general population. These included an increased risk of lymphoma [17–19] and lung cancer [4,18] and a reduced risk of colorectal and breast cancers [1,20]. In this study, during the follow-up period, one lymphoma case was observed in a male RA patient and seven lung cancers (four men, three women) were identified. The SIR for lymphoma and lung cancers was not increased in RA patients, inconsistent with the findings of previous studies. Smoking is a well-known independent risk factor for lung cancer and interstitial lung disease (ILD) would thus be a potential risk factor for lung cancer [21,22]. In the KORONA cohort, the majority of participants had never smoked (84.5%) and the proportion of patients with ILD was only 0.8%. Given that most studies to date have been conducted in Western countries, the lower smoking rate in the KORONA cohort is characteristic of Korean RA patients. Therefore, our results are likely to be linked to the low smoking and ILD rates in this study cohort.
Regarding site-specific malignancy, the incidence rates of stomach cancer (SIR, 0.41; 95% CI, 0.21 to 0.74) and colon cancer (SIR, 0.13; 95% CI, 0.03 to 0.37) were significantly decreased in RA patients as compared with in the general population in this study. In a recent meta-analysis [4] and previous epidemiological studies [11,15,20,23], colorectal cancer displayed a decreased risk in RA patients as compared with in the general population, which is similar to as found in this study. It has been well noted that NSAIDs and aspirin reduce the risk of colorectal cancer [24]. In one meta-analysis [25], anti-inflammatory drug intake, especially cyclooxygenase (COX)-2 inhibitors, was associated with a statistically significant reduction in the risk of gastric cancer. NSAIDs inhibit the cyclooxygenase enzyme (COX-1 or COX-2) involved in the synthesis of prostaglandin, which are elevated in colorectal cancer. RA patients mostly use NSAIDs to reduce inflammation, which may impart a protective effect against the development of colorectal cancer.
The incidence of gastric cancer in patients with RA appears to be inconsistent. A Japanese cohort showed a lower risk for stomach cancer than that in the general population [26], while a Taiwanese cohort showed an increased risk for stomach cancer (SIR, 1.26; 95% CI, 1.22 to 1.29) [27]. In this study, the risk of stomach cancer with RA was decreased (SIR, 0.41; 95% CI, 0.21 to 0.74). There are significant known regional differences in gastric cancer incidence and mortality. Gastric cancer rates are significantly increased in East Asia, including in Korea [28], and Helicobacter pylori infection is one of the risk factors for gastric cancer as a carcinogen in Korea [29]. A recent nationwide multicenter study in Korea [30] reported a downward trend in H. pylori seroprevalence and an increase in H. pylori eradication, which could affect development of gastric cancer. In addition, RA patients tend to receive regular endoscopies due to their high gastrointestinal bleeding risk, leading to a greater chance to eradicate H. pylori in this group than in the general population.
In our study, HCQ was indicated to be a potential protective factor against developing malignancy in RA patients. HCQ is usually used as component of a triple-drug combination involving MTX and sulfasalazine as well for RA treatment and is also widely used in the treatment of systemic lupus erythematous (SLE). As suggested in previous research [31,32], HCQ treatment in patients with SLE could reduce the risk of malignancy. It is also suggested that HCQ might have a role in preventing neoplasia in SLE. In our study, HCQ may have played a protective role against developing malignancy in RA patients, in contrast with a previous study [33]. Interest has grown in highlighting the antitumoral effects of HCQ and chloroquine, which may be related to strong antiproliferative, antimutagenic, and lysosomotropic-autophagy inhibition effects [34,35]. However, given that HCQ monotherapy was found only in nonmalignant group and HCQ monotherapy is generally used for patients with low disease activity, it seems difficult to rule out the possibility that malignant group might have higher disease activity than nonmalignant group. Although statistically insignificant between two groups, it also raises a possibility that malignant group with higher disease activity have been treated with leflunomide, corticosteroids, or biologic DMARDs more frequently than nonmalignant group, resulting in similar disease activity.
The malignant group also tends to have higher monthly income than the nonmalignant group even though it is not statistically significant. Considering the fact that the most common diagnosed cancer is thyroid cancer, it is possible the malignant group with higher income levels could increase their chance of diagnosing cancer by accessing medical services, particularly in preventive health services, such as cancer screening.
Some limitations of this study should be mentioned. Precision in some of the analyses, particularly in the assessment of specific cancer types, was limited because the occurrence of malignancy was obtained through interviews and based on patient self-reports. Therefore, the existence of missing cases or inaccurate patient reports cannot be ruled out. Another limitation is selection bias due to some patients being lost to follow-up. Also, some patients may have been unable to visit the clinic regularly because their RA was exacerbated or comorbidities such as malignancy had worsened. This bias may be associated with an underestimation of malignancy incidence.
Still, our results suggest that the underlying disease independently informs the risk of malignancy. Indeed, we found that thyroid disease increased the risk of malignancy by 2.5-fold among patients with RA. Thyroid disease at baseline was demonstrated to be a risk factor for malignancy in RA patients in our analysis. Recently, Li et al. [36] reported that the risk of thyroid dysfunction was increased among RA patients. In an Asian population, thyroid disorder increased the risk of breast cancer [37]. In real practice, thyroid function testing is part of the regular medical checkup for RA patients. This might be one of the reasons for increased thyroid disease in RA patients.
No association was found between the incidence of overall malignancy and baseline measurements of disease severity, physical dysfunction, or medication use. Presumably, these baseline data reflect characteristics years after diagnosis and would change with subsequent treatment.
The greatest strength of this study is its use of real-world clinical data obtained from a large, observational cohort study on RA with a long duration of follow-up, thus providing more precise estimates of the malignancy risk in RA patients.
In conclusion, the overall incidence of malignancy in Korean patients with RA was demonstrated to be decreased as compared with in the general Korean population. Regarding site-specific cancer, the risk of stomach and colon cancers was decreased.
1. Our study shows that the prevalence and incidence rates of malignancy in patients with rheumatoid arthritis (RA) in Korea were decreased as compared with in general population.
2. The patients with thyroid disease and longer RA disease duration are at increased risk of developing malignancy, while hydroxychloroquine users are at lower risk.
Conflict of Interest
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Acknowledgments
The authors acknowledged the contributions from the Korean Observational Study Network for Arthritis (KORONA) team located all over South Korea. The authors wish to acknowledge the financial support of the Catholic Medical Center Research Foundation made in the program year of 2015 and The Catholic University of Korea Uijeongbu, St. Mary’s Hospital Clinical Trials Center, provided in the program year of 2020.
Figure 1
Inclusion and exclusion criteria of patients with rheumatoid arthritis (RA) in the present study. KORONA, Korean Observational Study Network for Arthritis.

Figure 2
Frequency of cancer-type in the cross-sectional analysis. (A) Women (n = 88). (B) Men (n = 17).
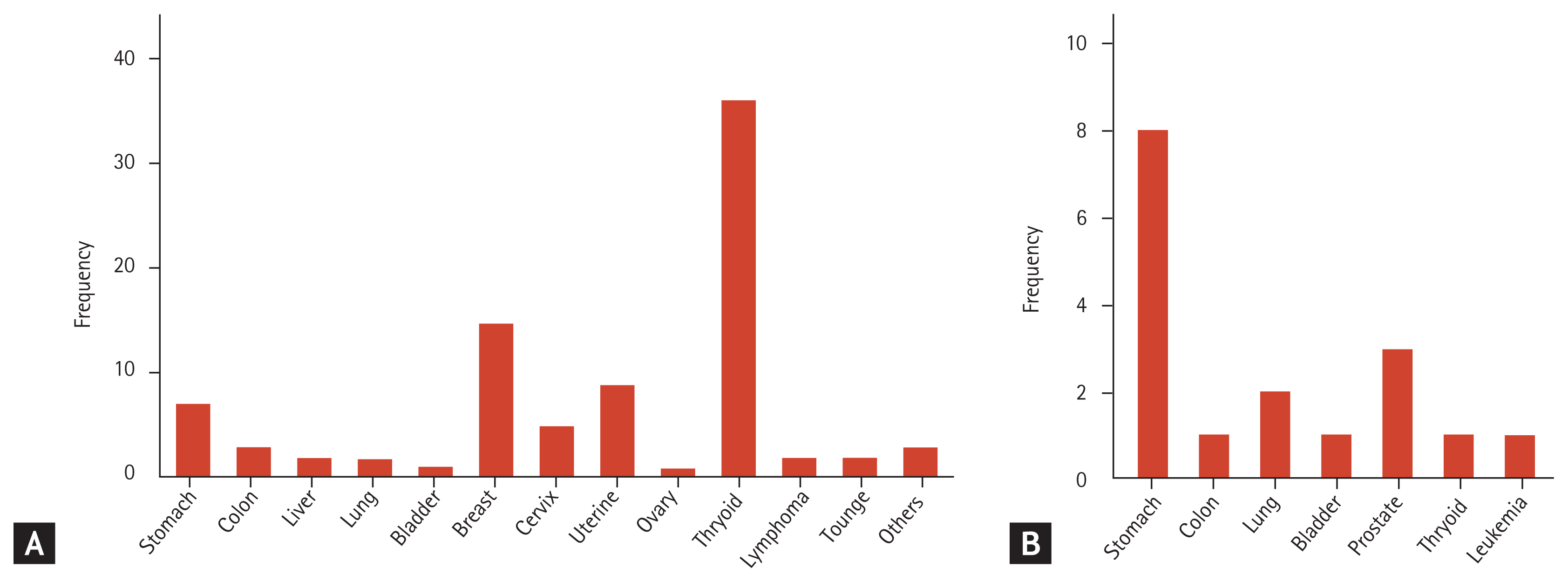
Table 1
Baseline characteristics of the KORONA cohort
Table 2
Comparison of RA patients with malignancy and without malignancy in the KORONA cohort
RA, rheumatoid arthritis; KORONA, Korean Observational Study Network for Arthritis; ACR, American College of Rheumatology; PIP, proximal interphalangeal; MCP, metacarpal phalangeal; ACPA, anti-citrullinated protein antibody; DAS-28, Disease Activity Score 28; HAQ, Health Assessment Questionnaire; EQ5D, EuroQol five-dimensional questionnaire; ESR, erythrocyte sediment rate; CRP, C-reactive protein; COPD, chronic obstructive pulmonary disease; NSAID, nonsteroidal anti-inflammatory drug; bDMARD, biologic disease-modified antirheumatic drug; IL-17, interleukin 17.
Table 3
SIRs of specific malignancy types stratified by sex in patients with RA
Table 4
Poisson regression models estimating the malignancy risk in RA (n = 4,143)
REFERENCES
1. Smitten AL, Simon TA, Hochberg MC, Suissa S. A meta-analysis of the incidence of malignancy in adult patients with rheumatoid arthritis. Arthritis Res Ther 2008;10:R45.



2. Kauppi M, Pukkala E, Isomaki H. Excess risk of lung cancer in patients with rheumatoid arthritis. J Rheumatol 1996;23:1484–1485.

3. Kauppi M, Pukkala E, Isomaki H. Low incidence of colorectal cancer in patients with rheumatoid arthritis. Clin Exp Rheumatol 1996;14:551–553.

4. Simon TA, Thompson A, Gandhi KK, Hochberg MC, Suissa S. Incidence of malignancy in adult patients with rheumatoid arthritis: a meta-analysis. Arthritis Res Ther 2015;17:212.



5. Jin S, Li M, Fang Y, et al. Chinese registry of rheumatoid arthritis (CREDIT): II. Prevalence and risk factors of major comorbidities in Chinese patients with rheumatoid arthritis. Arthritis Res Ther 2017;19:251.




6. Gross RL, Schwartzman-Morris JS, Krathen M, et al. A comparison of the malignancy incidence among patients with psoriatic arthritis and patients with rheumatoid arthritis in a large US cohort. Arthritis Rheumatol 2014;66:1472–1481.



7. Askling J, Berglind N, Franzen S, et al. How comparable are rates of malignancies in patients with rheumatoid arthritis across the world? A comparison of cancer rates, and means to optimise their comparability, in five RA registries. Ann Rheum Dis 2016;75:1789–1796.


8. Sung YK, Cho SK, Choi CB, et al. Korean Observational Study Network for Arthritis (KORONA): establishment of a prospective multicenter cohort for rheumatoid arthritis in South Korea. Semin Arthritis Rheum 2012;41:745–751.


9. Arnett FC, Edworthy SM, Bloch DA, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988;31:315–324.


10. Korean Statistical Information Service. Cancer Registration Statistics [Internet] Daejeon (KR): Statistics Korea, 2013. [cited 2021 Jul 20]. Available from: http://kosis.kr
.
11. Huang WK, Chiou MJ, Kuo CF, Lin YC, Yu KH, See LC. No overall increased risk of cancer in patients with rheumatoid arthritis: a nationwide dynamic cohort study in Taiwan. Rheumatol Int 2014;34:1379–1386.


12. Kim YJ, Shim JS, Choi CB, Bae SC. Mortality and incidence of malignancy in Korean patients with rheumatoid arthritis. J Rheumatol 2012;39:226–232.


13. Franklin J, Lunt M, Bunn D, Symmons D, Silman A. Influence of inflammatory polyarthritis on cancer incidence and survival: results from a community-based prospective study. Arthritis Rheum 2007;56:790–798.


14. Harigai M, Nanki T, Koike R, et al. Risk for malignancy in rheumatoid arthritis patients treated with biological disease-modifying antirheumatic drugs compared to the general population: a nationwide cohort study in Japan. Mod Rheumatol 2016;26:642–650.


15. Cibere J, Sibley J, Haga M. Rheumatoid arthritis and the risk of malignancy. Arthritis Rheum 1997;40:1580–1586.


16. Lee H. The risk of malignancy in Korean patients with rheumatoid arthritis. Yonsei Med J 2019;60:223–229.



17. Hellgren K, Baecklund E, Backlin C, Sundstrom C, Smedby KE, Askling J. Rheumatoid arthritis and risk of malignant lymphoma: is the risk still increased? Arthritis Rheumatol 2017;69:700–708.


18. Lim XR, Xiang W, Tan JWL, et al. Incidence and patterns of malignancies in a multi-ethnic cohort of rheumatoid arthritis patients. Int J Rheum Dis 2019;22:1679–1685.


19. Parikh-Patel A, White RH, Allen M, Cress R. Risk of cancer among rheumatoid arthritis patients in California. Cancer Causes Control 2009;20:1001–1010.



20. Yamada T, Nakajima A, Inoue E, et al. Incidence of malignancy in Japanese patients with rheumatoid arthritis. Rheumatol Int 2011;31:1487–1492.


21. Jung HI, Park JS, Lee MY, et al. Prevalence of lung cancer in patients with interstitial lung disease is higher than in those with chronic obstructive pulmonary disease. Medicine (Baltimore) 2018;97:e0071.



22. Naccache JM, Gibiot Q, Monnet I, et al. Lung cancer and interstitial lung disease: a literature review. J Thorac Dis 2018;10:3829–3844.



23. Gridley G, McLaughlin JK, Ekbom A, et al. Incidence of cancer among patients with rheumatoid arthritis. J Natl Cancer Inst 1993;85:307–311.


24. Tomic T, Dominguez-Lopez S, Barrios-Rodriguez R. Non-aspirin non-steroidal anti-inflammatory drugs in prevention of colorectal cancer in people aged 40 or older: a systematic review and meta-analysis. Cancer Epidemiol 2019;58:52–62.


25. Kong P, Wu R, Liu X, et al. The effects of anti-inflammatory drug treatment in gastric cancer prevention: an update of a meta-analysis. J Cancer 2016;7:2247–2257.



26. Hashimoto A, Chiba N, Tsuno H, et al. Incidence of malignancy and the risk of lymphoma in Japanese patients with rheumatoid arthritis compared to the general population. J Rheumatol 2015;42:564–571.


27. Chen YJ, Chang YT, Wang CB, Wu CY. The risk of cancer in patients with rheumatoid arthritis: a nationwide cohort study in Taiwan. Arthritis Rheum 2011;63:352–358.


28. Balakrishnan M, George R, Sharma A, Graham DY. Changing trends in stomach cancer throughout the world. Curr Gastroenterol Rep 2017;19:36.



29. Peleteiro B, Bastos A, Ferro A, Lunet N. Prevalence of Helicobacter pylori infection worldwide: a systematic review of studies with national coverage. Dig Dis Sci 2014;59:1698–1709.


30. Lim SH, Kim N, Kwon JW, et al. Trends in the seroprevalence of Helicobacter pylori infection and its putative eradication rate over 18 years in Korea: a cross-sectional nationwide multicenter study. PLoS One 2018;13:e0204762.

31. Lee SJ, Silverman E, Bargman JM. The role of antimalarial agents in the treatment of SLE and lupus nephritis. Nat Rev Nephrol 2011;7:718–729.


32. Ruiz-Irastorza G, Ugarte A, Egurbide MV, et al. Antimalarials may influence the risk of malignancy in systemic lupus erythematosus. Ann Rheum Dis 2007;66:815–817.



33. Bernatsky S, Clarke A, Suissa S. Antimalarial drugs and malignancy: no evidence of a protective effect in rheumatoid arthritis. Ann Rheum Dis 2008;67:277–278.


34. Plantone D, Koudriavtseva T. Current and future use of chloroquine and hydroxychloroquine in infectious, immune, neoplastic, and neurological diseases: a mini-review. Clin Drug Investig 2018;38:653–671.


35. Shi TT, Yu XX, Yan LJ, Xiao HT. Research progress of hydroxychloroquine and autophagy inhibitors on cancer. Cancer Chemother Pharmacol 2017;79:287–294.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



