 |
 |
| Korean J Intern Med > Volume 16(2); 2001 > Article |
|
Abstract
Objectives:
Primary pulmonary hypertension (PPH) that affects predominantly young and productive people is a progressive fatal disease of unknown cause. The objectives of this study were to characterize mortality in patients with PPH and to investigate the factors associated with their survival.
Methods:
Thirteen patients with PPH were enrolled between 1988 and 1996 and followed-up through July 1999. Measurements at diagnosis included hemodynamic and pulmonary function variables in addition to information on demographic data and medical history.
Primary pulmonary hypertension (PPH) is an uncommon disease that affects predominantly young and productive people1–3). Although important conceptual advances and information about the pathogenesis, histopathology, characterization and management of PPH have been generated in the past decade, many questions remain to be answered. Part of the reason for the lack of complete knowledge in this regard is a rare entity and a short mean survival of 2 to 4 years2–5). Patients with a survival period > 5 to 10 years have also been well documented5–9) and even spontaneous regression of the disease, although uncommon, has been described10–11). We analyzed determinants associated with the survival and prognosis of patients with PPH.
The study series included 13 patients in whom PPH was diagnosed at our institution between 1988 and 1996 and who were followed-up through July 1999. As in other studies, the PPH was diagnosed by a thorough workup, including clinical history, physical examination, laboratory tests, chest radiography, ECG, pulmonary function tests, echocardiography, radionuclide perfusion lung scan and cardiac catheterization. Criteria used to establish the diagnosis of PPH include a mean pulmonary arterial pressure of more than 25 mmHg at rest or 30 mmHg during exercise, a normal pulmonary capillary wedge pressure (PCWP) and absence of other disease known to cause or to be associated with secondary pulmonary hypertension12). Particular care was taken to exclude patients with evidence of congenital heart disease or acquired vascular or myocardial disease, obstructive or restrictive lung disease, or both, parasitic disease involving the lung, pulmonary thromboembolic and clearly defined collagen vascular disease and the antiphospholipid syndrome.
Cardiac output and cardiac index were measured by the thermodilution method and pulmonary artery pressure, PCWP, right atrial pressure, right ventricular pressure and oxygen saturation were obtained by a Swan-Ganz catheter. The radial artery was also cannulated with a needle for arterial oxygen blood sampling. We did not evaluate the hemodynamic data after taking a vasodilator drug during cardiac catheterization.
Some patients in this study had been taking a vasodilator and diuretics. All patients were not treated with coumadin.
For the survival analysis, we used the initial clinical and hemodynamic characteristics by cardiac catheterization as an index for determining survival. All values are expressed as mean ± SD. The Kaplan-Meier method was used to estimate overall survival distribution and Cox proportional hazards model was used to examine the relation between survival and variables. A p value <0.05 was significant in all analysis.
The mean age of the patients with PPH entered into the study was 36.1 ± 9.3 years and women were predominant (female/male ratio 1.75:1). None of the patients had histories of appetite suppressant drug use and 3 were cigarette smokers. Of the women of reproductive age, one had taken oral contraceptive and none had a family history of familial pulmonary hypertension. The frequency of symptoms at diagnosis was dyspnea 100%, syncope 2 (16.7%), chest pain 2 (16.7%), effort related palpitation 1 (8.3%), hoarseness 4 (30.7%) and leg edema 3 (23.1%). The functional status of the patients at diagnosis according to the New York Heart Association (NYHA) classification was as follows; 8.3% of the patients were in class I, 41.7% in class II, 41.7% in class III and 16.7% in class IV (Table 1). Mean time from onset of the first symptom to diagnosis of PPH was 4.2 years (range 0 to 9.6).
The chest radiograph showed the typical changes associated with pulmonary hypertension, namely prominence of the main pulmonary artery in all patients and enlarged hilar vessels. The mean cardiothoracic ratio was 0.6. The ECG showed a sinus rhythm and evidence of right ventricular hypertrophy in all cases. The echocardiogram confirmed right ventricular hypertrophy and showed variable degrees of right ventricular enlargement. A lung perfusion scan was performed in 8 patients and was considered to be normal in 4 (50%) and showed diffuse bilateral patchy pattern in 4 (50%). The mean hemoglobin and hematocrit levels were 13.9 ± 5 g/dL and 41.7 ± 4.5%, respectively. The total platelet count was normal. The antinuclear antibody test was negative in all cases.
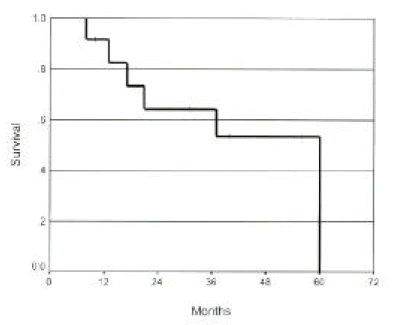
As of July 1999, 7 of the 13 PPH patients have died, and the mean survival time of this group was 3.4 ± 0.6 years. Among the 7 patients one survived <1 year, 3 < 2 years and 3 > 3 years (Figure 1). Mortality was not associated with age, sex, functional status (NYHA) and pulmonary function test. Only cardiac index was associated with the prognosis (Table 3).
PPH was first described over 100 years ago in a patient with right-heart failure whose necropsy showed no obvious reason for pulmonary arteriosclerosis13). PPH is clinically defined as a mean pulmonary arterial pressure of more than 25 mmHg at rest or 30 mmHg during exercise without evidence of secondary pulmonary hypertension14–15). The estimated annual incidence of PPH in European and US studies is 1–2 cases per one million people per year in the general population, and necropsy studies have shown a prevalence of 1300 per million16). The incidence of PPH among users of appetite suppressants may be as high as 25–50 per million per year17). The mean age at diagnosis of PPH is 36 years and the female is means predominant (1.7–3.5) than the male18). Familial PPH accounts for roughly 10% of cases. We had similar results showing the mean age was 36 years and the female was in excess (1.75) of the male. The pathogenesis of PPH envisage individual susceptibility, triggering a stimulus as the initiating factor for pulmonary vascular injury and repair, and genetic susceptibility in case of familial PPH. The stimuli that trigger PPH are drugs such as appetite suppressants, infections particularly HIV-1, and inflammatory disorders. Circulating vasoactive mediators (serotonin) may play a part in pulmonary hypertension19). The earliest symptom in most cases of PPH is the gradual onset of shortness of breath after physical exertion. Other symptoms include chest pain, syncope, fatigue, leg edema and hoarseness. We found 4 cases (30.7%) of PPH whose initial symptom was hoarseness. So, in the case of patients with hoarseness, PPH should be considered a differential diagnosis. The signs of PPH are an accentuated second heart sound in the pulmonary region, and a right ventricular S4 gallop. The aim of diagnostic testing in patients with suspected PPH is to exclude secondary causes of pulmonary hypertension. At initial screening, blood tests should include liver function tests and assays for antibodies to HIV-1, and serological studies should aim to exclude occult collagen vascular disease. Pulmonary function tests should be done to exclude significant parenchyma or airway disorders. Patients with severe PPH may have a mild restrictive pattern. Cardiac catheterization is the most important test in the assessment of pulmonary hypertension. Catheterization is necessary to fully assess right and left heart hemodynamics, the presence of shunts and vasoreactivity during acute drug trials. Acute vasodilator testing is an important component of the hemodynamic assessment, since the responses to acute challenge with vasodilators is predictive of the long-term response to oral vasodilator therapy20). We did not perform the vasodilator testing during the cardiac catheterization. Almost every type of vasodilator has been tried in the past, but there have been no prospective randomized trials of oral vasodilator therapy for PPH. Non-controlled studies have shown improved hemodynamics, exercise tolerance and survival in some patients treated with oral vasodilators20). A therapy using a high dose of calcium channel antagonists that are titrated to the maximal response of the pulmonary artery pressure and pulmonary vascular resistance has been described, and associated with a dramatic improvement in quality of life and lifestyle, regression of right ventricular hypertrophy and improved survival21). Continuous intravenous epoprostenol (prostacyclin, PGI2) has been shown to improve hemodynamics, to improve tolerance of exercise and to prolong survival in severe PPH22–24). Two trials have suggested improved survival for PPH patients treated with anticoagulants4,21). The prognosis for untreated PPH is poor. In a series of 137 cases from the UK, the median survival time was 3.4 years25). Among 200 patients enrolled on the US National Institute of Health Registry, the mean life expectancy was 2.5 years from diagnosis of PPH. This study showed 64% survival at 1 year and 48% survival at 3 years. The results were not affected by age, age at onset, sex, symptom duration, a positive test for antinuclear antibodies, family history, use of oral contraceptives, pregnancy or smoking status25). Stroke volume index, cardiac index, right atrial pressure and mean pulmonary artery pressure at catheterization are linked to survival7). Patients who respond to chronic therapy with calcium-channel blockers have a 95% chance of a 5-year life-expectancy when anticoagulant therapy is used at the same time21). Epoprostenol has increased survival in patients who are unresponsive to oral vasodilators, and is associated with a 5-year survival comparable with or better than survival after lung transplantation26). In regard to our results, the mean survival time was 3.4 years and the cardiac index is only associated with survival.
The limitation of this study includes that we have tried a common conventional vasodilator drug without a test of the vasodilator during the cardiac catheterization, so we excluded the drug for prognostic factor of survival. Not all patients have taken anticoagulation and prostacyclin. Although, there are a small number of cases and incomplete data of our study, no data has been published about survival and prognostic factor in all patients with PPH in Korea. Multicenter trial is needed to evaluate the survival and prognostic factor in all patients with PPH in Korea. In conclusion, patients with PPH have a poor survival expectancy and in this limited study with a small number of patients, mortality is largely associated with decreased cardiac index.
Table 1.
Baseline characteristics
Table 2.
Pulmonary function and hemodynamic findings of 13 patients at entry into the study
FVC indicates forced vital capacity; FEV1, forced expiratory volume in 1 second; MMEF, maximal mid-expiratory flow; PO2, oxygen pressure; PCO2, carbon dioxide pressure; RAP, right atrial pressure; RVEDP, right ventricular end-diastolic pressure; PCWP, pulmonary capillary wedge pressure; CI, cardiac index.
Table 3.
Variables associated with poor survival in 13 patients with primary pulmonary hypertension
FVC indicates forced vital capacity; FEV1, forced expiratory volume in 1 second; MMEF, maximal mid-expiratory flow; PO2, oxygen pressure; PCO2, carbon dioxide pressure; RAP, right atrial pressure; RVEDP, right ventricular end-diastolic pressure; PCWP, pulmonary capillary wedge pressure; CI, cardiac index.
REFERENCES
1. Dresdale DJ, Schultz M, Michtom RJ. Primary pulmonary hypertension, I; clinical and hemodynamic study. Am J Med 11:686–7051951.


4. Fuster V, Steele PM, Edwards WD, Gersh BJ, McGoon MD, Frye R. Primary pulmonary hypertension: natural history and the importance of thrombosis. Circulation 70:560–71984.

5. Hughes JD, Rubin LJ. Primary pulmonary hypertension: an analysis of 28 cases and a review of the literature. Medicine 65:56–721986.


6. Glanville AR, Burke CM, Theodore J, Robin ED. Primary pulmonary hypertension: length of survival in patients referred for heart-lung transplantation. Chest 91:675–811987.


7. D’Alonzo G, Barst RJ, Ayres SM. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med 115:343–91991.


8. Charters Ad, Baker W de C. Primary pulmonary hypertension of unusually long duration. Br Heart J 32:130–31970.



9. Trell E. Benign, idiopathic pulmonary hypertension? Two further cases of unusually long duration. Acta Med Scand 193:137–431973.


10. Bourdillon PD, Oakley CM. Regression of primary pulmonary hypertension. Br Heart J 38:264–701976.



11. Fujii A, Rabinovitch M, Matthews S. A case of spontaneous resolution of idiopathic pulmonary hypertension. Br Heart J 46:574–71981.



13. Romberg E. Ueber sklerose der lungen arterie. Dutch Archiv Klin Med 48:197–2061891.
14. Rich S, Dantzker DR, Ayres SM. Primary pulmonary hypertension. A national prospective study. Ann Intern Med 107:216–231987.


15. Kanemoto N, Sasamoto H. Pulmonary hemodynamics in primary pulmonary hypertension. Jap Heart J 20:395–4051979.


16. McDonnell P, Toye P, Hutchins G. Primary pulmonary hypertension and cirrhosis: are they related. Am Rev Respir Dis 127:437–411983.


17. Abenhaim L, Moride Y, Brenot F. Appetite-suppressant drugs and the risk of primary pulmonary hypertension: International Primary Pulmonary Hypertension study group. N Eng J Med 335:609–161996.

18. Loyd J, Newman J. Familial primary pulmonary hypertension: clinical patterns. Am Rev Respir Dis 129:194–71984.

19. Herve P, Launay JM, Scrobohaci ML. Increased plasma serotonin in primary pulmonary hypertension. Am J Med 99:249–541995.


20. Gaine SP, Rubin LJ. Primary pulmonary hypertension 352:719–251998.
21. Rich S, Kaufmann E, Levy PS. The effect of high doses of calcium channel blockers on survival in primary pulmonary hypertension. N Eng J Med 327:76–811992.

22. Barst RJ, Rubin LJ, Long WA. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension: the Primary Pulmonary Hypertension study group. N Eng J med 334:296–3021996.

23. Higenbottam T, Wells F, Wheeldon D, Wallwork J. Long-term treatment of primary pulmonary hypertension with continuous intravenous epoprostenol (prostacyclin). Lancet i:1046–471984.

24. Rubin LJ, Mendoza J, Hood M. Treatment of primary pulmonary hypertension with continuous intravenous prostacyclin (epoprostenol): results of a randomized trial. Ann Intern Med 112:485–911990.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



