1. Foretz M, Guigas B, Viollet B. Understanding the glucoregulatory mechanisms of metformin in type 2 diabetes mellitus. Nat Rev Endocrinol 2019;15:569–589.


4. El-Mir MY, Nogueira V, Fontaine E, Averet N, Rigoulet M, Leverve X. Dimethylbiguanide inhibits cell respiration via an indirect effect targeted on the respiratory chain complex I. J Biol Chem 2000;275:223–228.


13. Towler MC, Hardie DG. AMP-activated protein kinase in metabolic control and insulin signaling. Circ Res 2007;100:328–341.


15. Blagih J, Coulombe F, Vincent EE, et al. The energy sensor AMPK regulates T cell metabolic adaptation and effector responses in vivo. Immunity 2015;42:41–54.


18. Speirs C, Williams J, Riches K, Salt IP, Palmer TM. Linking energy sensing to suppression of JAK-STAT signalling: a potential route for repurposing AMPK activators? Pharmacol Res 2018;128:88–100.


19. Nerstedt A, Johansson A, Andersson CX, Cansby E, Smith U, Mahlapuu M. AMP-activated protein kinase inhibits IL-6-stimulated inflammatory response in human liver cells by suppressing phosphorylation of signal transducer and activator of transcription 3 (STAT3). Diabetologia 2010;53:2406–2416.


24. Vasamsetti SB, Karnewar S, Kanugula AK, Thatipalli AR, Kumar JM, Kotamraju S. Metformin inhibits monocyte-to-macrophage differentiation via AMPK-mediated inhibition of STAT3 activation: potential role in atherosclerosis. Diabetes 2015;64:2028–2041.


26. McInnes IB, Schett G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet 2017;389:2328–2337.


28. Firestein GS, Zvaifler NJ. How important are T cells in chronic rheumatoid synovitis?: II. T cell-independent mechanisms from beginning to end. Arthritis Rheum 2002;46:298–308.


29. Panayi GS. Even though T-cell-directed trials have been of limited success, is there reason for optimism? Nat Clin Pract Rheumatol 2006;2:58–59.


34. Nie H, Zheng Y, Li R, et al. Phosphorylation of FOXP3 controls regulatory T cell function and is inhibited by TNF-α in rheumatoid arthritis. Nat Med 2013;19:322–328.


35. Aravena O, Pesce B, Soto L, et al. Anti-TNF therapy in patients with rheumatoid arthritis decreases Th1 and Th17 cell populations and expands IFN-γ-producing NK cell and regulatory T cell subsets. Immunobiology 2011;216:1256–1263.


36. McGovern JL, Nguyen DX, Notley CA, Mauri C, Isenberg DA, Ehrenstein MR. Th17 cells are restrained by Treg cells via the inhibition of interleukin-6 in patients with rheumatoid arthritis responding to anti-tumor necrosis factor antibody therapy. Arthritis Rheum 2012;64:3129–3138.


37. Samson M, Audia S, Janikashvili N, et al. Brief report: inhibition of interleukin-6 function corrects Th17/Treg cell imbalance in patients with rheumatoid arthritis. Arthritis Rheum 2012;64:2499–2503.


39. Ju JH, Heo YJ, Cho ML, et al. Modulation of STAT-3 in rheumatoid synovial T cells suppresses Th17 differentiation and increases the proportion of Treg cells. Arthritis Rheum 2012;64:3543–3552.


40. Kang KY, Kim YK, Yi H, et al. Metformin downregulates Th17 cells differentiation and attenuates murine autoimmune arthritis. Int Immunopharmacol 2013;16:85–92.


43. Chen K, Lin ZW, He SM, et al. Metformin inhibits the proliferation of rheumatoid arthritis fibroblast-like synoviocytes through IGF-IR/PI3K/AKT/m-TOR pathway. Biomed Pharmacother 2019;115:108875.


45. Bartok B, Boyle DL, Liu Y, et al. PI3 kinase δ is a key regulator of synoviocyte function in rheumatoid arthritis. Am J Pathol 2012;180:1906–1916.


46. Bartok B, Hammaker D, Firestein GS. Phosphoinositide 3-kinase δ regulates migration and invasion of synoviocytes in rheumatoid arthritis. J Immunol 2014;192:2063–2070.


49. Kim HR, Kim KW, Kim BM, Lee KA, Lee SH. N-acetyl-l-cysteine controls osteoclastogenesis through regulating Th17 differentiation and RANKL production in rheumatoid arthritis. Korean J Intern Med 2019;34:210–219.


51. Indo Y, Takeshita S, Ishii KA, et al. Metabolic regulation of osteoclast differentiation and function. J Bone Miner Res 2013;28:2392–2399.


52. Matsuoka Y, Morimoto S, Fujishiro M, et al. Metformin repositioning in rheumatoid arthritis. Clin Exp Rheumatol 2021;39:763–768.


53. Lockwood TD. The lysosome among targets of metformin: new anti-inflammatory uses for an old drug? Expert Opin Ther Targets 2010;14:467–478.


55. Jhun J, Lee S, Kim SY, et al. Combination therapy with metformin and coenzyme Q10 in murine experimental autoimmune arthritis. Immunopharmacol Immunotoxicol 2016;38:103–112.


57. Koh JH, Park YJ, Lee S, et al. Distinct urinary metabolic profile in rheumatoid arthritis patients: a possible link between diet and arthritis phenotype. J Rheum Dis 2019;26:46–56.

58. Son KM, Kang SH, Seo YI, Kim HA. Association of body composition with disease activity and disability in rheumatoid arthritis. Korean J Intern Med 2021;36:214–222.


60. Gallagher L, Cregan S, Biniecka M, et al. Insulin-resistant pathways are associated with disease activity in rheumatoid arthritis and are subject to disease modification through metabolic reprogramming: a potential novel therapeutic approach. Arthritis Rheumatol 2020;72:896–902.


61. El-Sayyad SM, Ali MA, Kandil LS, Ragab GM, Abdelhamid Ibrahim SS. Metformin and omega-3 fish oil elicit anti-inflammatory effects via modulation of some dysregulated micro RNAs expression and signaling pathways in experimental induced arthritis. Int Immunopharmacol 2021;92:107362.


62. Park YH, Jang YJ, Choi Y, et al. Combination of LMT-28 and metformin improves beneficial anti-inflammatory effect in collagen-induced arthritis. Pharmacology 2021;106:53–59.


64. Naffaa ME, Rosenberg V, Watad A, et al. Adherence to metformin and the onset of rheumatoid arthritis: a population-based cohort study. Scand J Rheumatol 2020;49:173–180.


65. Abdallah MS, Alarfaj SJ, Saif DS, et al. The AMPK modulator metformin as adjunct to methotrexate in patients with rheumatoid arthritis: a proof-of-concept, randomized, double-blind, placebo-controlled trial. Int Immunopharmacol 2021;95:107575.


66. Bijlsma JW, Berenbaum F, Lafeber FP. Osteoarthritis: an update with relevance for clinical practice. Lancet 2011;377:2115–2126.


67. Deveza LA, Loeser RF. Is osteoarthritis one disease or a collection of many? Rheumatology (Oxford) 2018;57(suppl 4):iv34–iv42.


69. Zhuo Q, Yang W, Chen J, Wang Y. Metabolic syndrome meets osteoarthritis. Nat Rev Rheumatol 2012;8:729–737.


71. Barnett LA, Jordan KP, Edwards JJ, van der Windt DA. Does metformin protect against osteoarthritis? An electronic health record cohort study. Prim Health Care Res Dev 2017;18:623–628.


73. Schett G, Kleyer A, Perricone C, et al. Diabetes is an independent predictor for severe osteoarthritis: results from a longitudinal cohort study. Diabetes Care 2013;36:403–409.


75. Kevorkian L, Young DA, Darrah C, et al. Expression profiling of metalloproteinases and their inhibitors in cartilage. Arthritis Rheum 2004;50:131–141.


77. Abramson SB. Osteoarthritis and nitric oxide. Osteoarthritis Cartilage 2008;16:Suppl 2. S15–S20.


78. Attur M, Al-Mussawir HE, Patel J, et al. Prostaglandin E2 exerts catabolic effects in osteoarthritis cartilage: evidence for signaling via the EP4 receptor. J Immunol 2008;181:5082–5088.


79. Roach HI, Yamada N, Cheung KS, et al. Association between the abnormal expression of matrix-degrading enzymes by human osteoarthritic chondrocytes and demethylation of specific CpG sites in the promoter regions. Arthritis Rheum 2005;52:3110–3124.


81. Stanton H, Rogerson FM, East CJ, et al. ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 2005;434:648–652.


82. Yatabe T, Mochizuki S, Takizawa M, et al. Hyaluronan inhibits expression of ADAMTS4 (aggrecanase-1) in human osteoarthritic chondrocytes. Ann Rheum Dis 2009;68:1051–1058.


83. Glasson SS, Askew R, Sheppard B, et al. Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 2005;434:644–648.


84. Majumdar MK, Askew R, Schelling S, et al. Double-knockout of ADAMTS-4 and ADAMTS-5 in mice results in physiologically normal animals and prevents the progression of osteoarthritis. Arthritis Rheum 2007;56:3670–3674.


85. Schadler P, Lohberger B, Stundl N, et al. The effect of body mass index and metformin on matrix gene expression in arthritic primary human chondrocytes. Cartilage 2021;13(2 Suppl):1004S–1018S.

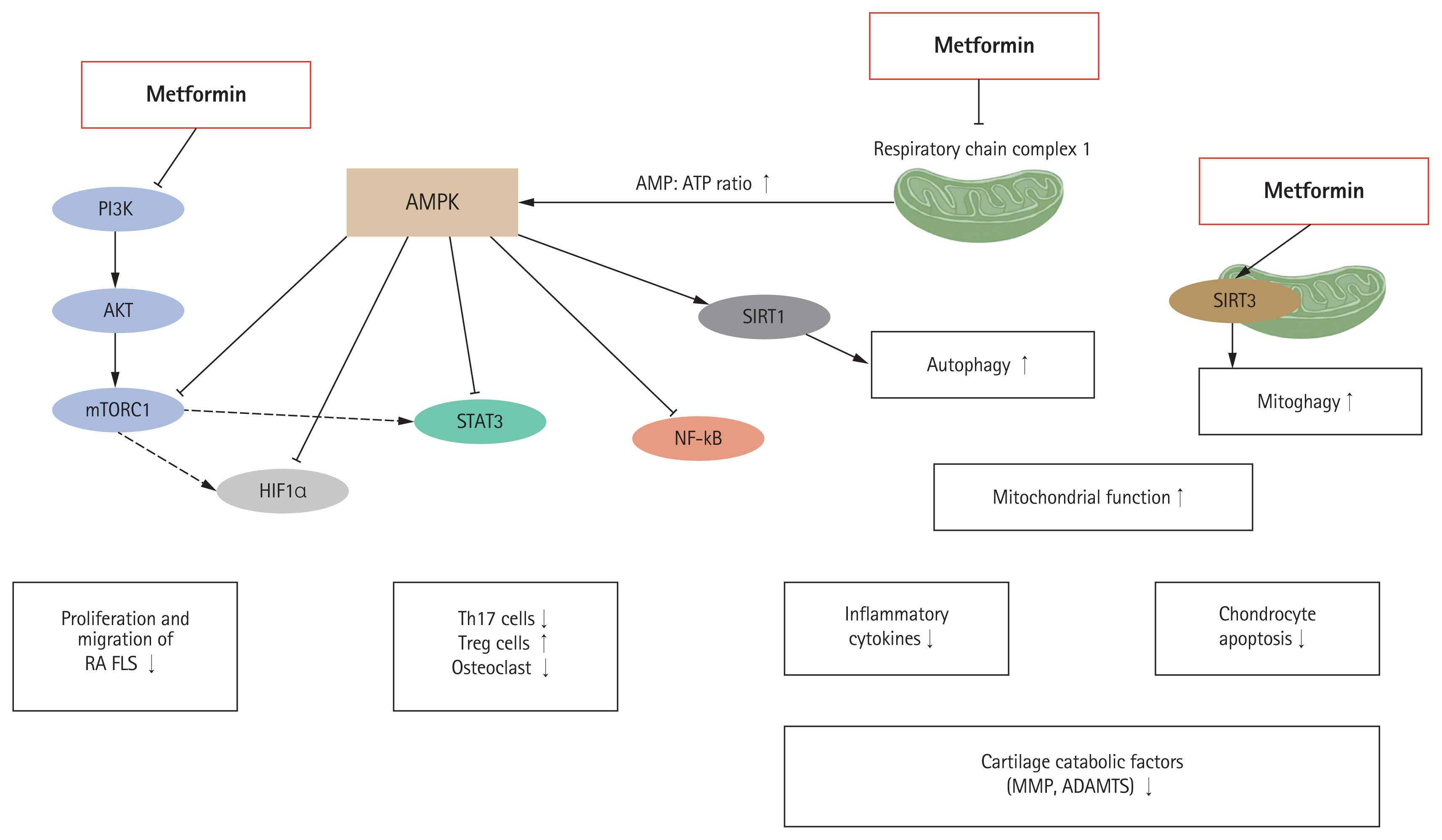
88. Li J, Zhang B, Liu WX, et al. Metformin limits osteoarthritis development and progression through activation of AMPK signalling. Ann Rheum Dis 2020;79:635–645.


90. Thomas CM, Fuller CJ, Whittles CE, Sharif M. Chondrocyte death by apoptosis is associated with cartilage matrix degradation. Osteoarthritis Cartilage 2007;15:27–34.


93. Sasaki H, Takayama K, Matsushita T, et al. Autophagy modulates osteoarthritis-related gene expression in human chondrocytes. Arthritis Rheum 2012;64:1920–1928.


94. Wang C, Yang Y, Zhang Y, Liu J, Yao Z, Zhang C. Protective effects of metformin against osteoarthritis through upregulation of SIRT3-mediated PINK1/Parkin-dependent mitophagy in primary chondrocytes. Biosci Trends 2019;12:605–612.


97. Sellam J, Berenbaum F. The role of synovitis in pathophysiology and clinical symptoms of osteoarthritis. Nat Rev Rheumatol 2010;6:625–635.


98. Dawood AF, Alzamil N, Ebrahim HA, et al. Metformin pretreatment suppresses alterations to the articular cartilage ultrastructure and knee joint tissue damage secondary to type 2 diabetes mellitus in rats. Ultrastruct Pathol 2020;44:273–282.


99. Zhang M, Liu Y, Huan Z, Wang Y, Xu J. Metformin protects chondrocytes against IL-1β induced injury by regulation of the AMPK/NF-κB signaling pathway. Pharmazie 2020;75:632–636.


100. Park MJ, Moon SJ, Baek JA, et al. Metformin augments anti-inflammatory and chondroprotective properties of mesenchymal stem cells in experimental osteoarthritis. J Immunol 2019;203:127–136.


101. Tsokos GC, Lo MS, Costa Reis P, Sullivan KE. New insights into the immunopathogenesis of systemic lupus erythematosus. Nat Rev Rheumatol 2016;12:716–730.


102. Lee YH, Song GG. Associations between circulating interleukin-17 levels and systemic lupus erythematosus and between interleukin-17 gene polymorphisms and disease susceptibility: a meta-analysis. J Rheum Dis 2020;27:37–44.

103. Yin Y, Choi SC, Xu Z, et al. Normalization of CD4
+ T cell metabolism reverses lupus. Sci Transl Med 2015;7:274ra18.



104. Yin Y, Choi SC, Xu Z, et al. Glucose oxidation is critical for CD4
+ T cell activation in a mouse model of systemic lupus erythematosus. J Immunol 2016;196:80–90.


105. Wang H, Li T, Chen S, Gu Y, Ye S. Neutrophil extracellular trap mitochondrial DNA and its autoantibody in systemic lupus erythematosus and a proof-of-concept trial of metformin. Arthritis Rheumatol 2015;67:3190–3200.


106. Titov AA, Baker HV, Brusko TM, Sobel ES, Morel L. Metformin inhibits the type 1 IFN response in human CD4
+ T cells. J Immunol 2019;203:338–348.


108. Jang SG, Lee J, Hong SM, Kwok SK, Cho ML, Park SH. Metformin enhances the immunomodulatory potential of adipose-derived mesenchymal stem cells through STAT1 in an animal model of lupus. Rheumatology (Oxford) 2020;59:1426–1438.


109. Cornaby C, Elshikha AS, Teng X, et al. Efficacy of the combination of metformin and CTLA4Ig in the (NZB × NZW)F1 mouse model of lupus nephritis. Immunohorizons 2020;4:319–331.


112. Karatas A, Celik C, Oz B, et al. Secukinumab and metformin ameliorate dermal fibrosis by decreasing tissue interleukin-17 levels in bleomycin-induced dermal fibrosis. Int J Rheum Dis 2021;24:795–802.


114. Ursini F, Grembiale RD, D’Antona L, et al. Oral metformin ameliorates bleomycin-induced skin fibrosis. J Invest Dermatol 2016;136:1892–1894.


115. Wang Y, Zhang S, Liang Z, et al. Metformin attenuates bleomycin-induced scleroderma by regulating the balance of Treg/Teff cells and reducing spleen germinal center formation. Mol Immunol 2019;114:72–80.


116. Qin X, Jiang T, Liu S, et al. Effect of metformin on ossification and inflammation of fibroblasts in ankylosing spondylitis: an in vitro study. J Cell Biochem 2018;119:1074–1082.


117. Vazirpanah N, Ottria A, van der Linden M, et al. mTOR inhibition by metformin impacts monosodium urate crystal-induced inflammation and cell death in gout: a prelude to a new add-on therapy? Ann Rheum Dis 2019;78:663–671.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



