 |
 |
| Korean J Intern Med > Volume 14(2); 1999 > Article |
|
Abstract
Objectives
This study examined the effect of cilostazol, a potent phosphodiesterase inhibitor, on the progression of neuropathies associated with streptozotocininduced diabetes mellitus in Sprague-Dawley rats.
Methods
Eight weeks after streptozotocin treatment, a pelleted diet containing 0.03% cilostazol (15mg/kg body weight) was given for four weeks. Body weight, blood glucose level, motor nerve conduction velocity(MNCV), myelinated fiber density and size distribution of sciatic nerves were compared between age-matched normal rats (Group 1), control diabetic rats (Group 2) and cilostazol-treated diabetic rats (Group 3).
Results
Body weight was significantly reduced and blood glucose level was significantly increased in diabetic rats (Group 2 and 3) compared to normal rats. MNCV and cAMP content of sciatic nerves were significantly reduced in diabetic rats 12 weeks after streptozotocin treatment. Myelinated fiber size and density were also significantly reduced, and thickening of the capillary walls and duplication of the basement membranes of the endoneural vessels were observed in the diabetic rats. Whereas both body weight and blood glucose level of Group 3 did not differ significantly from those of Group 2, cilostazol treatment significantly increased MNCV and cAMP content of sciatic nerves in Group 3 but not to the levels observed in Group 1. MNCV positively correlated with cAMP content of sciatic nerves (r=0.86; p < 0.001). Cilostazol treatment not only restored myelinated fiber density and size distribution but reversed some of the vascular abnormalities.
Both metabolic 1, 2) and vascular defects3ŌĆō5) have been implicated in the pathogenesis of diabetic neuropathy, but the precise mechanisms causing peripheral neuropathy have not been elucidated. Nerve conduction is impaired in overt diabetic neuropathy by a combination of structural and metabolic defects in the peripheral nerve6). Many studies attribute the slowing of nerve conduction in diabetic rats to alterations in nerve Na+ and Na+-related metabolism mediated by a reversible Na+, K+-ATPase defect1, 7, 8)
The decreases in Na+, K+-ATPase activity and motor nerve conduction velocity (MNCV) in diabetic rats have been reported to be ameliorated by treatment with aldose reductase inhibitors8ŌĆō11), dietary myo-inositol supplementation12), gangliosides13, 14), prostaglandin E115), anti-oxidants16 or essential fatty acids17, 18). More recent experiments showed that there was a positive correlation between cAMP content and Na+,K+-ATPase activity, and MNCV and Na+,K+-ATPase activity in the rat sciatic nerve19).
Haemodynamic abnormalities of the peripheral nerve have also been suggested as a major cause of the functional deterioration in the neuropathies observed in streptozotocin (STZ)-induced diabetic rats3, 20. In addition to marked biochemical and functional abnormalities, morphological changes have been demonstrated in the nerves of diabetic rats21).
Cilostazol(6-[4-cyclohexyl-1H-tetrazol-5-yl)butyl}3,4-dihydro-2(1H)-quinolinone]), a potent phosphodiesterase inhibitor, increases the cAMP content of the sciatic nerves of rats19). In this study, we examined the effects of cilostazol on biochemical, functional and morphological aspects of experimental diabetic neuropathies induced by STZ in the rat.
Eight-week-old male Sprague Dawely rats (Charles River, Japan) were acclimatized to their new environment for one week before the experiment. After an overnight fast, the rats were rendered diabetic by i.p. injection of STZ (35 mg/kg) (Sigma Chemical Co., St. Louis, MO, USA) in 10mM citrate buffer, pH 4.5. A diabetic rat was identified by a nonfasting tail-vein plasma glucose level exceeding 16.7mM/L in the first week after injection with STZ.
Diabetic rats at 8 weeks after STZ treatment were divided into two groups: the first group was given a pelleted diet containing 0.03% cilostazol(15mg/kg/day) (Otsuka Pharmaceutical Co. Tokyo, Japan) for another four weeks and the second group was the untreated age-matched diabetic rats. Age-matched normal rats were used as non-diabetic controls. Motor nerve conduction velocity (MNCV) was measured before and after treatment with the drug. Rats in each group were killed by cardiac puncture under light ether anesthesia in the 12th week after STZ-treatment for the measurement of cAMP and light and electron microscope examination of sciatic nerve samples.
MNCVs were measured in the sciatic nerves of rats using a Nuropack II (Nihon Kohden Co., Tokyo, Japan), as previously reported.26). Briefly, animals were lightly anesthetized with an i.p. injection of 40 mg/kg pentobarbital sodium. Body temperature was monitored using a rectal probe and maintained at 37 ┬░C with a warming pad. Sciatic-tibial NCV was determined non-invasively by stimulating proximally at the sciatic notch and distally at the ankle via bipolar electrodes with supramaximal stimulation. The proximal and distal latencies of the compound muscle action potentials, recorded via bipolar electrodes from the first interosseous muscle of the hind paw, were measured from the stimulus artifact to the onset of the negative M-wave deflection, subtracted, and divided into the distance between the stimulating and recording electrodes, giving a value for MNCV in m/s.
Nonfasting plasma glucose was measured in tail-vein blood samples, using Beckman glucose analyzer II (Beckman Instruments, Inc., Fullerton, CA). Sciatic nerves were removed from the rats under ether anesthesia and weighed. The whole or prepared nerves were immediately boiled for 5 min with sodium acetate buffer (pH 4.0), homogenized using a glass homogenizer, and then centrifuged at 2000 ├Ś g 5 min. The supernatant was used for cAMP determination by radioimmunoassay (RIA) (Incstar Co., Stillwater, Minnesota, U.S.A.)
On the day after the final MNCV determination, non-fasted animals were anesthetized with ether. Midthigh segments of the sciatic nerve were surgically removed and fixed in a 2.5% cacodylate-buffered glutaraldehyde fixative, dehydrated and embedded in Epon. Embedded sciatic nerves were sectioned and examined microscopically. Ultrathin cross sections were stained with toluidine blue.
Results are expressed as means┬▒SD. Multiple between-group comparisons were performed using one-way analysis of variance (ANOVA). Statistical comparisons in three groups were made using StudentŌĆÖs t test. The correlation between cAMP content and MNCV was analyzed by linear regression analysis.
As shown in Table 1, blood glucose levels were significantly elevated and body weights significantly decreased compared to normal control rats 12 weeks after STZ administration. cAMP levels were decreased in the sciatic nerves of the diabetic rats. Administration of cilostazol (weeks 9ŌĆō12) had no effect on blood glucose levels and body weights but significantly increased cAMP levels of sciatic nerves compared to age-matched, untreated diabetic rats. However, cilostazol treatment did not restore cAMP levels of diabetic rats to those of age-matched normal control rats.
As shown in Table 2, significant reduction in MNCVs were observed in diabetic rats compared to those in age-matched normal rats at the 8th and 12th weeks after STZ treatment (p<0.001). Cilostazol treatment significantly increased MNCV in the sciatic nerves of diabetic rats but they were not restored to the levels noted in age-matched normal control rats.
As shown in Fig. 1, there was a positive correlation between cAMP content and MNCV in the sciatic nerve at 12 weeks after STZ treatment (r=0.86; P<0.001).
cAMP and MNCV in untreated diabetic rats were scatterd in the lower left part of the curve and cilostazol treatment shifted the data points to the upper right part of the curve.
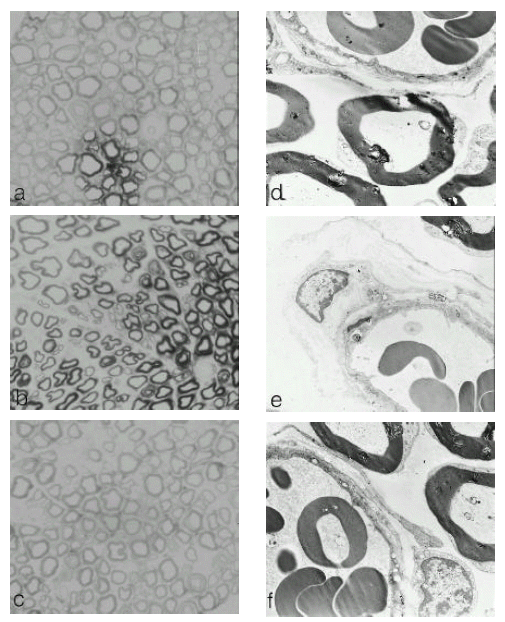
Morphological changes were examined in the sciatic nerve by light and electron microscopy. In the peripheral nerves of the diabetic rat, there was reduction in the number of myelinated fibers and there appeared to be more small fibers in the nerve when compared to non-diabetic control rats. Myelinated fiber density and size distribution were restored after treatment of cilostazol (Fig 2. a, b, c).
In electron micrographs, a thickening of capillary walls and duplication of basement membranes in the nerves of diabetic rats were noted. These vascular abnormalities were restored towards normal after cilostazol treatment, (Fig 2. c, d, f).
The streptozotocin (STZ)-induced diabetic rat is the most commonly used animal model of human diabetic neuropathies. Early changes in nerve function are characterized by reduced nerve conduction velocity in this animal model3, 22, 23). Diabetic rats of eight weeksŌĆÖ duration show a significant reduction in sciatic MNCV which can be normalized by insulin treatment and near normoglycemia24). Acute hyperglycemia induced by STZ elicits activation of the polyol pathway with sorbitol accumulation, myo-inositol depletion and reduction of Na+,K+-ATPase activity25, 26) The decrease in myo-inositol causes depletion of phosphoinositides, followed by poor calcium mobilization and impaired protein kinase C activity, eventually resulting in impaired Na+, K+-ATPase activity27).
Thus, nerve Na+/K+ pump activity is reduced; fibers become Na+ loaded, Na+ channels become inactivated, and MNCV decreases. Decreasd Na+,K+ -ATPase activity and motor nerve conduction velocity(MNCV) have been reported to be restored by aldose reductase inhibitors8ŌĆō11), dietary myo-inositol supplementation8, 12), gangliosides,13, 14) prostaglandin E115), anti-oxidants16) or essential fatty acid17, 18) in diabetic rats. Recent studies have shown that cAMP levels are decreased in the peripheral nerves of STZ-diabetic rats19). cAMP is one of the intrinsic intracellular modulators which regulates Na+,K+-ATPse activity or the Na+/K+ pump in various tissues28, 29). Therefore, the possibility was suggested that increased cAMP may improve MNCV by restoration of Na+,K+-ATPse activity. It has been reported that cilostazol increased cAMP content in the nerves of diabetic rats and improved MNCV without changing myo-inositol levels, and the positive correlation between cAMP content and Na+,K+-ATPse activity was noted also19). In addition to marked biochemical and functional abnormalities, morphological changes have been demonstrated in the peripheral nerves of STZ-induced diabetic rats21).
As reported by other investigators, STZ in the present study also increased blood glucose levels and reduced body weights of the rats. In addition, the cAMP level and MNCV of sciatic nerves were decreased by STZ as reported earlier.19 Blood glucose and body weights were unaffected by cilostazol treatment but cAMP levels and MNCV were significantly increased compared to untreated diabetic rats but were not restored to the levels observed in age-matched, normal control animals as previously reported19). This discrepancy may be explained by the different strains of rats used to produce the diabetic animals or by differences in the duration and/or the severity of the experimental diabetes. Since rats with STZ-induced diabetes were not treated with insulin, we intentionally used rats with a milder diabetic state to ensure survival of the animals to the end of this prolonged study. A positive correlation between cAMP content and MNCV in the sciatic nerve was also observed. Whether the association of a reduced cAMP content with decreased MNCV implies the involvement of cAMP in motor neuron function is unknown at the present time. Reduced cAMP levels could modulate the function of motor neurons and precipitate the neuropathic condition. Clear mechanisms by which cAMP enhances MNCV are unknown, but it is possible that an increased cAMP content could affect cAMP-dependent protein kinase activity, followed by an enhancement of Na+,K+-ATPase activity in the sciatic nerve. It has been reported that protein kinase C agonists normalized Na+,K+-ATPase activity in the diabetic rabbit nerve27, 30) and cAMP stimulates protein kinase C activity in cultured renal cells31). In addition to various metabolic factors, reduced nerve blood flow, caused by rheological changes coupled with vasa nervorsum microangiopathy, leads to endoneural hypoxia of sufficient magnitude to impair nerve function3, 20).
Some experimental studies have reported that hyperglycemia-induced blood flow reduction and the resultant endoneural hypoxia were important factors underlying nerve conduction deficits early in the development of diabetic neuropathy3). The alteration of nerve blood flow, accompanied by nerve ischemia in diabetic rats, may be related to functional abnormalities3) and the reduction of Na+,K+-ATPase activity observed32). Uehara et al33) reported that the anti-platelet effects of cilostazol improved nerve blood flow, increased mean myelinated fiber size and enlarged the lumen of endoneural microvessels in diabetic rats. In our study, STZ reduced myelinated fiber size and there appeared to be more small fibers, thickening of capillary walls and duplication of basement membranes in the endoneural microvessels of sciatic nerves when compared to those of the non-diabetic, control rats. The myelinated fiber density, size distribution and vascular abnormalities were restored after treatment of cilostazol. It is possible that correction of nerve ischemia by cilostazol may result in the prevention of structural changes also. Cilostazol is a potent phosphodiesterase inhibitor, which increases the cAMP content in the sciatic nerve of rats19). Our study revealed that cilostazol can elevate cAMP levels and prevent impairment of MNCV in the sciatic nerves of STZ-induced diabetic rats, functionally and morphologically. This suggests that there is a relationship between the decreased rate of nerve regeneration and the decreased cAMP contents, which may be a possible pathophysiological link in the peripheral motor nerves of diabetic rats. Cilostazol may be useful for the prevention of diabetic neuropathies from functional and structural aspects of its action.
Acknowledgments
The authors thank Otsuka Pharmaceutical Company for providing cilostazol used in this study.
Fig.┬Ā1.
The correlation between cAMP content and MNCV in the sciatic nerve of rats at 12 weeks Eight-week-old, male Sprague-Dawley rats were divided into two groups: an age-matched, normal group and a group rendered diabetic by administration of streptozotocin (STZ; 35mg/kg, i.p.). Eight weeks later, the STZ-treated diabetic rats were further subdivided into two groups: one group consisted of age-matched untreated diabetic rats and, in the second group, the diabetic rats were treated with 0.03% cilostazol in their daily diets for four weeks.

Fig.┬Ā2.
Light (a, b, c. ├Ś 400) and electron (d, e, f. ├Ś 5100) photomicrography of semi-thin transverse section of rat sciatic nerves. Eight-week-old, male Sprague-Dawley rats were divided into two groups: an age-matched, normal control (a, d), and a group rendered diabetic by administration of streptozotocin. Eight weeks later, the STZ-treated diabetic rats were further subdivided into two groups: one group was an age-matched untreated diabetic rat (b, e) and the second group was treated with 0.03% cilostazol in their daily diets for four weeks (c, f).
Table┬Ā1.
Blood glucose, body weight and cAMP contents of rat sciatic nerves
| Group | n |
Blood glucose (mM) |
Body weight (g) |
cAMP (fmol/mg wt) |
|---|---|---|---|---|
| Age-matched, Normal control | 10 | 5.9┬▒0.7a | 376.0┬▒11.6a | 93.9+10.8a |
| Age-matched, Diabetic untreated | 7 | 18.5┬▒0.9 | 214.1┬▒7.5 | 62.6┬▒3.8 |
| Diabetic cilostazol-treated | 8 | 17.8┬▒0.9 | 218.5┬▒6.0 | 80.8┬▒5.3a,b |
Eight-week-old, male Sprague-Dawley rats were divided into two groups: an age-matched, normal group and a group rendered diabetic by administration of streptozotocin (STZ; 35mg/kg, i.p.). Eight weeks later, the STZ-treated diabetic rats were further subdivided into two groups: one group consisted of age-matched, untreated diabetic rats and, in the second group, the diabetic rats were treated with 0.03% cilostazol in their daily diets for four weeks.
Table┬Ā2.
Motor nerve conduction velocity (MNCV) of rat sciatic nerves at 8 and 12 weeks after streptozotocin treatment
|
MNCV (m/sec)
|
|||
|---|---|---|---|
| Group | n | 8 weeks | 12 weeks |
| (treatment for 4 weeks) | |||
| Age-matched, Normal control | 10 | 47.5┬▒3.4a | 51.8┬▒4.3a |
| Age-matched, Diabetic untreated | 7 | 30.9┬▒3.2 | 31.6┬▒4.4 |
| Diabetic cilostazol ŌĆō treated | 8 | 31.4┬▒5.1 | 41.4┬▒2.9a,b |
The experimental conditions were the same as those described in Table 1.
REFERENCES
1. Greene DA, Lattimer SA, Sima AF. Sorbitol, phosphoinositide and sodium-potassium-ATPase in the pathogenesis of diabetic complications. N Engl J Med 1987;316:599ŌĆō606.


2. Greene DA, Sima AF, Stevens MJ, Feldman EL, Lattimer SA. Complications; neuropathy, pathogenetic considerations. Diabetes Care 1992;15:1902ŌĆō1925.


3. Cameron NE, Cotter MA, Low PA. Nerve blood flow in early experimental diabetes; relation to conduction deficits. Am J Physiol 1991;261:E1ŌĆōE8.


4. Cameron NE, Cotter MA, Ferguson K, Robertson S, Radecliff MA. Effects of chronic alpha-adrenergic receptor blockage on peripheral nerve conduction, hypoxic resistance, polyols, Na/K-ATPase activity and vascular supply in STZ-D rats. Diabetes 1991;40:1652ŌĆō1658.


5. Cameron NE, Cotter MA, Roberton S. Ansiotensin converting enzyme inhibition prevents development of muscle and nerve dysfunction and stimulated angiogenesis in streptozotocin-diabetic rats. Diabetologia 1992;35:12ŌĆō18.


7. Greene DA, Yagihashi S, Lattimer SA, Sima AAF. Nerve Na+-K+-ATPase, conduction and myo-inositol in the insulin deficient BB-rat. Am J Physiol 1984;247:E534ŌĆō549.


8. Mayer JH, Tomlinson DR. Prevention of defects of axonal transport and nerve conduction velocity by oral administration of myo-inositol or an aldose reductase inhibitor in streptozotocin-diabetic rats. Diabetologia 1983;25:433ŌĆō438.


9. Cameron NE, Cotter MA, Dines KC, Maxfield EK, Carey F, Mirrlees DJ. Aldose reductase inhibition, nerve perfusion, oxygenation and function in streptozotocin-diabetic rats: Dose-response considerations and independence from a myo-inositol mechanism. Diabetologia 1994;37:651ŌĆō663.


10. Yue DK, Hanwell MA, Satchell PM, Turtle JR. The effect of aldose reductase inhibition on motor nerve conduction velocity in diabetic rats. Diabetes 1982;31:789ŌĆō794.


11. Finegold D, Lattimer SA, Nolle S, Bernstein M, Greene DA. Poyol pathway activity and myo-inositol metabolism. A suggested relationship in the pathogenesis of diabetic neuropathy. Diabetes 1983;32:988ŌĆō992.


12. Gillon KR, Hawthorne JN, Tomlinson DR. Myo-inositol and sorbitol metabolism in relation to peripheral nerve function in experimental diabetes in the rat. The effect of aldose reductase inhibition. Diabetoloagia 1983;25:365ŌĆō371.

13. Bianchi R, Berti-Mattera LN, Fiori MG, Eichberg J. Correction of altered metabolic activities in sciatic nerve of streptozotocin-induced diabetic rats. Diabetes 1990;39:782ŌĆō788.


14. Bianchi R, Marini P, Merlini S, Fabris M, Triban C, Mussini E, Fiori MG. ATPase activity defects in alloxan-induced diabetic sciatic nerve recovered by ganglioside treatment. Diabetes 1988;37:1340ŌĆō1345.


15. Sonobe M, Yasuda H, Hisanaga T, Maeda K, Yamashita M, Kawabata T, Kikkawa R, Taniguchi Y, Shigeta Y. Amelioration of nerve Na+,K+-ATPase activity independently of myo-inositol level by PGE1 analogue OP-1206 ╬▒-CD in streptozotocin-induced diabetic rats. Diabetes 1991;40:726ŌĆō730.


16. Cameron NE, Cotter MA, Archibald V, Dines KC, Maxfield EK. Anti-oxidant and pro-oxidant effects on nerve conduction velocity, endoneurial blood flow and oxygen tension in non-diabetic and streptozotocin-diabetic rats. Diabetologia 1994;37:449ŌĆō459.


17. Cameron NE, Cotter MA, Hohman TC. Interactions between essential fatty acid, prostanoid, poyol pathway and nitric oxide mechanisms in the neurovascular deficit of diabetic rats. Diabetologia 1996;39:172ŌĆō182.


18. Dines KC, Cotter MA, Cameron NE. Contrasting effects of treatment with Žē-3 and Žē-6 essential fatty acid on peripheral nerve function and capillarization in streptozotocin-diabetic rats. Diabetologia 1993;36:1132ŌĆō1138.


19. Sindo H, Tawata M, Onaya T. Cyclic Adenosine 3ŌĆ▓,5ŌĆ▓-Monophosphate enhances sodium, potassium-adenosine triphosphatase activity in the sciatic nerve of streptozotocin -induced diabetic rats. Endocrinology 1993;132:510ŌĆō516.


20. Low PA, Schmelzer JD, Ward KK, Yao JK. Experimental chronic hypoxic neuropathy: relevance to diabetic neuropathy. Am J Physiol 1986;250:E94ŌĆō99.


21. Jakobsen J. Axonal dwindling in early experimental diabetes. I A study of cross-sectioned nerves. Diabetologia 1976;12:539ŌĆō546.


22. Cameron NE, Cotter MA, Dines KC, Maxfield EK. Pharmacological manipulation of vascular endothelium function in non-diabltic and streptozotocin-diabetic rats: effects on nerve conduction, hypoxic resistance and endoneurial capillarization. Diabetologia 1993;36:516ŌĆō522.


23. Sima AAF, Brisma T. Reversible diabetic nerve dysfunction; structural correlates to electrophysiological abnormalities. Ann Neurol 1985;18:21ŌĆō29.


24. Cameron NE, Cotter MA, Robertson S. Chronic low frequency electrical activation for one week corrects nerve conduction velocity deficits in rats. Diabetologia 1989;32:759ŌĆō761.


25. Greene DA, Lattimer SA. Impaired rat sciatic nerve, sodium-potassium adenosine triphosphatase in acute streptozotocin diabetes and its correction by dietary myo-inositol supplementation. J Clin Invest 1983;72:1058ŌĆō1063.



26. Greene DA, Lattimer SA, Sima AAF. Sorbitol, phosphoinositides and sodium-potassium-ATPase in the pathogenesis of diabetic complication. N Engl J Med 1987;316:599ŌĆō606.


27. Lattimer SA, Sima AAF, Greene DA. In vitro correction of impaired Na+-K+-ATPase in diabetic nerve by protein kinase C agonists. Am J Physio 1989;256:E264ŌĆōE269.

28. Giesen EM, Imbs JL, Grima M, Schmidt M, Schwartz J. Modulation of renal ATPase activities by cycle AMP. Biocham Biophys Res commun 1984;120:619ŌĆō624.

29. Hughs BA, Miller SS, Joseph DP, Edelman JL. cAMP stimulates the Na+,K+-ATPase pump in frog retinal pigment epithelium. Am J Physiol 1988;254:C84ŌĆō98.


30. Greene DA, Lattimer SA. Protein kinase C agonists acutely normalize decreased ouabain-inhibitable respiration in diabetic rabbit nerve; implications for (Na+,K+)-ATPase regulation and diabetic complications. Diabetes 1986;35:242ŌĆō245.


31. Anderson RJ, Breckon R. cMP stimulates protein kinase C activity in cultured renal LLC-PK1 cells. Am J Pysiol 1991;261:F945ŌĆō950.
32. Takahashi K, Kako KJ. Ischemia-induced changes in arcolemal (Na,K)-ATPase, K+-pNPPase, sialic acid, and phospholipid in the dog and effects of the nisoldipine and chlorpromazine treatment. Biocchem Med 1984;31:271ŌĆō286.

33. In: Uehara K, Sugimoto K, Yoshikawa T, Marukawa K, Yasuda Y, Yagihashi SAAF, Boulton AJM, eds. Diabetic neuropathy; new concepts and Effects of cilostazol on peripheral neuropathy in STZ-induced diabetic rats. In: Hotta N, Greene DA, Ward JD, Sima AAF, Boulton AJM, eds. Diabetic Neuropathy new concept and insights. Amsterdam: Elservier Science BV, 1995;153ŌĆō158.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



