 |
 |
| Korean J Intern Med > Volume 41(2); 2026 > Article |
|
Abstract
Background/Aims
The Psoriatic Arthritis Impact of Disease (PsAID-12) questionnaire was developed to measure quality of life and disease impact in patients with psoriatic arthritis (PsA). We aimed to translate the English PsAID-12 into Korean and to evaluate its reliability and validity.
Methods
The PsAID-12 questionnaire was translated into Korean following international cross-cultural adaptation guidelines. After expert review and cognitive debriefing with 10 patients, the final Korean version of the PsAID-12 was established. For validation, 21 patients with PsA completed the Korean PsAID-12 questionnaire, and a retest was conducted one week later to assess reliability. Demographic data and disease activity indices, including the Disease Activity in Psoriatic Arthritis (DAPSA) and Visual Analog Scale (VAS) pain were collected. Correlations between PsAID-12 scores and disease activity indices were analyzed.
Results
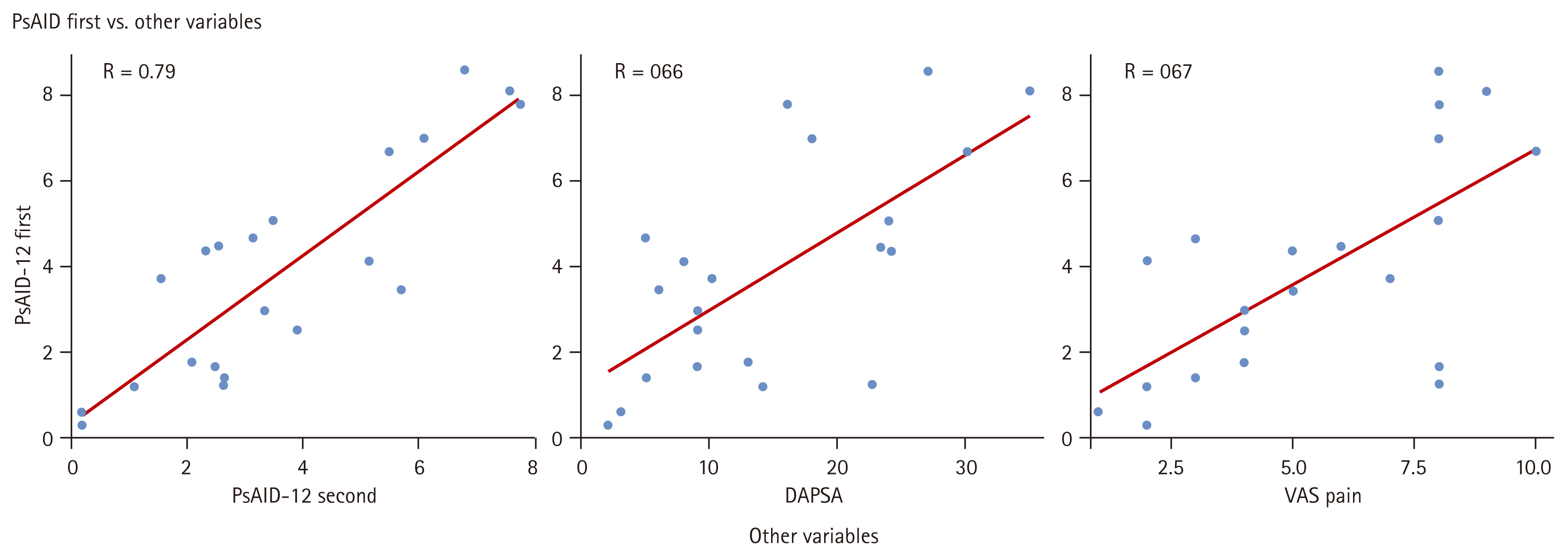
Minor discrepancies in the translation process were resolved during the synthesis and review. The mean PsAID-12 scores were 3.9 ┬▒ 2.6 and 3.6 ┬▒ 2.3 for the first and second assessments, respectively. The mean response time was 2.6 ┬▒ 1.0 min. The intraclass correlation coefficient was 0.856 (95% CI 0.682ŌĆō0.939, p < 0.050), indicating good reliability. The first PsAID-12 score was strongly correlated with the second PsAID-12 (r = 0.79), DAPSA (r = 0.66), and VAS pain (r = 0.67).
Psoriatic arthritis (PsA) is a chronic inflammatory disease characterized by skin and nail psoriasis, enthesitis, and fingernail involvement [1,2]. The prevalence of PsA is reported to range from 0.05% to 0.25%, with 6ŌĆō41% of patients with psoriasis developing PsA [3]. Although the prevalence of PsA in Korea is unknown, the overall prevalence of PsA among patients with psoriasis has been reported to be 11.2% [4].
The diverse symptoms of PsA affect both physical and mental health, impair daily activities, and limit social participation, ultimately reducing the quality of life. When evaluating PsA, it is essential to consider the psychological and social effects of the symptoms [5ŌĆō7]. While physicians often focus on measures of pain and disease activity, incorporating tools that capture patient experience provides valuable insights into the everyday challenges faced by patients [5,8]. Patient-reported outcome measures (PROMs) enable patients to directly assess their feelings and functions in relation to their health [9,10]. This is particularly important for Korean patients with PsA, who may exhibit clinical manifestations and concerns that differ from those observed in other countries, highlighting the need for research that emphasizes PROMs.
The Psoriatic Arthritis Impact of Disease (PsAID-12) questionnaire was developed by the European League Against Rheumatism (EULAR) task force involving patients and clinicians from 13 countries, which aims to provide a tool to assess the impact of PsA from the patientŌĆÖs perspective [8]. The questionnaire was developed in two versions: clinical practice (PsAID-12) and clinical trial (PsAID-9). The PsAID-12 questionnaire is grouped into three categories: physical impact, impact related to skin, and psychological and social impact, and is useful in clinical practice for the longitudinal monitoring of patients [11].
Information on PsA in Korean patients remains limited, and reliable data are scarce. Given that the PsAID-12 questionnaire has been validated in multiple countries, it may serve as a valuable tool for assessing the multidimensional impact of the disease on Korean patients. Therefore, we aimed to evaluate the reliability of the PsAID-12 questionnaire translated into Korean when applied to Korean patients with PsA.
The study was conducted between September 2024 and January 2025. The English version of the PsAID-12 questionnaire was translated into Korean and then validated in 21 patients from a single center. All participants were patients with PsA who met the Classification Criteria for Psoriatic Arthritis (CASPAR) criteria [12]. Their baseline characteristics, medication status, and disease activity, including the Disease Activity in Psoriatic Arthritis (DAPSA), C-reactive protein (CRP), Visual Analog Scale (VAS) for pain were assessed.
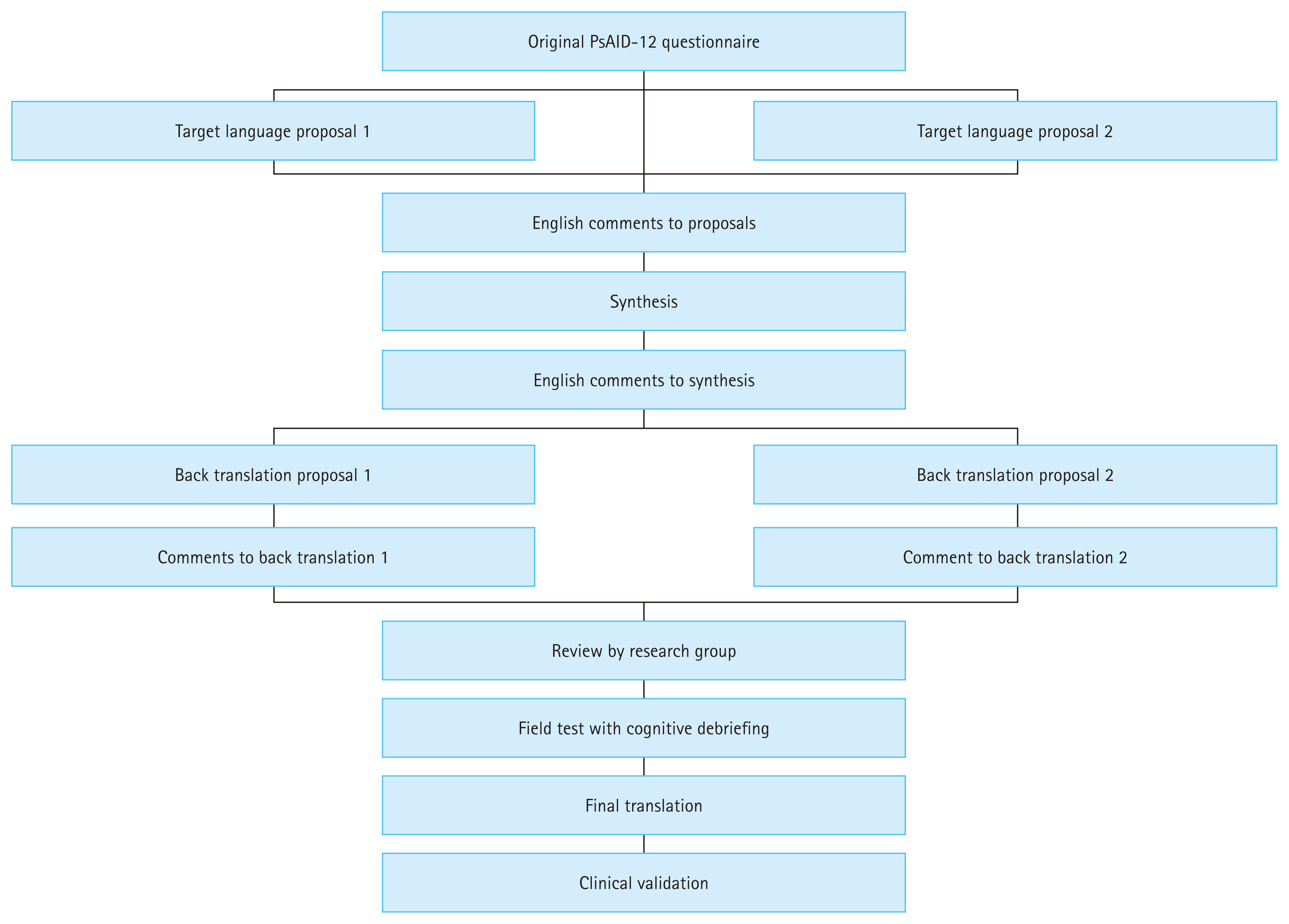
The PsAID-12 questionnaire was translated into Korean according to international translation and cross-cultural adaptation guidelines [13]. The translation process was carried out in five stages as follows: initial translation, synthesis, back-translation, review by an expert committee (comprising professionals involved in the translation and representatives from the Spondylitis Research Society of the Korean Rheumatology Association), and pretesting with patients (Fig. 1). Two native Koreans with different professional backgrounds independently forward translated the PsAID-12 questionnaire from English to Korean. The translations were then synthesized and back-translated. The back-translation from Korean to English was independently performed by two bilingual native English speakers who were not shown the original. An expert committee then reviewed the Korean translations. The Korean translation of the PsAID-12 questionnaire was completed after review by the Spondylitis Research Society of the Korean Rheumatology Association. For cognitive debriefing interviews, 10 patients were interviewed regarding the validity and comprehensibility of the questionnaire items in the final Korean translation. The time to complete the items, comprehensibility, relevance, response option clarity, comprehensiveness, and additional feedback were evaluated (Supplementary Table 1).
During a face to face consultation, the Korean PsAID-12 questionnaire was administered with the disease activity assessments. One week later, the PsAID-12 questionnaire was readministered online. The agreement in the PsAID-12 scores over a 1-week interval was evaluated.
All variables are expressed as mean ┬▒ standard deviation (SD) and percentage. The agreement between each item and the total PsAID-12 questionnaire over a 1-week interval was measured using the intraclass correlation coefficient (ICC). Internal consistency was assessed using CronbachŌĆÖs alpha coefficient for the 12 items of the Korean PsAID-12 questionnaire. SpearmanŌĆÖs correlation coefficient between each disease activity index and the total PsAID-12 score was analyzed. Statistical significance was set at p < 0.05. The analysis was conducted using R version 4.4.2 (The R Foundation for Statistical Computing).
After obtaining informed consent from patients, the validation study was conducted. This study was approved by the Institutional Review Board of Inje University Ilsan Paik Hospital Institutional Review Board (IRB No. ISPAIK 2024-10-027-001) and was conducted in accordance with the Declaration of Helsinki and Ethical Guidelines for Medical Research Involving Human Subjects.
A total of 21 patients with PsA completed the PsAID-12 questionnaire. The baseline characteristics are presented in Table 1. Of the participants, 14 (66.7%) used biological agents. The CRP level was mostly normal; however, the mean DAPSA was 15.0 ┬▒ 9.6, showing moderate activity.
Cognitive debriefing interviews demonstrated that all participants found the instructions and items of the Korean PsAID-12 questionnaire to be clear, unambiguous, and culturally appropriate (Supplementary Table 1). The response options were considered suitable, and the time required to complete the questionnaire was appropriate, with a mean completion time of 2.6 ┬▒ 1.0 minutes (range, 1ŌĆō4 min). Only one participant reported difficulty distinguishing whether fatigue was attributable to PsA or daily life, but no further substantial issues or additional suggestions were identified. Based on these results, the final Korean version of the PsAID-12 was established (Supplementary materials).
The results of the two surveys conducted 1 week apart, including the 12 PsAID-12 items, are shown in Table 2. In the first survey, the ŌĆ£fatigueŌĆØ item had the highest score (5.5 ┬▒ 2.7), while ŌĆ£embarrassment and/or shameŌĆØ had the lowest (2.6 ┬▒ 3.1). This same pattern was observed in the second survey. The total PsAID-12 scores were 3.9 ┬▒ 2.6 and 3.6 ┬▒ 2.3, respectively, with no significant difference in their mean values based on a paired t-test. The ICC was 0.856 (95% confidence interval [CI] 0.682ŌĆō0.939, p < 0.050), indicating good reliability. The BlandŌĆōAltman plot showed that the data were mainly distributed within the limit line, indicating that the reliability was acceptable and the bias was not large (Supplementary Fig. 1).
For individual items, no significant differences were found using the paired t-test; however, the ICC analysis showed that the coping ŌĆ£dealing with the diseaseŌĆØ item had poor reliability, with an ICC of 0.348 (95% CI ŌłÆ0.105 to 0.676, p = 0.063). Similarly, the ŌĆ£anxiety, fear, and uncertaintyŌĆØ item demonstrated poor reliability, with an ICC of 0.398 (95% CI 0.000ŌĆō0.697, p < 0.050). In contrast, the remaining items exhibited moderate-to-good reliability, with ICC values of 0.6 or higher. The Korean version of the PsAID-12 questionnaire demonstrated excellent internal consistency, with CronbachŌĆÖs alpha coefficients of 0.941 at the first and 0.951 at the second assessments (Supplementary Table 2).
Table 3 shows the SpearmanŌĆÖs correlation coefficient between the PsAID-12 score and disease activity indices, and Figure 2 shows the correlation between the first PsAID-12 questionnaire and disease activity indices in a dot plot. The PsAID-12 questionnaire showed strong correlations with most of the other variables.
We translated the English PsAID-12 questionnaire into the Korean version following established translation guidelines. The translation process was conducted by individuals with prior experience in translating English language questionnaires and was reviewed by both patients and experts [14]. The reliability of the translated Korean PsAID-12 questionnaire was assessed, and the agreement between the two surveys conducted 1 week apart was found to be good. Additionally, it demonstrated a strong correlation with disease activity, suggesting that the Korean version of the PsAID-12 questionnaire may be a valuable tool for evaluating patients with PsA.
Similar to other translation studies of the PsAID-12 questionnaire [15,16], the Korean version demonstrated high ICC values and strong correlations with disease activity indices, supporting its reliability and its ability to reflect the original questionnaire. Additionally, the total PsAID-12 score showed good reliability (ICC = 0.856). Items assessing physical domains, such as fatigue, discomfort, and pain, generally exhibited high ICC values, whereas items assessing coping and anxiety (items 8 and 9) showed lower values. This may be due to their sensitivity to psychological and situational factors, which can fluctuate over short periods. Furthermore, some participants might have reported reduced anxiety and uncertainty simply through study participation and receiving medical attention, which may have lowered scores at follow-up. Finally, conducting the follow-up survey online may also have contributed to variability in responses [17].
The importance of PROMs in PsA is well recognized. In a multinational short-term study conducted in Europe and the United States, quality of life indicators decreased significantly in patients with PsA as the severity of skin disease and peripheral joint symptoms increased [18]. It showed that in the disease management of PsA, an integrated assessment of the overall symptoms from the patientŌĆÖs perspective and an accurate clinical assessment are necessary. In particular, they confirmed that the EuroQol-VAS and PsAID-12 questionnaire are sensitive tools for assessing the quality of life of patients with PsA. Therefore, the PsAID-12 questionnaire is a useful tool for easily identifying patientsŌĆÖ needs that cannot be identified through disease activity.
The effect of PsA on daily life was investigated in 1,286 patients from eight countries [5]. The study reported findings based on adjusted PsAID-12 question categories, revealing that many patients experienced the effects of PsA on physical function and activity as well as on social, emotional, and work life. Additionally, differences in specific items among countries were observed, indicating that the burden of disease perceived by patients varies owing to social and cultural factors in addition to disease activity [6,7]. This highlights the need for a scale that captures patientsŌĆÖ experiences beyond disease activity when assessing patients with PsA. The translation of the PsAID-12 questionnaire into Korean was initiated with this objective, allowing us to better understand the characteristics and unmet needs of Korean patients with PsA and to compare them with those in other countries.
In our study, ŌĆ£fatigueŌĆØ had the highest score, whereas ŌĆ£embarrassment and/or shameŌĆØ and ŌĆ£social participationŌĆØ had the lowest scores. Most patients had been receiving long-term treatment, with many taking biologics and experiencing physical discomfort; however, despite limitations in physical activity or skin discomfort, they may not perceive significant impairments in social activities. Interestingly, in a multinational survey, the percentage of Taiwanese patients who reported that PsA affected their psychological and social aspects was lower than that of patients in European and American countries [5].
Several noteworthy observations can be drawn from this study. The relatively moderate mean DAPSA score despite a low swollen joint count may be attributed to the predominance of pain and fatigue over objective joint swelling in our study population. This pattern may reflect the characteristics of the recruited patients rather than a unique feature of Korean PsA patients, and further studies with larger multicenter cohorts are warranted to clarify this observation. Approximately 30% of patients in our study were receiving glucocorticoids. While systemic glucocorticoids are generally not recommended as a standard treatment for PsA, they are frequently used in clinical settings as a temporary measure to manage symptoms until disease-modifying anti-rheumatic drugs or biologics take effect. This concomitant use of glucocorticoids may also have contributed to the relatively moderate DAPSA scores observed in our cohort.
This study has some limitations, primarily related to its sample size. Although only 21 participants were included, which may have limited the statistical power for item-level psychometric analyses and contributed to relatively lower ICCs for some items, we emphasize that the sample size was not arbitrarily chosen. Specifically, a pilot cognitive debriefing with the first 10 patients yielded an ICC of 0.78 for the total PsAID-12 score. Using BonettŌĆÖs formula for estimating sample size with a desired 95% CI half-width of ┬▒ 0.10 and 80% statistical power, the required minimum sample size was calculated as 19 participants. Taking into account a 10% anticipated dropout rate, the final target was set at 21, which was successfully achieved. Therefore, we believe that the sample size was not only statistically justified but also sufficient to meet the precision requirements of the studyŌĆÖs psychometric objectives. Additionally, external factors such as the online administration of the second assessment and possible changes in treatment following outpatient visits may have influenced disease activity, thereby impacting testŌĆōretest reliability.
Despite these limitations, the Korean version of the PsAID-12 was carefully developed through a systematic process involving forwardŌĆōbackward translation, multidisciplinary expert review, and cognitive debriefing, ensuring both linguistic accuracy and cultural relevance. The translated questionnaire demonstrated good reliability and correlated well with established disease activity measures.
Therefore, the Korean PsAID-12 is a suitable tool for assessing the diverse impact of PsA in Korean patients. Its use in future clinical trials and longitudinal studies may provide deeper insights into the disease burden and treatment outcomes within this population.
1. The PsAID-12 questionnaire was successfully adapted into Korean through a rigorous translation process. The finalized Korean version demonstrated good reliability (ICC = 0.856) and strong correlations with disease activity indices, confirming its validity as a PROM for patients with PsA.
2. In Korea, where comprehensive data on PsA are limited, the PsAID-12 questionnaire is essential for capturing the multifaceted impact of the disease, including the physical, psychological, and social burdens, which may not be fully reflected in standard clinical assessments.
3. This study emphasizes that the PsAID-12 questionnaire can be useful for assessing the multidimensional impact of PsA in Korean patients, supporting its application in future clinical studies and cohort-based research.
Notes
Acknowledgments
We express our gratitude to the Spondyloarthritis Research Committee under the KCR for their support in finalizing the translated version. Eun Young Lee ŌĆō Seoul National University; Hong Ki Min ŌĆō Konkuk University; Hoon-Suk Cha ŌĆō Sungkyunkwan University; Hye Won Kim ŌĆō Seoul National University; Ji Hyeon Ju ŌĆō The Catholic University of Korea; Min-Chan Park ŌĆō Yonsei University; Sang-Hoon Lee ŌĆō Kyung Hee University; Seong-Kyu Kim ŌĆō Daegu Catholic University; Seong-Ryul Kwon ŌĆō Inha University; Seung-Cheol Shim ŌĆō Chungnam National University; Tae-Hwan Kim ŌĆō Hanyang University; Yeon-Ah Lee ŌĆō Kyung Hee University; Yong-Gil Kim ŌĆō University of Ulsan; and special thanks to Laure Gossec ŌĆō Sorbonne Universit├®; Piti├®-Salp├¬tri├©re Hospital, for her review.
CRedit authorship contributions
Bon San Koo: conceptualization, methodology, resources, investigation, data curation, formal analysis, validation, software, writing - original draft, writing - review & editing, visualization, project administration, funding acquisition; Jung Gon Kim: conceptualization, methodology, resources, investigation, writing - review & editing; Bo Young Yoon: conceptualization, methodology, resources, investigation, data curation, writing - review & editing; Ji-Hyoun Kang: methodology, resources, investigation, data curation, validation, writing - review & editing; Sung-Eun Choi: methodology, resources, investigation, data curation, writing - review & editing; Sooin Jung: methodology, resources, investigation, data curation; Seyeong Kim: methodology, resources, investigation, data curation, writing - review & editing; Tae-Jong Kim: conceptualization, methodology, resources, investigation, data curation, validation, writing - original draft, writing - review & editing, supervision, project administration
Figure┬Ā1
Flow diagram of the translation and cross-cultural adaptation process of the PsAID-12 questionnaire into Korean. The process followed established international guidelines and included five steps: forward translation, synthesis of the translations, backward translation, expert committee review, and field testing with 30 patients using cognitive debriefing interviews. PsAID-12, Psoriatic Arthritis Impact of Disease.
Figure┬Ā2
Correlations between the Korean PsAID-12 questionnaire and disease activity indices. Scatter plots demonstrate the relationship between PsAID-12 scores, DAPSA, pain VAS scores. Strong positive correlations were observed, supporting the construct validity of the Korean translation. PsAID-12, Psoriatic Arthritis Impact of Disease; DAPSA, Disease Activity in Psoriatic Arthritis; VAS, Visual Analog Scale.
Table┬Ā1
Baseline characteristics of patients with PsA
| Variables | Value (n = 21) |
|---|---|
| Age (yr) | 50.5 ┬▒ 9.3 |
| Female | 11 (52.4) |
| Disease duration (mo) | 79.8 ┬▒ 68.6 |
| BMI (kg/m2) | 24.7 ┬▒ 3.6 |
| CASPAR criteriaa) | |
| ŌĆā3 | 4 (19.0) |
| ŌĆā4 | 12 (57.1) |
| ŌĆā5 | 5 (23.8) |
| PsA subtype | |
| ŌĆāAxial PsA | 2 (9.5) |
| ŌĆāPeripheral PsA | 19 (90.5) |
| Skin biopsy | 9 (42.9) |
| Medication | |
| Methotrexate | 18 (85.7) |
| Tacrolimus | 2 (9.5) |
| ŌĆāLeflunomide | 2 (9.5) |
| ŌĆāSulfasalazine | 3 (14.3) |
| ŌĆāHydroxichlorquine | 1 (4.8) |
| ŌĆāTumor necrosis factor inhibitor | 7 (33.3) |
| ŌĆāInterleukin-17 inhibitor | 7 (33.3) |
| ŌĆāNonsteroidal anti-inflammatory drug | 10 (47.6) |
| ŌĆāGlucocorticoid | 6 (28.6) |
| C-reactive protein (mg/dL) | 0.2 ┬▒ 0.3 |
| Tender joint count | 4.7 ┬▒ 4.7 |
| Swollen joint count | 0.6 ┬▒ 1.5 |
| Disease activity indices | |
| ŌĆāVAS pain | 5.5 ┬▒ 2.7 |
| ŌĆāDAPSA | 15.0 ┬▒ 9.6 |
Table┬Ā2
Scores and agreements for each item in the Korean version of the PsAID-12 questionnaire
Table┬Ā3
Spearman correlation coefficients and p values between PsAID-12 questionnaire and disease activity indices
| PsAID-12 first | PsAID-12 second | DAPSA | VAS pain | |
|---|---|---|---|---|
| PsAID-12 first | 1 | 0.79*** | 0.66** | 0.67*** |
| PsAID-12 second | 0.79*** | 1 | 0.43 | 0.59** |
| DAPSA | 0.66** | 0.43 | 1 | 0.79*** |
| VAS pain | 0.67*** | 0.59** | 0.79*** | 1 |
REFERENCES
1. Coates LC, Helliwell PS. Psoriatic arthritis: state of the art review. Clin Med (Lond) 2017;17:65ŌĆō70.



2. Schett G, Rahman P, Ritchlin C, McInnes IB, Elewaut D, Scher JU. Psoriatic arthritis from a mechanistic perspective. Nat Rev Rheumatol 2022;18:311ŌĆō325.



3. Ogdie A, Weiss P. The epidemiology of psoriatic arthritis. Rheum Dis Clin North Am 2015;41:545ŌĆō568.



4. Shin D, Kim HJ, Kim DS, et al. Clinical features of psoriatic arthritis in Korean patients with psoriasis: a cross-sectional observational study of 196 patients with psoriasis using psoriatic arthritis screening questionnaires. Rheumatol Int 2016;36:207ŌĆō212.



5. Coates LC, Orbai AM, Azevedo VF, et al. Results of a global, patient-based survey assessing the impact of psoriatic arthritis discussed in the context of the Psoriatic Arthritis Impact of Disease (PsAID) questionnaire. Health Qual Life Outcomes 2020;18:173.




6. Husni ME, Merola JF, Davin S. The psychosocial burden of psoriatic arthritis. Semin Arthritis Rheum 2017;47:351ŌĆō360.


7. Puig L, van de Kerkhof PCM, Reich K, et al. A European subset analysis from the population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis shows country-specific features: results from psoriasis patients in Spain. J Eur Acad Dermatol Venereol 2017;31:1176ŌĆō1182.




8. Gossec L, de Wit M, Kiltz U, et al. A patient-derived and patient-reported outcome measure for assessing psoriatic arthritis: elaboration and preliminary validation of the Psoriatic Arthritis Impact of Disease (PsAID) questionnaire, a 13-country EULAR initiative. Ann Rheum Dis 2014;73:1012ŌĆō1019.


9. Acquadro C, Berzon R, Dubois D, et al. Incorporating the patientŌĆÖs perspective into drug development and communication: an ad hoc task force report of the Patient-Reported Outcomes (PRO) Harmonization Group meeting at the Food and Drug Administration, February 16, 2001. Value Health 2003;6:522ŌĆō531.


10. Choi AR, Park KJ, Kang JH, et al. The effectiveness of tumor necrosis factor-╬▒ blocker therapy in patients with axial spondyloarthritis who failed conventional treatment: a comparative study focused on improvement in ASAS Health Index. J Rheum Dis 2024;31:171ŌĆō177.



11. T├żlli S, Etcheto A, Fautrel B, et al. Patient global assessment in psoriatic arthritis - what does it mean? An analysis of 223 patients from the Psoriatic arthritis impact of disease (PsAID) study. Joint Bone Spine 2016;83:335ŌĆō340.


12. Tillett W, Costa L, Jadon D, et al. The ClASsification for Psoriatic ARthritis (CASPAR) criteria--a retrospective feasibility, sensitivity, and specificity study. J Rheumatol 2012;39:154ŌĆō156.


13. Beaton DE, Bombardier C, Guillemin F, Ferraz MB. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine (Phila Pa 1976) 2000;25:3186ŌĆō3191.



14. Choi JH, Kim TJ, Shin K, et al. The reliability and validity of a Korean translation of the ASAS Health Index and Environmental Factors in Korean patients with axial spondyloarthritis. J Korean Med Sci 2014;29:334ŌĆō337.




15. da Cruz Ribeiro E, Souza E, da Silva Carneiro SC, Yazbek MA, et al. Validation and clinical interpretability of PsAID - psoriatic arthritis impact of disease. Adv Rheumatol 2020;60:49.

16. Ziad├® N, Abbas N, Hmamouchi I, et al. Is the patient-perceived impact of psoriatic arthritis a global concept? An international study in 13 Arab countries (TACTIC study). Rheumatol Int 2024;44:885ŌĆō899.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement figure 1
Supplement figure 1 Print
Print



