 |
 |
| Korean J Intern Med > Volume 41(2); 2026 > Article |
|
Abstract
Background/Aims
Thyroid hormone suppression therapy (THST) is widely used for long-term management of differentiated thyroid cancer (DTC). However, studies have suggested that THST may increase the risk of atrial fibrillation (AF) in patients with DTC. We aimed to evaluate whether the risk of AF in patients with DTC differs according to treatment modality.
Methods
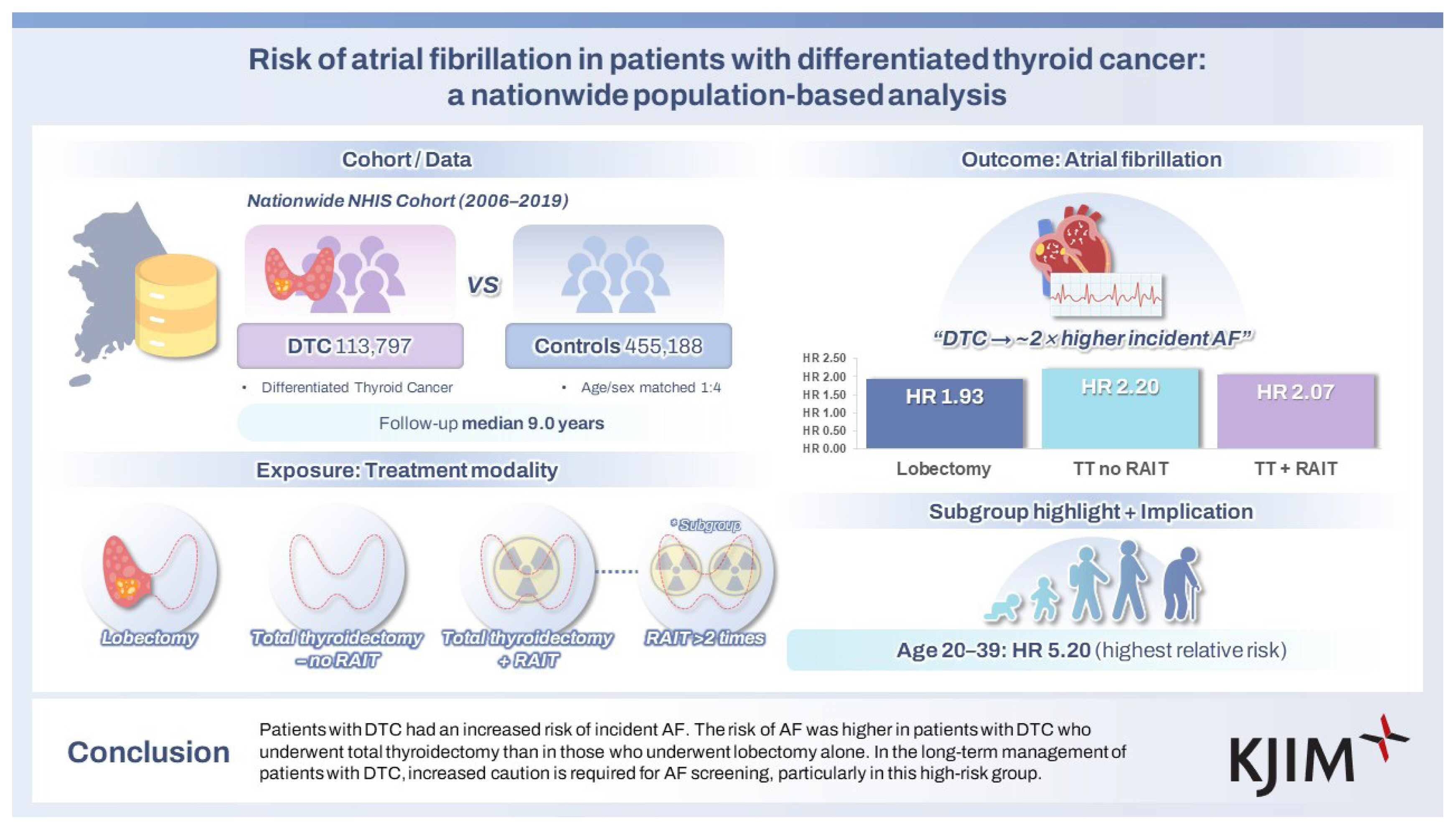
This retrospective cohort study used data from the Korean National Health Information Database between 2006 and 2019. We included 113,791 patients with DTC and age- and sex-matched 455,188 controls. The risk of AF was assessed according to the following treatment modalities: lobectomy or total thyroidectomy with or without radioactive iodine therapy (RAIT). A Cox proportional hazards model was used to compute hazard ratios (HRs) and 95% confidence intervals (CIs).
Results
Patients with DTC had a two-fold higher risk of incident AF than controls (HR, 2.07; 95% CI, 1.98ŌĆō2.17). All the treatment groups showed a higher risk of AF than the control group. Patients with DTC who underwent total thyroidectomy without RAIT (HR, 2.20; 95% CI, 2.06ŌĆō2.34) or with RAIT (HR, 2.07; 95% CI, 1.95ŌĆō2.20) had a higher risk of AF in contrast to those who underwent lobectomy (HR, 1.93; 95% CI, 1.72ŌĆō2.15).
Conclusions
Patients with DTC had an increased risk of incident AF. The risk of AF was higher in patients with DTC who underwent total thyroidectomy than those who underwent lobectomy alone. For long-term management of patients with DTC, increased vigilance is required for AF screening, particularly in this high-risk group.
Differentiated thyroid cancer (DTC), which includes papillary and follicular thyroid carcinomas, is the most common type of thyroid cancer. DTC has an excellent prognosis, with a 10-year overall survival rate > 90% [1] . The conventional treatment comprises thyroidectomy with or without radioactive iodine therapy (RAIT) and thyroid hormone suppression therapy (THST).
THST with levothyroxine replacement is the mainstay of DTC management following thyroidectomy [2] . The rationale for THST is to minimize thyroid stimulating hormone (TSH)-mediated tumor growth and reduce the risk of recurrence [3] . According to the current American Thyroid Association guidelines, postoperative TSH targets are recommended based on the risk of recurrence: < 0.1 mIU/L for high-risk patients, 0.1ŌĆō0.5 mIU/L for intermediate-risk patients, and 0.5ŌĆō2.0 mIU/L for low-risk patients [2] . In patients at low-to intermediate-risk who have undergone lobectomy, immediate initiation of levothyroxine therapy may not be necessary; however, routine levothyroxine replacement is commonly performed [4,5] . RAIT is typically performed after total thyroidectomy, either for remnant ablation or as an adjuvant therapy to reduce recurrence in intermediate-to-high-risk patients [2] .
Overt and subclinical hyperthyroidism are well-known risk factors for atrial fibrillation (AF) in the general population [6] . Furthermore, low TSH levels are associated with an increased risk of AF in clinically euthyroid individuals [7ŌĆō9] . Most patients with DTC undergo lifelong THST, especially those who undergo total thyroidectomy [2], concerns pertaining to the potential cardiovascular complications of iatrogenic hyperthyroidism induced by THST have been raised [10ŌĆō14] . Although previous studies have reported an increased risk of AF in patients with DTC [11ŌĆō13, 15], few have assessed whether the difference is based on treatment modalities. Therefore, in this retrospective cohort study, we aimed to evaluate the risk of AF in patients with DTC according to the treatment method.
We used health claims data from the National Health Information Database (NHID) of South Korea from 2006 to 2019. The NHID, maintained by the National Health Insurance Service of South Korea, is a public database that covers more than 97% of the Korean population. It includes demographic data, socioeconomic status, lifestyle questionnaire responses, National Health Checkup data, inpatient and outpatient diagnoses based on the International Classification of Diseases (ICD) codes, and medical treatment information [14] . This retrospective cohort study was approved by the Institutional Review Board of Myongji Hospital (IRB No.2024-12-019) and adhered to the tenets of the Declaration of Helsinki. The requirement for informed consent was waived because all data were deidentified and anonymized.
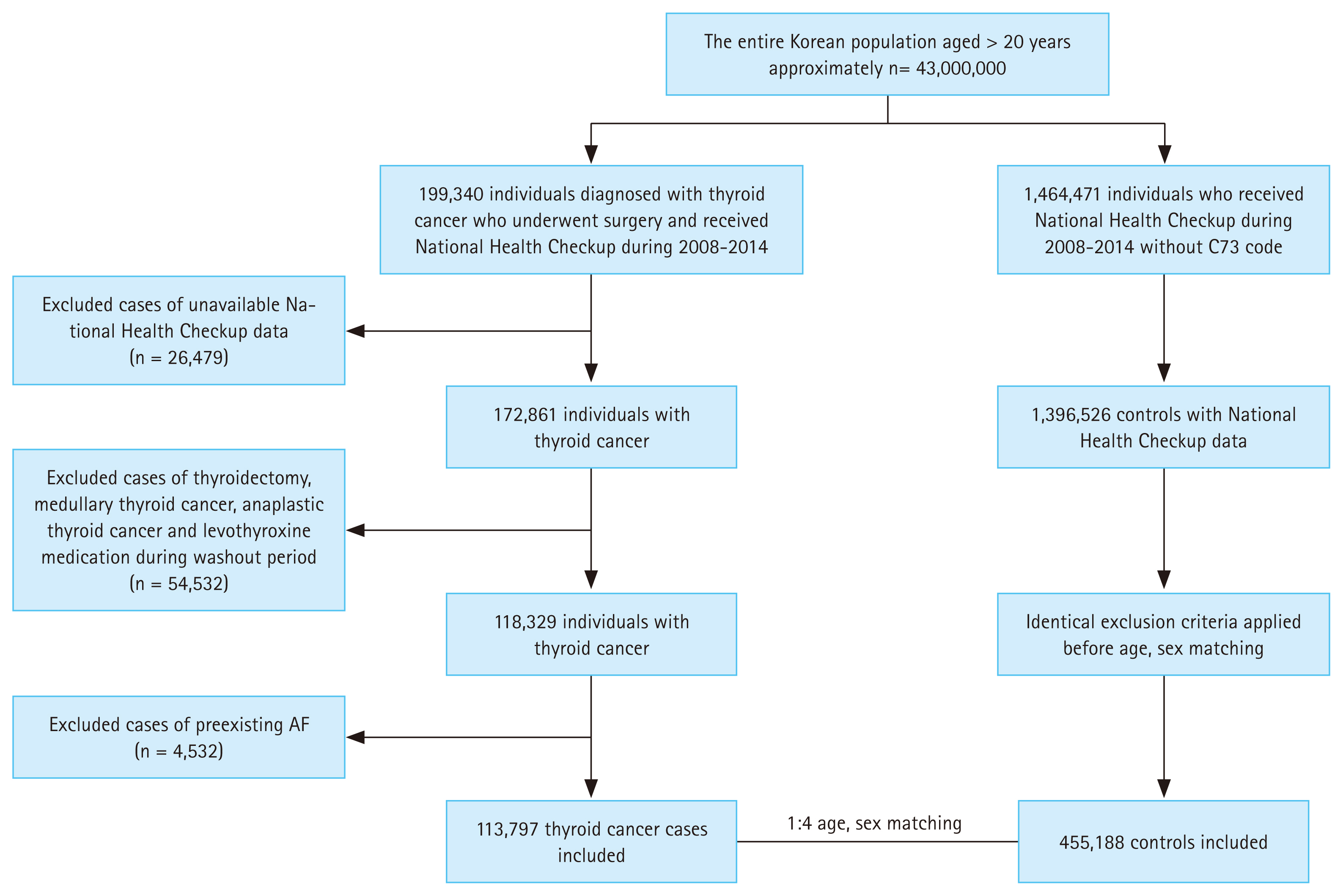
Data of patients diagnosed with DTC between January 2008 and December 2014 were extracted from the NHID. A total of 199,340 patients with DTC were initially identified using the ICD codes for thyroid cancer (C73) and those who underwent thyroidectomy [16] . A 2-year washout period (January 2006 to December 2007) was applied to exclude any previous history of thyroid cancer or thyroidectomy; thus, we identified newly diagnosed cases of DTC. The exclusion criteria were defined as patients with (1) missing demographic data, (2) medullary thyroid cancer (MTC) or anaplastic thyroid cancer, (3) history of levothyroxine use or thyroidectomy (P4551, P4552, P4553, P4554, and P4561) during the washout period, and (4) history of AF before the thyroid cancer index date (Fig. 1). The date of thyroidectomy was defined as the index date. MTC was defined as patients who underwent calcitonin testing more than twice within 1 year after the C73 code was assigned, and anaplastic thyroid cancer was defined as patient death occurring within 1 year of that assignment [16,17] . In total, 113,797 patients with DTC were included in the final cohort.
The control group data were extracted from the NHID using a random number generator. For each patient with DTC, four thyroid cancer-free individuals with available National Health Checkup data were randomly selected and matched by age and sex. To prevent bias due to differences in follow-up periods, we matched the follow-up periods for both groups. Identical exclusion criteria were applied to the control cohort before matching them to those of the DTC cohort.
The primary end point was the first occurrence of AF during the follow-up period. AF was defined as at least two outpatient visits or at least one hospitalization with ICD codes I480ŌĆōI484 and I489. This definition was adopted from previous epidemiological studies that used data from the NHID [18] .
Diabetes was defined as the presence of ICD-10 codes (E10ŌĆōE14) in conjunction with the prescription of antidiabetic medications. Hypertension was defined as a combination of ICD-10 codes (I10) and the prescription of antihypertensive drugs. Dyslipidemia was identified using the ICD-10 code (E78), along with the prescription of dyslipidemia medications. Household income was categorized into quartiles, with absolute poverty defined as a monthly household income below the minimum cost of living. Smoking status was classified as nonsmoker, former smoker, or current smoker.
Descriptive statistics were presented as mean values with standard deviations for continuous variables, whereas categorical variables were represented as numerical values and percentages. Group comparisons were conducted using t-tests for continuous variables and chi-square tests for categorical variables. Cumulative incidence rates (IRs) of AF during the follow-up period were plotted using a cumulative incidence function. Follow-up started on the index date and was censored on the date of outcome occurrence, date of death, or the last day of follow-up (December 31, 2019).
Cox proportional hazards regression models were used to compute the adjusted hazard ratios (HRs) when patients in the DTC and control groups were compared. The HRs were adjusted for age, sex, diabetes, hypertension, dyslipidemia, smoking, drinking, income, and body mass index (BMI). Subgroup analyses were performed according to age group (20ŌĆō39, 40ŌĆō64, and Ōēź 65 yr), sex (male or female), comorbidities, smoking status, alcohol intake, and BMI (< 25, Ōēź 25 kg/m2). All statistical analyses were performed using SAS version 14.0, and results with p values < 0.05 were considered significant.
This study included 113,797 patients with DTC and 455,188 matched controls. A comparison of the baseline characteristics of patients with DTC and controls is presented in Table 1. The mean age of the patients with DTC was 49.6 ┬▒ 11.1 years, and 15.6% were male. Patients with DTC have a higher prevalence of hypertension, diabetes, and dyslipidemia. They also had a higher BMI and a lower proportion of smokers than those in the control group. Among the patients with DTC, 83.5% underwent total thyroidectomy and 44.2% received RAIT.
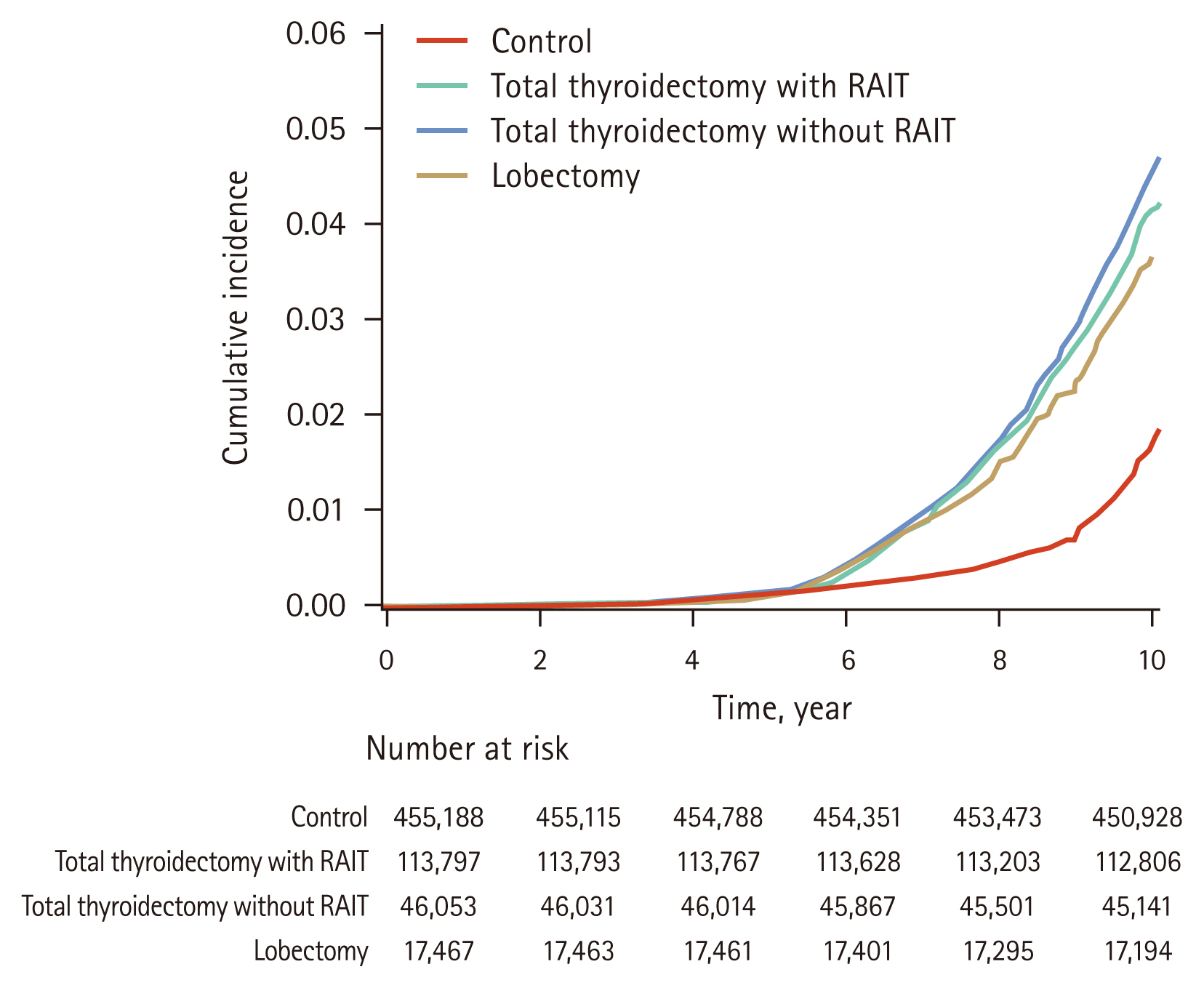
During a median follow-up period of 9.0 years (7.0ŌĆō10.1 yr), a total of 9,184 newly developed cases of AF were noted, with 2,868 in the patients with DTC and 6,316 in the controls (Table 2). The cumulative incidence of AF was higher in the DTC group than in the control group (Fig. 2). The IR per 1,000 person-years of AF was 3.17 in patients with DTC and 1.65 in the control group. In multivariate Cox-regression analysis, patients with DTC had a 2.07-fold higher risk of developing AF (adjusted HR, 2.07; 95% confidence interval [CI], 1.98ŌĆō2.17), compared to controls.
When analyzing the risk of AF according to the treatment method for DTC, the risk of AF was significantly increased in all treatment groups compared to that in the control group (Table 2, Fig. 2). IR of AF was 3.35 in the total thyroidectomy with RAI group, 3.25 in the total thyroidectomy without RAI, and 2.43 in the lobectomy group. The IR was 4.1 among patients with DTC who underwent total thyroidectomy with RAIT more than double. The adjusted HR for AF was 2.07 (95% CI, 1.95ŌĆō2.20) in the total thyroidectomy with RAIT group, 2.20 (95% CI, 2.06ŌĆō2.34) in the total thyroidectomy without RAIT group, and 1.93 (95% CI, 1.72ŌĆō2.15) in the lobectomy group, compared to the control group.
Results of subgroup analyses according to age, sex, presence of diabetes, hypertension, dyslipidemia, obesity (BMI Ōēź 25 kg/m2), and status of alcohol intake and smoking are presented in Table 3. The IR of AF was consistently higher in the DTC group than in the control group across all subgroup analyses. After adjusting for baseline demographic variables, patients with DTC still had a higher risk of AF than controls. In particular, the absolute IR of AF was higher in the older age group (Ōēź 65 yr) than in the younger ones (40ŌĆō64 yr and 20ŌĆō39 yr). However, HR increased in younger age groups. The HR for AF was highest among patients with DTC aged 20ŌĆō39 years (HR, 5.20; 95% CI, 4.10ŌĆō6.60), compared to those aged 40ŌĆō64 years (HR, 2.38; 95% CI, 2.24ŌĆō2.53) and those aged Ōēź 65 years (HR, 1.79; 95% CI, 1.66ŌĆō1.93). The HR for AF was also higher in males; patients without diabetes, hypertension, or dyslipidemia; smokers and those who consumed alcohol; and patients without obesity than in females; patients with diabetes, hypertension, and dyslipidemia; nonsmokers; nondrinkers; and patients with obesity. Significant interactions were observed between DTC and age, sex, diabetes, hypertension, alcohol intake, and BMI, whereas no significant interaction was found between DTC and dyslipidemia and smoking status.
In this nationwide retrospective cohort study, patients with DTC had a higher risk of incident AF than the controls, independent of various cardiovascular risk factors. Moreover, the risk of AF differed according to the treatment modality, with patients who underwent total thyroidectomy being at a higher risk than those who underwent lobectomy. An increased risk of AF in patients with DTC was repeatedly documented across all subgroup analyses and was particularly higher in younger age groups.
Previous studies have shown that patients with DTC have excellent overall survival rates, with similar mortality rates between patients with DTC and the general population [19,20] . Given the comparable life expectancy, the potential complications associated with cancer treatment should be considered in the long-term management of patients with DTC. Recent studies have reported that incident AF increases in patients with DTC who undergo THST with levothyroxine [11ŌĆō13, 15] . Our study also confirmed the elevated risk of AF in patients with DTC, which is consistent with previous findings. THST in patients with DTC can lead to iatrogenic hyperthyroidism, increasing the risk of AF through positive chronotropic effects and elevated conduction velocity [21] . Iatrogenic hyperthyroidism also induces myocardial strain, increases ventricular mass, and increases left atrial pressure, all of which promote AF development in patients with DTC [22] .
In our study, we further investigated whether the risk of AF differed according to treatment modality in patients with DTC. We found that patients who underwent total thyroidectomy had a higher risk of developing AF than those who underwent lobectomy, regardless of the RAIT status. In real-world practice, levothyroxine replacement is not routinely required in patients who undergo lobectomy to suppress TSH levels, with 14ŌĆō65% receiving levothyroxine therapy, in contrast to patients who were treated with total thyroidectomy [23,24] . Moreover, patients who undergo total thyroidectomy generally receive higher doses of levothyroxine to maintain low TSH levels than those who undergo lobectomy [25] . In a retrospective cohort study using a claims database, Suh et al. [10] found that patients with DTC receiving higher doses of levothyroxine were more likely to develop AF than those receiving lower doses. Collectively, these findings suggested that THST contributes to the development of AF in patients with DTC.
Notably, the patients who underwent total thyroidectomy without RAIT had a higher risk of AF than those who underwent total thyroidectomy with RAIT. Despite the lack of data on TSH levels and indications for RAIT in our study, this finding was unexpected because patients receiving RAIT are more likely to undergo more intensive THST. Our findings suggest that although THST is an important contributor to AF risk, the degree of TSH suppression may not be proportionally associated with an increased risk of AF in patients with DTC. Similarly, previous studies did not identify a linear relationship between TSH levels and AF in patients with DTC [26,27] . Klein Hesselink et al. [26] suggested that THST might trigger AF in patients predisposed to other cardiovascular risk factors or genetic susceptibilities. In such cases, the degree of TSH suppression may be less relevant, possibly explaining the loss of a dose-response relationship between AF and TSH levels. Overall, exogenous thyrotoxicosis induced by THST may have a distinct physiology in the cardiovascular system compared with endogenous hyperthyroidism [28] .
In this study, patients who underwent RAIT more than twice had the highest risk of developing AF, suggesting that repeated RAIT may play a role in the occurrence of AF. Several plausible explanations for these findings exist. In experimental studies, radioiodine is taken up by cells via the membrane sodium-iodide symporter, and its expression has been identified in certain extrathyroidal tissues, including in cardiac tissue [29] . Local uptake of radioiodine in the cardiac tissue may induce myocardial damage and rhythm disturbances. Another possible mechanism involves fluctuations in thyroid hormone levels during repeated levothyroxine withdrawal may exacerbate dysrhythmias [26,30] . Additionally, repeated administration of T3 along with variations in cholesterol and inflammatory cytokine levels may contribute to an increased risk of AF.
Epidemiological studies have shown that the prevalence of AF in the general population increases with age [31] . In the present study, higher IRs of AF were observed in older patients with DTC. The IR per 1,000 PYS was 10.64 in the subgroup aged Ōēź 65 years, which was more than ten-fold higher than in the subgroup aged 20ŌĆō39 years. However, the adjusted HRs for AF in patients with DTC compared to controls were higher in the younger age group than in the older age group. Therefore, the absolute incidence of AF was lower in patients with DTC in the younger age group, and the effect of DTC on AF was stronger in the younger age group than in the older age group. This may be attributed to the relatively stronger contribution of cardiovascular comorbidities to AF risk in the older group, which may attenuate the effect of DTC on AF occurrence compared to that in the younger age group. Given the excellent cancer-related prognosis of patients with DTC, these findings suggest that close monitoring for AF is needed, especially in younger patients with DTC.
In the general population, AF is more prevalent in males [31] . However, few studies have examined the effect of sex on AF development in patients with DTC. Zoltek et al. [32] reported that the standardized incidence ratio of AF was slightly higher in male patients than in females. In contrast, another study found that both males and females with DTC exhibited similar risks of cardiovascular morbidity, although the specific risk of AF was not addressed in their study [33] . In this study, the association between DTC and AF was more pronounced in males than in females. Although the exact mechanism underlying sex-related differences remains unclear, iatrogenic hyperthyroidism and RAIT-related myocardial damage may be more significant in men.
In the subgroup analyses, the association between AF and DTC was significantly modified by cardiovascular risk factors, as indicated by the interaction effects. While DTC was associated with a higher overall risk of AF, this association was more pronounced in individuals without diabetes, hypertension, or obesity. In contrast, among individuals with established risk factors, such as diabetes, hypertension, and obesity, the incremental risk attributable to DTC may be attenuated, possibly because of the already elevated baseline risk driven by metabolic and hemodynamic disturbances. Notably, alcohol consumption appeared to strengthen the association between DTC and AF, whereas no significant interaction was observed with dyslipidemia or smoking status. The complex interplay between the DTC-related treatment effects and conventional AF risk factors warrants further investigation.
This study has several limitations. First, detailed clinical information on the patients with DTC, such as TSH levels and total RAI dose, was not available. Although levothyroxine use was applied as an exclusion criterion during the initial cohort selection, data on levothyroxine dosage, including the weight-adjusted dose (mcg/kg), were not included in the analysis of AF risk. Second, we used data from the Korean NHID; therefore, the findings of our study may not be generalizable to other ethnic groups. Genetic components are known to strongly influence the pathogenesis of AF [34], therefore ethnic variability in the development of AF should be considered. Thirdly, comorbidities such as coronary artery disease, heart failure, and chronic kidney disease, which may influence the risk of AF, were not fully considered in the analyses. However, the key strengths of this study are its large-scale design and long-term follow-up, which enabled stratified analyses based on treatment modalities. We also carefully considered various confounders that affect the development of AF.
In conclusion, patients with DTC had a higher risk of incident AF than controls. The risk of AF differed according to the treatment modality, with a higher risk in patients who underwent total thyroidectomy than in those who underwent lobectomy. Given the excellent cancer-specific survival of patients with DTC, targeted screening is required in patients at high risk for AF.
Notes
Figure┬Ā2
Cumulative incidence function curves of AF by treatment modality. The solid, short-dashed, long-dashed, and dotted lines represent the control group, total thyroidectomy with RAIT group, total thyroidectomy without RAIT group, and lobectomy group, respectively. AF, atrial fibrillation; RAIT, radioactive iodine therapy.
Table┬Ā1
Baseline characteristics of study cohort
Table┬Ā2
Risk of AF in patients with differentiated thyroid cancer compared to controls
| Number | AF event rate | IR per 1,000 PYS | Unadjusted HR (95% CI) | Adjusted HR (95% CI)a) | |
|---|---|---|---|---|---|
| Controls | 455,188 | 6,316 | 1.65 | Reference (1) | Reference (1) |
| Thyroid cancer | 113,797 | 2,868 | 3.17 | 2.55 (2.44ŌĆō2.67) | 2.07 (1.98ŌĆō2.17) |
| ŌĆāTotal thyroidectomy with RAIT | 50,277 | 1,361 | 3.35 | 2.59 (2.44ŌĆō2.75) | 2.07 (1.95ŌĆō2.20) |
| ŌĆāŌĆāNumber of RAIT: 1 | 40,041 | 1,012 | 3.15 | 2.49 (2.34ŌĆō2.67) | 1.98 (1.85ŌĆō2.11) |
| ŌĆāŌĆāNumber of RAIT: Ōēź 2 | 10,236 | 349 | 4.10 | 2.90 (2.60ŌĆō3.24) | 2.41 (2.16ŌĆō2.68) |
| ŌĆāTotal thyroidectomy without RAIT | 46,053 | 1,176 | 3.25 | 2.68 (2.51ŌĆō2.85) | 2.20 (2.06ŌĆō2.34) |
| ŌĆāLobectomy | 17,467 | 331 | 2.43 | 2.08 (1.85ŌĆō2.32) | 1.93 (1.72ŌĆō2.15) |
Table┬Ā3
Risk of AF among patients with differentiated thyroid cancer compared to controls according to age, sex and cardiovascular risk factors
| Subgroup | Thyroid cancer | Number | AF event | IR per 1,000 PYS | Crude HR (95% CI) | Adjusted HR (95% CI)a) | p for interaction |
|---|---|---|---|---|---|---|---|
| Age (yr) | < 0.001 | ||||||
| ŌĆā20ŌĆō39 | No | 74,980 | 159 | 0.25 | Reference | Reference | |
| Yes | 18,745 | 155 | 1.04 | 5.71 (4.54ŌĆō7.19) | 5.20 (4.10ŌĆō6.60) | ||
| ŌĆā40ŌĆō64 | No | 335,492 | 3,660 | 1.30 | Reference | Reference | |
| Yes | 83,228 | 1,746 | 2.63 | 2.74 (2.58ŌĆō2.91) | 2.38 (2.24ŌĆō2.53) | ||
| ŌĆāŌēź 65 | No | 44,716 | 2,497 | 6.72 | Reference | Reference | |
| Yes | 11,824 | 967 | 10.64 | 1.98 (1.84ŌĆō2.14) | 1.79 (1.66ŌĆō1.93) | ||
| Sex | 0.025 | ||||||
| ŌĆāMale | No | 70,952 | 1,491 | 2.49 | Reference | Reference | |
| Yes | 17,738 | 689 | 5.03 | 2.97 (2.71ŌĆō3.26) | 2.40 (2.18ŌĆō2.64) | ||
| ŌĆāFemale | No | 384,236 | 4,825 | 1.50 | Reference | Reference | |
| Yes | 96,059 | 2,179 | 2.84 | 2.47 (2.34ŌĆō2.60) | 2.14 (2.03ŌĆō2.25) | ||
| Diabetes | < 0.001 | ||||||
| ŌĆāNo | No | 423,170 | 5,156 | 1.45 | Reference | Reference | |
| Yes | 101,589 | 2,245 | 2.78 | 2.58 (2.45ŌĆō2.71) | 2.32 (2.20ŌĆō2.44) | ||
| ŌĆāYes | No | 32,018 | 1,160 | 4.48 | Reference | Reference | |
| Yes | 12,208 | 623 | 6.56 | 1.81 (1.64ŌĆō2.0) | 1.80 (1.63ŌĆō2.0) | ||
| Hypertension | < 0.001 | ||||||
| < 0.001ŌĆāNo | No | 366,311 | 3,032 | 0.99 | Reference | Reference | |
| Yes | 83,548 | 1,226 | 1.84 | 2.54 (2.37ŌĆō2.72) | 2.42 (2.26ŌĆō2.60) | ||
| ŌĆāYes | No | 88,877 | 3,284 | 4.39 | Reference | Reference | |
| Yes | 30,249 | 1,642 | 6.86 | 2.01 (1.89ŌĆō2.14) | 2.02 (1.90ŌĆō2.15) | ||
| Dyslipidemia | 0.074 | ||||||
| ŌĆāNo | No | 383,183 | 4,095 | 1.27 | Reference | Reference | |
| Yes | 81,189 | 1,537 | 2.35 | 2.43 (2.28ŌĆō2.58) | 2.30 (2.17ŌĆō2.45) | ||
| ŌĆāYes | No | 72,005 | 2,221 | 3.78 | Reference | Reference | |
| Yes | 32,608 | 1,331 | 5.35 | 1.93 (1.8ŌĆō2.07) | 2.01 (1.87ŌĆō2.16) | ||
| Smoke | 0.052 | ||||||
| ŌĆāYes | No | 76,190 | 1,266 | 2.04 | Reference | Reference | |
| Yes | 16,606 | 533 | 4.18 | 2.89 (2.60ŌĆō3.21) | 2.35 (2.11ŌĆō2.62) | ||
| ŌĆāNo | No | 378,998 | 5,050 | 1.58 | Reference | Reference | |
| Yes | 97,191 | 2,335 | 3.01 | 2.51 (2.39ŌĆō2.64) | 2.16 (2.05ŌĆō2.27) | ||
| Alcohol intake | < 0.001 | ||||||
| ŌĆāNo | No | 307,769 | 4,679 | 1.80 | Reference | Reference | |
| Yes | 81,506 | 2,205 | 3.37 | 2.38 (2.26ŌĆō2.51) | 2.07 (1.97ŌĆō2.19) | ||
| ŌĆāYes | No | 147,419 | 1,637 | 1.34 | Reference | Reference | |
| Yes | 32,291 | 663 | 2.66 | 3.01 (2.74ŌĆō3.30) | 2.66 (2.42ŌĆō2.92) | ||
| BMI (kg/m2) | < 0.001 | ||||||
| ŌĆā< 25 | No | 317,054 | 3,583 | 1.34 | Reference | Reference | |
| Yes | 73,577 | 1,554 | 2.65 | 2.60 (2.44ŌĆō2.76) | 2.29 (2.15ŌĆō2.44) | ||
| ŌĆāŌēź 25 | No | 138,134 | 2,733 | 2.36 | Reference | Reference | |
| Yes | 40,220 | 1,314 | 4.12 | 2.35 (2.19ŌĆō2.51) | 2.10 (1.96ŌĆō2.25) |
REFERENCES
1. Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin 2021;71:7ŌĆō33.




2. Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016;26:1ŌĆō133.


3. Carayon P, Thomas-Morvan C, Castanas E, Tubiana M. Human thyroid cancer: membrane thyrotropin binding and adenylate cyclase activity. J Clin Endocrinol Metab 1980;51:915ŌĆō920.


4. Wilson M, Patel A, Goldner W, Baker J, Sayed Z, Fingeret AL. Postoperative thyroid hormone supplementation rates following thyroid lobectomy. Am J Surg 2020;220:1169ŌĆō1173.


5. Park S, Kim WG, Han M, et al. Thyrotropin suppressive therapy for low-risk small thyroid cancer: a propensity score-matched cohort study. Thyroid 2017;27:1164ŌĆō1170.


6. Cho YY, Kim B, Choi D, et al. GravesŌĆÖ disease, its treatments, and the risk of atrial fibrillation: a Korean population-based study. Front Endocrinol (Lausanne) 2022;13:1032764.



7. Sawin CT, Geller A, Wolf PA, et al. Low serum thyrotropin concentrations as a risk factor for atrial fibrillation in older persons. N Engl J Med 1994;331:1249ŌĆō1252.



8. Auer J, Scheibner P, Mische T, Langsteger W, Eber O, Eber B. Subclinical hyperthyroidism as a risk factor for atrial fibrillation. Am Heart J 2001;142:838ŌĆō842.


9. Jabbar A, Pingitore A, Pearce SH, Zaman A, Iervasi G, Razvi S. Thyroid hormones and cardiovascular disease. Nat Rev Cardiol 2017;14:39ŌĆō55.



10. Suh B, Shin DW, Park Y, et al. Increased cardiovascular risk in thyroid cancer patients taking levothyroxine: a nationwide cohort study in Korea. Eur J Endocrinol 2019;180:11ŌĆō20.


11. Lee EK, Ahn HY, Ku EJ, et al. Cardiovascular outcomes in thyroid cancer patients treated with thyroidectomy: a meta-analysis. J Clin Endocrinol Metab 2021;106:3644ŌĆō3654.



12. Yang X, Guo N, Gao X, Liang J, Fan X, Zhao Y. Meta-analysis of TSH suppression therapy and the risk of cardiovascular events after thyroid cancer surgery. Front Endocrinol (Lausanne) 2022;13:991876.



13. Yu J, Kaur R, Ayeni FE, Eslick GD, Edirimanne S. Cardiovascular outcomes of differentiated thyroid cancer patients on long term TSH suppression: a systematic review and meta-analysis. Horm Metab Res 2023;55:379ŌĆō387.


14. Shin DW, Cho B, Guallar E. Korean National Health Insurance Database. JAMA Intern Med 2016;176:138.


15. Kostopoulos G, Doundoulakis I, Antza C, et al. Incident atrial fibrillation in patients with differentiated thyroid cancer: a meta-analysis. Endocr Relat Cancer 2021;28:325ŌĆō335.



16. Ahn HY, Lee J, Kang J, Lee EK. Increased risk of diabetes mellitus and hyperlipidemia in patients with differentiated thyroid cancer. Eur J Endocrinol 2024;190:248ŌĆō255.



17. Ahn HY, Chae JE, Moon H, Noh J, Park YJ, Kim SG. Trends in the diagnosis and treatment of patients with medul lary thyroid carcinoma in Korea. Endocrinol Metab (Seoul) 2020;35:811ŌĆō819.




18. Lee SR, Choi EK, Han KD, Cha MJ, Oh S. Trends in the incidence and prevalence of atrial fibrillation and estimated thromboembolic risk using the CHA2DS2-VASc score in the entire Korean population. Int J Cardiol 2017;236:226ŌĆō231.


19. Kim KJ, Jang S, Kim KJ, et al. Actual causes of death in thyroid cancer patients in Korea: a Nationwide Case Control Cohort Study. Eur J Endocrinol 2020;182:103ŌĆō110.


21. Bielecka-Dabrowa A, Mikhailidis DP, Rysz J, Banach M. The mechanisms of atrial fibrillation in hyperthyroidism. Thyroid Res 2009;2:4.




22. Biondi B, Palmieri EA, Lombardi G, Fazio S. Effects of thyroid hormone on cardiac function: the relative importance of heart rate, loading conditions, and myocardial contractility in the regulation of cardiac performance in human hyperthyroidism. J Clin Endocrinol Metab 2002;87:968ŌĆō974.


23. Cooper D, Kaur R, Ayeni FE, Eslick GD, Edirimanne S. Hypothyroidism after hemithyroidectomy: a systematic review and meta-analysis. Thyroid Res 2024;17:18.




24. Park S, Jeon MJ, Song E, et al. Clinical features of early and late postoperative hypothyroidism after lobectomy. J Clin Endocrinol Metab 2017;102:1317ŌĆō1324.


25. Kim MS, Lee JW, Hyun MK, Song YS. Risk of subsequent primary cancers in thyroid cancer survivors according to the dose of levothyroxine: a nationwide cohort study. Endocrinol Metab (Seoul) 2024;39:288ŌĆō299.




26. Klein Hesselink EN, Lefrandt JD, Schuurmans EP, et al. Increased risk of atrial fibrillation after treatment for differentiated thyroid carcinoma. J Clin Endocrinol Metab 2015;100:4563ŌĆō4569.


27. Wang Y, Liu C, Wang J, Zhang Y, Chen L. Iodine-131 induces apoptosis in human cardiac muscle cells through the p53/Bax/caspase-3 and PIDD/caspase-2/tŌĆÉBID/cytochrome c/caspase-3 signaling pathway. Oncol Rep 2017;38:1579ŌĆō1586.



28. Hoermann R, Midgley JEM, Larisch R, Dietrich JW. Heterogenous biochemical expression of hormone activity in subclinical/overt hyperthyroidism and exogenous thyrotoxicosis. J Clin Transl Endocrinol 2020;19:100219.



29. Spitzweg C, Joba W, Eisenmenger W, Heufelder AE. Analysis of human sodium iodide symporter gene expression in extrathyroidal tissues and cloning of its complementary deoxyribonucleic acids from salivary gland, mammary gland, and gastric mucosa. J Clin Endocrinol Metab 1998;83:1746ŌĆō1751.


30. An JH, Song KH, Kim DL, Kim SK. Effects of thyroid hormone withdrawal on metabolic and cardiovascular parameters during radioactive iodine therapy in differentiated thyroid cancer. J Int Med Res 2017;45:38ŌĆō50.




31. Staerk L, Sherer JA, Ko D, Benjamin EJ, Helm RH. Atrial fibrillation: epidemiology, pathophysiology, and clinical outcomes. Circ Res 2017;120:1501ŌĆō1517.


32. Zoltek M, Andersson TM, Hedman C, Ihre-Lundgren C, Nordenvall C. Cardiovascular incidence in 6900 patients with differentiated thyroid cancer: a Swedish nationwide study. World J Surg 2020;44:436ŌĆō441.



-
METRICS

-
- 0 Crossref
- 0 Scopus
- 208 View
- 85 Download
- Related articles
-
Role of catheter ablation in patients with heart failure and atrial fibrillation2025 July;40(4)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


