 |
 |
| Korean J Intern Med > Volume 37(2); 2022 > Article |
|
Abstract
Background/Aims
Germline mutations of the rearranged during transfection (RET) gene cause multiple endocrine neoplasia type 2 (MEN2). About 85% of RET mutations in MEN2 occur in codon Cys634. The RET D631Y mutation has recently been discovered, and we have studied its molecular expression and clinical consequences.
Methods
We analyzed the clinical characteristics of a total of 34 D631Y variant MEN2 individuals from seven families. We also constructed wild-type and mutant C630Y, D631Y, and C634R/W expression vectors and investigated their effects on signaling pathways and ability to correct the phenotypes of RET mutant cells.
Results
The median ages at diagnosis of pheochromocytoma and medullary thyroid carcinoma (MTC) were higher in patients with RET D631Y variant MEN2 than in those with the C634R/W variant (49:53.5 years vs. 33.5:27 years, respectively), and the penetration of the D631Y mutation with respect to MTC was lower than that of the C634R/W mutation (32.3% vs. 90%). The effects of the mutant vectors on phosphorylation of RET signaling molecules and focus formation were significantly different from those of wild type, but there were no significant differences between the mutants. D631Y scored significantly higher for chemotaxis and wound healing than C630Y, but lower than C634R and C634W.
Multiple endocrine neoplasia type 2 (MEN2) is an autosomal dominant disease that causes tumors in two or more endocrine organs due to mutations in the rearranged during transfection (RET) gene [1,2]. RET is located on chromosome 10 (10q11.2), consists of 21 exons, and encodes a receptor tyrosine protein kinase (RTK) [1,3]. RET is composed of a cysteine-rich extracellular transmembrane domain, and an intracellular domain with tyrosine kinase activity [1,4–6]. MEN2 is associated with medullary thyroid carcinoma (MTC), pheochromocytoma (PCC), and primary hyperparathyroidism (PHPT), while MEN type 3 (MEN3) causes MTC and PCC, but is not associated with PHPT [1,2,5,7,8]. In MEN2, 95% of the RET mutations occur in the cysteine-rich extracellular domain, and 85% of these are in the Cys634 codon [1,2,6,9]. About half of the Cys634 codon mutations found in MEN2 are Cys634Arg (C634R) substitutions [1,6,8,10].
There is a high correlation between the location of RET gene mutations and their clinical phenotypes in MEN2 [2,11]. Identification of the locations of RET mutations has facilitated the prognosis and management of MEN2/MEN3 patients, who have to take thyroxine throughout their lives after prophylactic thyroidectomy [1,10]. The original American Thyroid Association (ATA) guidelines categorize RET mutations from D to A based on their risk levels for MTC (highest to lowest) and provide recommendations as to the timing of prophylactic thyroidectomy [12]. Recently, the task force recommends that category D be changed to a new category, ‘highest risk’ and category C to ‘high risk,’ and that categories A and B be combined into a new category, ‘moderate risk’ [2]. For MTC in patients with mutations in RET, the ATA classifies the risk of MTC for Cys634Phe/Gly/Arg/Ser/Trp/Tyr variant MEN2 as “high.” The risk of MTC in RET Asp631Tyr (D631Y) MEN2 mutants, like that of MEN2 mutants at other sites, is currently classified as “moderate” [2]. However, this classification appears to be based on one of the first reports of this mutation [13], in which the age of onset and clinical details of MTC were not provided. On the other hand, since Elston et al. [14] reported that the youngest patient with MTC associated with the D631Y mutation was 22 years old, this mutation should be reclassified. However, because not many RET D631Y cases have been reported so far, it is unclear if the risk of MTC is actually moderate [1]. The ATA’s MTC management guidelines [2,6] are strict, and recommend prophylactic thyroidectomy before the age of 5 for Cys634 variants [2,15]. However, their guidelines for RET D631Y variant MEN2, which has been reported recently in Korea [16,17], New Zealand [14], and the United States [18], still lack evidence-based data. Furthermore, no criterion for the timing of prophylactic thyroidectomy has been recommended, and clinical and/or basic research data comparing the MTC risks in patients with the D631Y and Cys634 mutations are limited. The purpose of this study was to analyze the clinical features of 7 D631Y families, including four families already described in the literature and three diagnosed in our hospital. In addition, the effects of the D631Y variant on RET-associated signaling molecules and tumor formation were investigated in a laboratory setting. In this way we aimed to obtain evidence-based data for the risk level of D631Y variant MEN2.
This case was previously reported in Korean [17]. Briefly, a 62-year-old woman presented with palpitation and headache. She had been taking anti-hypertensive and oral hypoglycemic agents for 10 years. Twenty-four-hour urine metanephrine (MN) and vanillylmandelic acid (VMA) were elevated, as was serum calcitonin level (154.87 pg/mL; normal <11.5 pg/mL). Ultrasonography of the thyroid gland showed hypoechoic masses (right side size, 1.5 cm; left side size, 0.5 cm) with microcalcifications. Abdominal computed tomography (CT) revealed heterogeneously-enhanced bilateral adrenal masses (right side, 3.5 cm; left side, 6.0 cm). 123I-meta-iodobenzylguanidine scintigraphy revealed increased uptake over the bilateral adrenal masses. The patient underwent staged surgery for bilateral adrenalectomy and total thyroidectomy. Gene testing revealed a germline D631Y mutation in RET. One of the proband’s children tested positive for the D631Y mutation, but she declined screening (Fig. 1A).
A 66-year-old woman was referred to an endocrinologist due to a high calcitonin level of 45.6 pg/mL after total thyroidectomy with therapeutic central node dissection for MTC. Two tumors, 1.2 cm, and 0.7 cm in size, respectively, were buried in the surgical specimen. A total of 27 lymph nodes were removed from level II to level VI on both sides. Metastasis was found in 2 lymph nodes, one at right level IV and the other at level VI. According to a medical history review, her preoperative calcitonin level was 1,750 pg/mL. She had been treated for high blood pressure for 15 years and diabetes for 2 years. On abdominal CT, adrenal tumors of 1.0 cm on the right and 0.6 cm on the left were observed. Twenty-four-hour urine MN and VMA were slightly above upper normal limits. Genetic testing revealed the D631Y mutation in the patient and his eldest son. A cervical ultrasound examination of the son revealed two hypoechoic nodules of 0.3 cm, and blood calcitonin was slightly elevated at 13 pg/mL, but all screening results for PCCs were normal. He declined surgery on the thyroid gland (Fig. 1B)
A 64-year-old woman was admitted to the emergency room due to dizziness and vomiting and was hospitalized after tumors of 3.3 cm on the right and 4.6 cm on the left were found by abdominal CT. She had been under treatment for high blood pressure and diabetes for 7 years. There was a marked increase in 24-hour urine MN and VMA, and blood calcitonin level was normal. The patient underwent bilateral adrenal resection. After examination based on family history, her 66-year-old brother also underwent bilateral adrenal resection, due to an adrenal mass of 4.2 cm on the right and one of 4.5 cm on the left. The proband and her son, brother and nephew all carried a D631Y mutation, and the son and nephew were diagnosed with bilateral PCC on CT scan, but no further clinical information is available (Fig. 1C).
Genomic DNA (gDNA) was extracted from peripheral leukocytes of each family’s proband and family members using a FlexiGene DNA Kit (Qiagene, Hilden, Germany), and the coding exons and flanking intronic sequences of RET were amplified by standard polymerase chain reaction (PCR). The amplified products were analyzed for purity and size by electrophoresis on 2% agarose gels, and sequenced with an ABI 3730XL Genetic Analyzer (Applied Biosystems, Foster City, CA, USA).
We screened members of the seven families already mentioned, comprising four families previously described in the literature and three diagnosed in our hospital. The clinical phenotypes of 34 D631Y mutant MEN2 individuals identified in the seven families were analyzed (Tables 1 and 2). The study was approved by the Institutional Review Board of Incheon St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea (IRB approval No. OC16TISI0099). All participating members of the three families diagnosed in our hospital provided written informed consent.
To investigate the effects of RET mutations on signaling pathways as shown in Fig. 2A, we first created an expression vector carrying the human RET gene (GenBank accession no. NM_020975.6). Wild type RET was amplified from control cDNA using KOD Plus Neo Taq polymerase (Toyobo, Japan). A HindIII site was introduced into upstream primer (5′-GGGAGACCCAAGCTTATGGCGAAGGCGACGTCCGG-3′), and a KpnI site into downstream primer (5′-GTCCTTGTAATCGGTACCGAATCTAGTAAATGCATGGGA-3′). The PCR product was cloned into pcDNA3 vector with a C-terminal flag tag using an EZ fusion cloning kit (Enzynomics, Daejeon, Korea). The various RET mutation types (C630Y, D631Y, C634R, and C634W) were constructed by mutating the wild type expression vector (RET-wild-Flag/pcDNA3) using a QuikChange site-directed mutagenesis kit (Agilent Technologies, Santa Clara, CA, USA) (Fig. 2B). All expression vectors were sequenced directly by BigDye Terminator Cycle sequencing with a 3730XL DNA Analyzer (Applied Biosystems).
Prior to the experiments, we confirmed that monkey kidney fibroblast (COS7) and mouse embryonic fibroblast (NIH3T3) cell lines did not express endogenous RET. Experimental results on the signaling effects showed that COS7 cells were more differentiated. In the cell proliferation experiments, NIH3T3 cells were used for direct comparison with the results of Asai et al. [19,20]. COS7 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM, Gibco BRL, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (Gibco BRL) and 1% penicillin-streptomycin (Gibco BRL). NIH3T3 cells were maintained in DMEM supplemented with 10% bovine calf serum (BCS, Gibco BRL). Cells were transfected with an implemented electroporation device (Neon Transfection System, Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. For COS7 cells, parameters were set at 950 pulse voltage and 30 pulse widths, and two pulses were used; for NIH3T3 cells, parameters were set at 950 pulse voltage and 40 pulse widths, and two pulse were again used. For transfection, cells (5 × 106) were suspended in buffer containing 2.5 μg plasmid DNA. After electroporation, they were seeded in 100 mm culture dishes in DMEM without antibiotics.
Twenty-four hours after transfection, COS7 cells were treated with 50 ng/mL of glial cell line-derived neurotrophic factor (GDNF) for 60 minutes, after serum starvation. They were lysed with lysis buffer and centrifuged. The pellet was discarded, and aliquots of the supernatant containing 60 μg protein were separated on 8% and 10% acrylamide gels under denaturing conditions. After transfer to a membrane, the membrane was treated with first antibody. Antibodies used were rabbit anti-phospho-RET (Y1062) antibody (R&D System, Minneapolis, MN, USA). Rabbit anti-RET (E1N8X), rabbit anti-phospho-Akt (Ser473), rabbit anti-Akt, rabbit anti-phospho-phosphadidyl-inositol-3-kinase (PI3K) p85 (Try458)/p55 (Tyr199), rabbit anti-PI3K p85 (19H8), rabbit anti-phospho-extracellular regulated kinase (Erk)1/2, rabbit anti-Erk1/2, rabbit Bad, rabbit anti-cyclin D1 (92G2), rabbit anti-phospho-glycogen synthase kinase 3β (GSK3β; Ser9), rabbit anti-GSK3β (27C10) antibodies were purchased from Cell Signaling Technologies (Danvers, MA, USA). Rabbit anti-beta actin antibody was purchased from Abcam (Cambridge, UK) and mouse anti-c-Myc (9E10) antibody from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The membrane was then incubated with goat-anti mouse immunoglobulin G-horseradish peroxidase (IgG-HRP) and goat-anti rabbit IgG-HRP (GenDEPOT, Barker, TX, USA). Signals were detected with Enhanced Chemiluminescence (ECL) Select (GE Healthcare Life Sciences, Uppsala, Sweden). Bands were identified using an Image Analyzer system (PXi4, Syngene, UK) and quantified with Multi Gauge V3.0 software (Fujifilm, Tokyo, Japan).
Transfected NIH3T3 cells were harvested, and aliquots of 2 × 104 cells were seeded in 100 mm culture dishes in DMEM (Gibco BRL) with 5% BCS (Gibco BRL). After 3 weeks, the cells were fixed in ice-cold methanol for 10 minutes; 0.5% crystal violet solution in 25% methanol was added; the dishes were then incubated for 5 minutes at room temperature, and carefully rinsed with D.W. until no further color came off in the rinse. They were allowed to dry overnight on a benchtop and the cells were photographed with a gel documentation system (Uvitec, Cambridge, UK).
Twenty-four hours after transfection of NIH3T3 cells, 4 × 105 cells/well were cultured in 6-well dishes until 90% confluent. The wells were then scratched with a SPLScar (SPL, Pocheon, Korea) and incubation continued. Selected areas were analyzed under a microscope after 0, 12, 18, 24, and 36 hours using reference marks as guides, and the areas of the wound gaps were measured using ImageJ (National Institutes of Health, Bethesda, MD, USA) software with an magnetic resonance imaging wound healing tool.
Chemotaxis assays were performed using Transwell chambers (Corning Costar, Lowell, MA, USA) with 8 μm pore size membranes. Transfected NIH3T3 cells in 100 μL serum-free DMEM supplemented with 0.1% bovine serum albumin were placed in the upper chamber, and 600 μL DMEM supplemented with 10% BCS was placed in the bottom chamber. After incubating for 2 hours at 37°C, the cells were fixed with methanol and stained with crystal violet. The filters were mounted on slides lower surface uppermost and cells that had migrated onto the reverse side of the filter were counted with the assistance of a slide scanner (3DHISTECH Ltd., Budapest, Hungary).
Each experiment was repeated three times, and the results are expressed as mean ± standard deviation. Statistical significance was tested by one-way analysis of variance (ANOVA) and set at p < 0.05. All analyses were performed using SPSS for Windows version 12.0 (SPSS Inc., Chicago, IL, USA).
The Asp631Tyr (D631Y) mutation in codon 631 of exon 11 of the RET gene was identified in all probands of the three Korean families. It was also identified in eight of the 16 family members of these three families (Fig. 1A–1C). It is a germline mutation (GAC to TAC) that substitutes tyrosine for aspartate (Fig. 1D).
The clinical characteristics of the 34 D631Y variant MEN2 patients belonging to the seven families are summarized in Table 1. PCC was found in 58.8% of the carriers of the D631Y mutation, 30.3% of whom had bilateral PCC, and MTC was found in 32.3% of the D631Y carriers (Table 2). The age of onset of both PCC and MTC was 22 years, and both appeared in the same patients [16]. In the D631Y variant MEN2 patients with PCC and MTC, the median age at the time the corresponding mutations were detected was higher than in the case of the C634R/W mutation (49.0:53.5 years vs. 33.5:27 years). The incidence of MTC in D631Y variant MEN2 was lower than in C634 variant MEN2 (32.3% vs. 90%) (Table 2). It is noteworthy that lymph node metastasis of MTC was found in the proband of the Case 2 family, and that PHPT has been observed in only one individual, in a report by Elston et al. [14].
The effects of transfection of the wild type and mutant RET expression vectors into COS7 cells on levels of RET signal transduction molecules (Fig. 2A) were investigated by Western blotting. The D631Y expression vector increased phosphorylation of RET, AKT, and PI3K kinases as did the C630Y, C634R, and C634W vectors. The D631Y vector also inhibited phosphorylation of the downstream signaling molecules Bad and Gsk3β and increased the expression of the downstream signaling molecules c-Myc and cyclin D1, as well as the phosphorylation of Erk1/2 (Fig. 3). Thus, like the C630Y, C634R, and C634W mutations, the RET D631Y mutation appears potentially capable of promoting tumor formation by affecting cell proliferation, differentiation, and migration. The effects on expression of signaling molecules resulting from transfection of RET harboring the C630Y, D631Y, C634R, and C634W mutations differed significantly from those resulting from transfection of the wild type RET, but the effects of the C630Y, D631Y, and C634R/W mutations were not significantly different from one another.
Wild type and mutant RET expression vectors were transfected into NIH3T3 cells, and focus formation, chemotaxis, and wound healing were examined. As in the case of the signaling molecule changes, expression of the D631Y variant RET had effects on focus formation, chemotaxis, and wound healing similar to those caused by expression of the C630Y, C634R, and C634W variants (Fig. 4). However, chemotaxis was significantly higher in response to expression of D631Y than in response to expression of C630Y and lower than in response to expression of C634R/W. Wound healing after 36 hours was similar in response to expression of C630Y and D631Y, but healing in response to expression of D631Y was significantly slower than in response to expression of C634R (Fig. 4C and 4D). On the basis of this test, it appears that the tumorigenic effect of the D631Y RET mutation is greater than that of the C630Y mutation and weaker than that of the C634R/W mutation.
The purpose of testing for mutations in the RET gene in MEN2 is to identify individuals needing treatment to prevent life-threatening disease, and to advise on the transmission/inheritance of the mutations [14,21]. Because the only treatment for MTC is surgery, the amino acid substitution in the mutant determines the optimum age for thyroidectomy [22]. We analyzed the clinical characteristics of MEN2 in seven families with the D631Y mutation: four families previously reported in the literature and three identified in this institute (Table 1). The D631Y germline mutation in MEN2 have also been reported in Germany [13,23], the Netherlands [24], and Turkey [25] but they were excluded from this analysis because results of thyroidectomy and family screening were not provided. We found that PCC occurred in 58.8% and MTC in 32.3% of the RET D631Y variant MEN2 patients, and PHPT in only one case (2.9%) (Table 2) [1,2,6,24,26–35]. This compares with previous reports that 90% of MEN2 patients developed MTC, 50% developed PCC, and 20% to 30% developed PHPT [1,5,6,15]. According to the literature, patients with D631Y variant MEN2 do not develop parathyroid tumors [16,18]. Therefore, whether the occurrence of PHPT in one patient in one family [14] was related to the D631Y mutation is unclear. PCC and MTC appeared to develop at a later age in D631Y mutant patients than in C634R/W patients, and the penetration of the D631Y mutation seemed to be significantly lower. In addition, the age of onset of MTC was significantly higher for the D631Y mutants than the C634R/W mutants (22 years vs. < 3 years). Therefore, it seems reasonable to consider deferring prophylactic thyroidectomy beyond the age of 5 in patients with the D631Y mutation. However more clinical information and laboratory data are needed to confirm this inference. To date, patients with the D631Y mutation have not been reported to suffer from lymph node metastases or distant metastatic disease [14]. However, it is notable that in case 2, lymph node metastasis of MTC was identified in two of 27 lymph nodes examined. We therefore recommend close screening and monitoring for lymph node metastasis of MTC in D631Y carriers.
Mutations in the RET gene associated with MEN2 occur most frequently in exons 10 and 11 (codons 630 and 634, respectively), which encode cysteine-rich extracellular domains [2,6,36]. RET receptor activation occurs when a GDNF family ligand (GFL) binds to the RET receptor [1,10,37]. When the GFL forms a complex with the GDNF family receptor (GFR) dimerization of the RET receptor occurs and autophosphorylation of specific tyrosine residues takes place (Fig. 2A) [6]. This eventually activates downstream signaling molecules along the RAS (rat sarcoma oncogene) and PI3K pathways, leading to tumor formation through stimulation of cell growth and survival, angiogenesis, migration and invasion [4,6,38,39]. To investigate the effects of expression of the various RET mutants (Fig. 2A), we constructed vectors for the C630Y and C634R/W mutations, which are reported to be the most common RET mutations with high risks of MTC in MEN2, as well as for the D631Y mutation, transfected them into COS7 cells and investigated the phosphorylation of RET signaling molecules mediated by activated RET. With the D631Y mutant vector, phosphorylation of RET signaling molecules was similar to that seen with the C630Y and C634R/W expression vectors. In other words, there was increased phosphorylation of RET, AKT, PI3K kinase, and Erk1/2 (Fig. 3). Focus formation after infecting NIH3T3 cells with the D631Y mutation was also similar to that seen with the C630Y and C634R/W mutations (Fig. 4A and 4B), and chemotaxis and wound healing resulting from expression of the D631Y variant was similar in response, pattern, and extent to that seem with C630Y and C634R/W, but stronger than with C630Y and weaker than with C634R/W (Fig. 4C and 4D). Our findings thus indicate that MEN2 patients with the D631Y mutation develop multiple endocrine tumors by a mechanism similar to that in patients with the C630Y and C634R/W mutations of MEN2.
The D631Y mutation has low tumorigenic potential, as it appears to present at a later age and behave in a more indolent manner [14]. Interestingly, the opposite was reported in an in vitro study in which the D631Y mutant was found to have high carcinogenic activity [19]. That was the only laboratory study performed so far; four vectors expressing Asp631Tyr/Gly/Asn/Ala were generated and transfected into NIH3T3 cell lines and their effects were observed, and high carcinogenic activity was observed only for the D631Y.
In our study, patients with RET D631Y variant MEN2 developed PCC and MTC later than patients with the C634R/W mutation, and the penetration of RET D631Y with respect to MTC was significantly lower. However, laboratory studies showed that the tumorigenic potential of the D631Y RET mutant vector was greater than that of C630Y mutant and weaker than that of C634R/W mutant. Thus, as also suggested by Elston et al. [14], D631Y mutant MEN2 led to later onset of PCC and MTC than C634R/W MEN2, and the earliest age of onset of MTC was 22 years. Therefore, it is reasonable to delay prophylactic thyroidectomy until 12 years of age in patients with D631Y mutation. In general, it is recommended that prophylactic thyroidectomy for MEN2 patients be performed 10 years before the earliest age of onset of MTC [14].
A limitation of this study is that we were only able to analyze the clinical phenotypes of seven families, some in Korea others abroad. This number of families was insufficient to allow us to analyze genetic penetrance and associated cancer risk, though the cancer risk appeared to be lower in the D631Y mutant than in the C634 codon variants. Caution should of course be exercised in predicting patients’ prognoses from the results of in vitro experiments using mutant RET expression vectors. In conclusion, we suggest that the tumorigenic potential conferred by the D631Y mutation is lower than that conferred by the C634R/W mutation, but higher than that conferred by C630Y. Thus, the risk level of the RET D631Y variant appears to be higher than that of C630Y and lower than that of C634R/W.
1. The D631Y mutation led to later onset of pheochromocytoma and medullary thyroid carcinoma than the C634R/W mutation.
2. The age of onset of medullary thyroid carcinoma (MTC) was significantly higher for D631Y mutants than for C634R/W mutants.
3. Close screening and monitoring for lymph node metastasis of MTC in D631Y carriers is likely to be necessary.
4. The risk level of the rearranged during transfection (RET) D631Y variant appears to be higher than that of the C630Y variant but lower than that of the C634R/W variant.
Acknowledgments
This study was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (Grant number NRF-2016R1A2B4008830).
Figure 1
Pedigrees and sequencing results of multiple endocrine neoplasia type 2 (MEN2) families with the D631Y mutation. (A) Pedigree of case 1, (B) pedigree of case 2, (C) pedigree of case 3, as summarized in Table 1. Males (squares); females (circles). Symbols filled in black indicate suffering from D631Y MEN2. Family members whose DNA was analyzed are indicated by superscript circles in the upper right corner. Arrows indicate probands. Family members are designated by generation (Roman numbers) and as individuals (Arabic numbers). (D) Sequencing results; in the proband and family members participating in the analysis, the sequence GAC encoding Asp in the wild type is changed to the sequence TAC encoding Tyr. LN, lymph node; Meta, metastasis; PCC, pheochromocytoma; MTC, medullary thyroid carcinoma.

Figure 2
Signaling pathways controlled by rearranged during transfection (RET), structure of RET expression plasmids and risk levels of RET codon changes. (A) Phosphorylation of RET tyrosine residues results in activation of several intracellular signaling pathways. Downstream targets include the extracellular regulated kinase (Erk) and phosphadidyl-inositol-3-kinase (PI3K)/Akt pathways. RET appears to play a central role in cell proliferation, differentiation, and migration via these pathways. (B) Schematic illustration of the RET expression plasmids. The entire coding regions of C630Y, D631Y, and C634R/W and wild type cDNAs were subcloned into the HindIII and KpnI sites of the pcDNA3 vector using a C-terminal flag tag. The positions of restriction enzyme sites, start (ATG) and stop (TGA) codons, and HindIII, KpnI, and XhoI sites are indicated. Exons are indicated by arabic numerals and the base changes (c.1889G>A [C630Y], c.1891G>T [D631Y], c.1900T>C [C634R], and c.1902C>G [C634W]) are listed. GSK3β, glycogen synthase kinase 3β; pCMV, promoter cytomegalovirus; SV40, simian virus 40. aStill lack of evidence-based data.
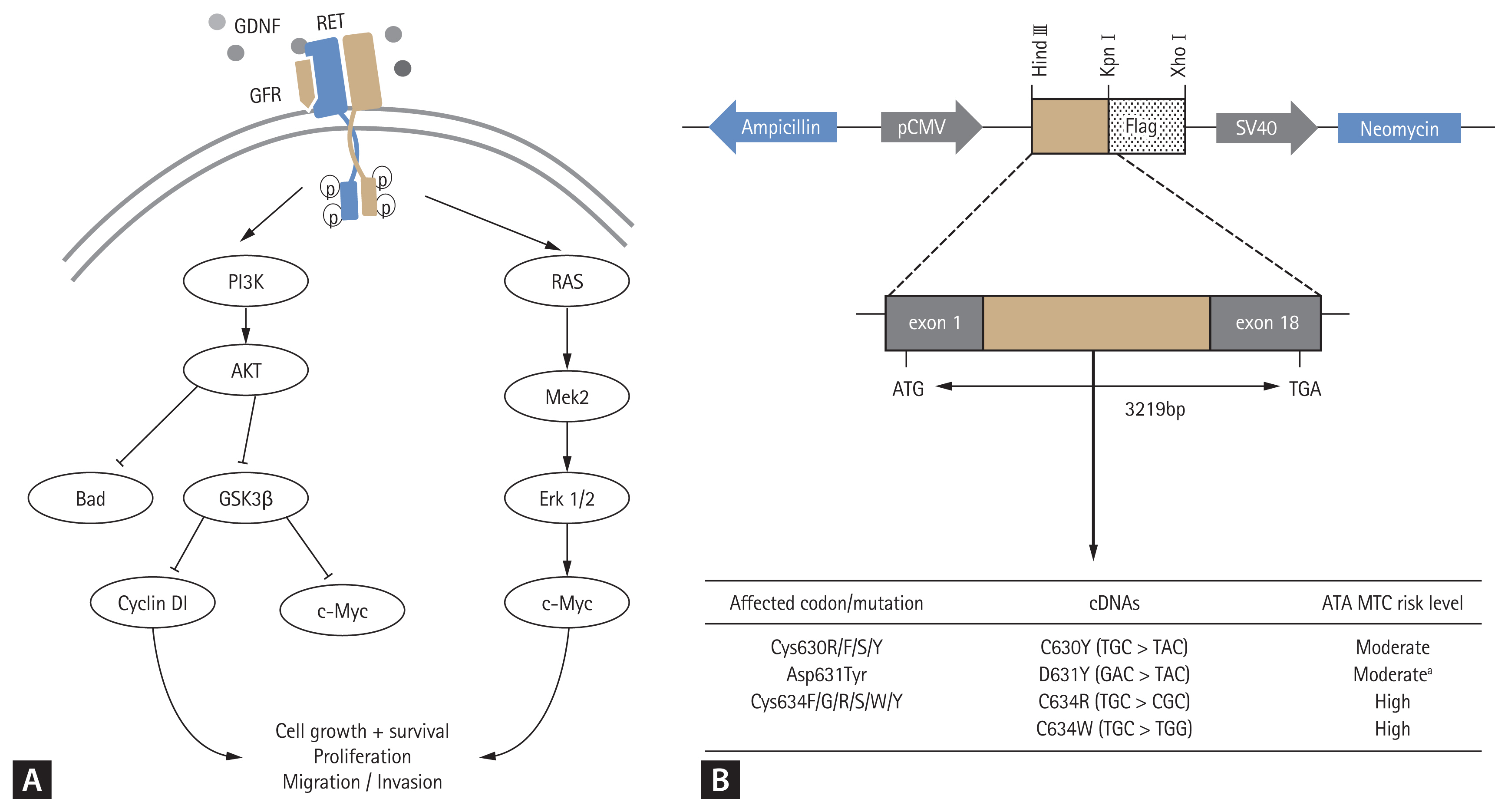
Figure 3
Western blot analysis of relevant rearranged during transfection (RET) downstream signaling molecules in COS7 cells expressing wild type and mutant RET vectors. Glial cell line-derived neurotrophic factor (GDNF)-treated COS7 cells were transfected with the four RET mutant vectors, wild type vector and empty vector, and phosphorylation of signaling intermediates was analyzed by Western blotting. All the mutations significantly increased phosphorylation of RET, phosphadidyl-inositol-3-kinase (PI3K), and AKT, and inhibited phosphorylation of Bad and glycogen synthase kinase 3β (GSK3β). As a result, increased expression of c-Myc and cyclin D1 was observed with all four vectors, along with increased phosphorylation of extracellular regulated kinase (Erk) 1/2. Data are expressed as mean ± standard deviation. p, phospho; t, total. ap < 0.05, bp < 0.01 vs. wild type.
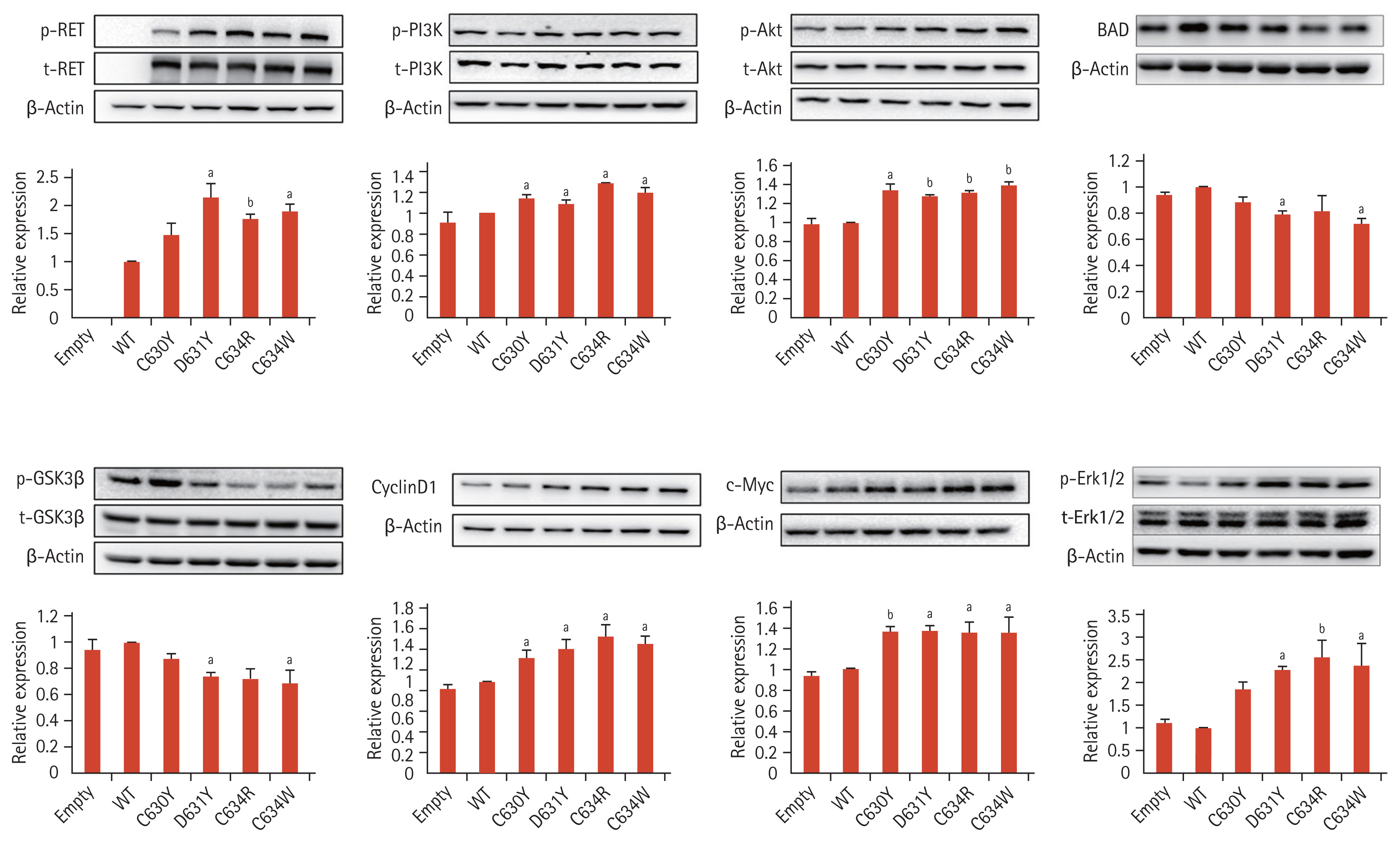
Figure 4
Focus formation, chemotaxis and wound healing by NIH3T3 cells transfected with wild type (WT) and mutant rearranged during transfection (RET) vectors. (A) Focus formation tests performed on crystal violet-stained plates 3 weeks after transfection, and summary graph showing numbers of colonies formed by the mutants and WT. (B) Results of chemotaxis assays using transwell chambers. Migrated cells were fixed with methanol, stained with crystal violet, and counted with a slide scanner. (C) Representative wound healing images observed in NIH3T3 cells transfected with RET WT and mutant vectors. (D) Summary of wound healing rates. Data are expressed as mean ± standard deviation. ap < 0.05, bp < 0.01, cp < 0.001 vs. wild type, dp < 0.05 vs. C630Y, ep < 0.05 vs. C634R, fp <0.05 vs. C634W.

Table 1
Clinical features of all patients in the seven families with the D631Y mutation
| Family | Age/Gender | PCC (age, yr) | MTC (age, yr) | PHPT | Reference | |
|---|---|---|---|---|---|---|
| 1 | 1a | 67/F | Bilat (46) | N | N | Elston et al. [14] |
| 2 | 66/M | Unilat (63) | CCH (60) | N | ||
| 3 | 41/M | Unilat (39) | Y (34) | Y | ||
| 4 | 37/F | N | N | N | ||
| 5 | 34/F | N | N | N | ||
| 2 | 6a | 46/F | Bilat (46) | Y (46) | N | Bae et al. [16] |
| 7 | 53/F | Bilat (53) | Y (53) | ? | ||
| 8 | 44/F | Unilat (44) | N | ? | ||
| 9 | 22/M | Unilat (22) | Possible (22) | ? | ||
| 10 | 30/F | Unilat (30) | Y (30) | ? | ||
| 11 | 75/M | N | N | N | ||
| 12 | 27/F | N | N | N | ||
| 3 | 13a | 54/F | Bilat (54) | Y (54) | N | |
| 14 | 53/F | Bilat (52) | N | N | ||
| 15 | 26/F | N | N | N | ||
| 16 | 20/M | N | N | N | ||
| 4 | 17 | 24/M | Unilat (24) | N | N | Ospina et al. [18] |
| 18 | 27/F | N | N | N | ||
| 19 | 54/M | Unilat (54) | N | N | ||
| 20 | 57/M | N | N | N | ||
| 21 | 53/F | N | N | N | ||
| 22 | 56/F | Unilat (56) | N | N | ||
| 23 | 24/M | N | N | N | ||
| 24 | 29/F | Unilat (29) | N | N | ||
| 25 | 26/F | N | N | N | ||
| 26a | 79/F | N | Y (79) | N | ||
| Case 1 | II-3a | 62/F | Bilat (62) | Y (62) | N |
Kim et al. [17] Fig. 1A |
| III-2 | 35/F | Ds | Ds | Ds | ||
| Case 2 | II-1a | 66/F | Bilat (66) | Y (66)b | N | Fig. 1B |
| III-1 | 43/M | N | Y (43) | N | ||
| Case 3 | II-7a | 64/F | Bilat (64) | N | N | Fig. 1C |
| III-4 | 40/M | Bilat (40) | N | N | ||
| II-3 | 66/M | Bilat (66) | N | N | ||
| III-1 | 42/M | Bilat (42) | N | N |
Table 2
Comparison of clinical characteristics according to RET variant type
| D631Y | C630Y | C634R/W | |
|---|---|---|---|
| ATA risk level (moderate/high/highest) | Moderate | moderate | high |
| Cases of MEN2, % | ? | 4–10 [6,24] | 85 [6] |
| Age at diagnosis, yr, median (range) | |||
| PCC | 49 (22–66) | ? | 33.5 (15–77) [2,27–32] |
| MTC | 53.5 (22–79) | 67 (11–71) [26] | 27 (<3–77) [1,2,28,30,32–34] |
| Penetrance | |||
| PCC | 58.8 (50% [1]) | 10–30 [2] | 25–88 [1,2] |
| bilateral PCC | 30.3 | ? | 38.8 [35] |
| MTC | 32.3 | ? | 90 [1] |
| PHPT | 2.9 | 10 [2] | 20–30 [1] |
REFERENCES
1. Melmed S, Auchus RJ, Goldfine AB, Koenig RJ, Rosen CJ, Williams RH. Williams Textbook of Endocrinology. 14 ed. Philadelphia (PA): Elsevier, 2020.
2. Wells SA Jr, Asa SL, Dralle H, et al. Revised American Thyroid Association guidelines for the management of medullary thyroid carcinoma. Thyroid 2015;25:567–610.



3. Brandi ML, Gagel RF, Angeli A, et al. Guidelines for diagnosis and therapy of MEN type 1 and type 2. J Clin Endocrinol Metab 2001;86:5658–5671.

4. Prazeres H, Torres J, Rodrigues F, et al. How to treat a signal? Current basis for RET-genotype-oriented choice of kinase inhibitors for the treatment of medullary thyroid cancer. J Thyroid Res 2011;2011:678357.



5. Waguespack SG, Rich TA, Perrier ND, Jimenez C, Cote GJ. Management of medullary thyroid carcinoma and MEN2 syndromes in childhood. Nat Rev Endocrinol 2011;7:596–607.


6. Jameson JL, De Groot LJ. Endocrinology: Adult and Pediatric. 2:7th ed. Philadelphia (PA): Elsevier Saunders, 2016.
7. Machens A, Lorenz K, Sekulla C, et al. Molecular epidemiology of multiple endocrine neoplasia 2: implications for RET screening in the new millenium. Eur J Endocrinol 2013;168:307–314.


10. de Groot JW, Links TP, Plukker JT, Lips CJ, Hofstra RM. RET as a diagnostic and therapeutic target in sporadic and hereditary endocrine tumors. Endocr Rev 2006;27:535–560.


11. Voss RK, Feng L, Lee JE, et al. Medullary thyroid carcinoma in MEN2A: ATA moderate- or high-risk RET mutations do not predict disease aggressiveness. J Clin Endocrinol Metab 2017;102:2807–2813.




12. American Thyroid Association Guidelines Task Force; Kloos RT, et al.; Eng C. Medullary thyroid cancer: management guidelines of the American Thyroid Association. Thyroid 2009;19:565–612.


13. Berndt I, Reuter M, Saller B, et al. A new hot spot for mutations in the ret protooncogene causing familial medullary thyroid carcinoma and multiple endocrine neoplasia type 2A. J Clin Endocrinol Metab 1998;83:770–774.


14. Elston MS, Meyer-Rochow GY, Holdaway I, Conaglen JV. Patients with RET D631Y mutations most commonly present with pheochromocytoma and not medullary thyroid carcinoma. Horm Metab Res 2012;44:339–342.


15. Eng C. Multiple endocrine neoplasia type 2. In: Adam MP, Ardinger HH, Pagon RA, eds, et al. GeneReviews Seattle (WA): University of Washington, Seattle, 1999. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1257
.

16. Bae SJ, Kim DJ, Kim JY, et al. A rare extracellular D631Y germline mutation of the RET proto-oncogene in two Korean families with multiple endocrine neoplasia 2A. Thyroid 2006;16:609–614.


17. Kim JH, Jeon EK, Kim KH, et al. Two cases of RET proto-oncogene mutation in multiple endocrine neoplasia type 2a. Korean J Med 2007;73:994–1002.
18. Ospina NS, Maraka S, Donegan D, Morris JC. Clinical features of a family with multiple endocrine neoplasia type 2A caused by the D631Y RET mutation. Thyroid 2017;27:1332–1334.


19. Asai N, Iwashita T, Murakami H, et al. Mechanism of Ret activation by a mutation at aspartic acid 631 identified in sporadic pheochromocytoma. Biochem Biophys Res Commun 1999;255:587–590.


20. Asai N, Iwashita T, Matsuyama M, Takahashi M. Mechanism of activation of the ret proto-oncogene by multiple endocrine neoplasia 2A mutations. Mol Cell Biol 1995;15:1613–1619.



22. Lips CJ. Clinical management of the multiple endocrine neoplasia syndromes: results of a computerized opinion poll at the Sixth International Workshop on Multiple Endocrine Neoplasia and von Hippel-Lindau disease. J Intern Med 1998;243:589–594.


23. Machens A, Dralle H. Familial prevalence and age of RET germline mutations: implications for screening. Clin Endocrinol (Oxf) 2008;69:81–87.


24. Mathiesen JS, Kroustrup JP, Vestergaard P, et al. Distribution of RET mutations in multiple endocrine neoplasia 2 in Denmark 1994–2014: a nationwide study. Thyroid 2017;27:215–223.



25. Aldemir O, Ozsan M, Gokce H, et al. A family diagnosed as MEN2A with a rare mutation D631Y in RET oncogene. West Indian Med J 2016;https://doi.org/10.7727/wimj.2016.231.

26. Yonekawa H, Sugitani I, Fujimoto Y, Arai M, Yamamoto N. A family of multiple endocrine neoplasia type 2A (MEN 2A) with Cys630Tyr RET germline mutation: report of a case. Endocr J 2007;54:531–535.


27. Quayle FJ, Fialkowski EA, Benveniste R, Moley JF. Pheochromocytoma penetrance varies by RET mutation in MEN 2A. Surgery 2007;142:800–805.


28. Zhou Y, Zhao Y, Cui B, et al. RET proto-oncogene mutations are restricted to codons 634 and 918 in mainland Chinese families with MEN2A and MEN2B. Clin Endocrinol (Oxf) 2007;67:570–576.


29. Jindrichova S, Vcelak J, Vlcek P, Neradilova M, Nemec J, Bendlova B. Screening of six risk exons of the RET proto-oncogene in families with medullary thyroid carcinoma in the Czech Republic. J Endocrinol 2004;183:257–265.


30. Chung YJ, Kim HH, Kim HJ, et al. RET proto-oncogene mutations are restricted to codon 634 and 618 in Korean families with multiple endocrine neoplasia 2A. Thyroid 2004;14:813–818.


31. Punales MK, Graf H, Gross JL, Maia AL. RET codon 634 mutations in multiple endocrine neoplasia type 2: variable clinical features and clinical outcome. J Clin Endocrinol Metab 2003;88:2644–2649.


32. Milos IN, Frank-Raue K, Wohllk N, et al. Age-related neoplastic risk profiles and penetrance estimations in multiple endocrine neoplasia type 2A caused by germ line RET Cys634Trp (TGC>TGG) mutation. Endocr Relat Cancer 2008;15:1035–1041.


33. Qi XP, Zhao JQ, Fang XD, et al. Spectrum of germline RET variants identified by targeted sequencing and associated multiple endocrine neoplasia type 2 susceptibility in China. BMC Cancer 2021;21:369.



34. Machens A. Early malignant progression of hereditary medullary thyroid cancer. N Engl J Med 2004;350:943.

35. Lang BH, Yu HW, Lo CY, et al. Bilateral pheochromocytomas in MEN2A syndrome: a two-institution experience. World J Surg 2015;39:2484–2491.


36. Colombo C, Minna E, Rizzetti MG, et al. The modifier role of RET-G691S polymorphism in hereditary medullary thyroid carcinoma: functional characterization and expression/penetrance studies. Orphanet J Rare Dis 2015;10:25.



-
METRICS

- Related articles
-
Clinical features of snoring patients during sedative endoscopy2019 March;34(2)




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


