 |
 |
|
|
|
Abstract
Background/Aims
Relatively little data are available on how the response to the coronavirus disease 2019 (COVID-19) pandemic has affected treatment outcomes in patients receiving chemotherapy for lymphoma or multiple myeloma. We aimed to determine the effect of COVID-19 countermeasures on treatment outcomes in this patient population.
Methods
We retrospectively analyzed data on patients treated for lymphoma or multiple myeloma in two tertiary hospitals in Seoul. Patients were divided into two groups: group 1 included patients who received chemotherapy between September and December 2019 (the control period), and group 2 included patients who received chemotherapy between September and December 2020 (the study period). Countermeasures to COVID-19 were applied to the patients in group 2. The countermeasures implemented included mask wearing and regular handwashing at home and in hospital; COVID-19 risk assessments on all hospital visitors; and pre-emptive COVID-19 screening for all newly hospitalized patients and their resident guardians.
Results
No differences in treatment outcomes, including treatment response, incidence and duration of neutropenia or neutropenic fever, delays in chemotherapy, or number of deaths during chemotherapy, were observed between the g roups. None of the patients in group 2 tested positive for COVID-19, and there were no COVID-19-related deaths during the study period.
Conclusions
Countermeasures to COVID-19 did not affect treatment outcomes in patients receiving chemotherapy for lymphoma or multiple myeloma. Data on the effect of countermeasures to COVID-19 on treatment outcomes should continue to be analyzed to ensure that treatment outcomes are not adversely affected.
Coronavirus disease 2019 (COVID-19) is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a positive-sense single-stranded RNA virus transmitted via respiratory droplets (direct contact) and contaminated objects and airborne contagion (indirect contact) [1,2]. Although the mortality rate of COVID-19, at approximately 3%, is lower than that of SARS-CoV-1, at approximately 10%, or Middle East respiratory syndrome, at approximately 40%, it is far more contagious, infecting approximately 10 times more people [3]. As the number of patients with COVID-19 increases, the strain on medical resources locally and globally is reaching a level unprecedented in recent times. As various efforts to contain the spread of COVID-19, to produce vaccines against it, and to develop therapeutic agents to treat it are underway, the pressure on medical facilities remains challenging as the need to optimally treat patients with COVID-19 is added to the need to optimally treat patients with other diseases, including malignancies.
Patients with malignancies are more likely to be infected by SARS-CoV-2 and are more likely to develop severe COVID-19 [4,5]. The mortality rate among patients with existing malignancies and COVID-19 can reach approximately 33% [6–10]. The risks of more severe disease and death tend to be higher in patients with hematologic malignancies [11,12], with the mortality rate reported to be as high as 62% [13,14]. Despite the challenges of COVID-19, chemotherapy for patients with hematologic malignancies cannot be delayed indefinitely. Countermeasures to prevent infection with SARS-CoV-2 are thus critical to ensure optimal treatment for both patients with COVID-19 and those with hematologic malignancies receiving chemotherapy.
This study aimed to analyze an example of countermeasures to COVID-19 and to evaluate the impact of these countermeasures on treatment processes and outcomes in patients who were hospitalized repeatedly and/or received outpatient treatment for lymphoma or multiple myeloma.
The study was approved by the appropriate Institutional Review Board (IRB) of Korea University Anam Hospital and Korea University Guro Hospital, and all data were fully anonymized (IRB No. 2020AN0559 and 2021GR0051). As this study was conducted using anonymous patient data, the requirement for informed consent was waived by the IRB.
In this non-interventional comparative cohort study, we retrospectively analyzed the data of patients who were consecutively enrolled in the Lymphoma and Myeloma Registry from September 2019 to December 2019 and from September 2020 to December 2020. Data from two tertiary hospitals affiliated with the Korea University Medical Center (Anam and Guro Hospitals) located in Seoul were used. The Anam and Guro Hospitals had a total of 1,048 and 1,075 beds and received approximately 3,665 and 3,985 outpatients daily, respectively, as of January 2021. Patients who met the following criteria were included: (1) a diagnosis of lymphoma or multiple myeloma according to the World Health Organization classification, and (2) provision of chemotherapy from September 2019 to December 2019 (the control period) or September 2020 to December 2020 (the study period).
Patients were divided into two groups: group 1 included patients with lymphoma or multiple myeloma who received chemotherapy during the control period, and group 2 included patients with lymphoma or multiple myeloma who received chemotherapy during the study period. Countermeasures to COVID-19 were applied to group 2. Group 1 was included as a control group to account for seasonal viral infectious diseases, such as influenza, that may have occurred in patients treated at a similar time before the COVID-19 pandemic. Both groups were defined as patients with lymphoma or multiple myeloma who received chemotherapy within a limited period, and the clinical outcomes measured within the period were compared.
The South Korean government initiated active quarantine to control the COVID-19 pandemic in February 2020. With the continuation of the COVID-19 pandemic, the two tertiary hospitals in this study also initiated active responses to the COVID-19 pandemic that applied to patients, their guardians, and hospital workers from September 2020. The countermeasures that were implemented and included this study are listed in Table 1.
Patients and their guardians were required to wear a mask, irrespective of whether they were indoors or outdoors at home or in a hospital, and to practice hand hygiene by washing their hands thoroughly with soap under running water. All patients and guardians who visited the hospital were permitted to enter only after confirming that they had no history of visiting any areas with COVID-19 outbreaks or foreign countries, no COVID-19-related symptoms, and no fever (temperature > 37.5°C). If one or more of the above conditions were not satisfied, they were not permitted to enter the hospital and were referred to an outdoor COVID-19 clinic or emergency department. All newly admitted patients and their resident guardians were confirmed to be negative for COVID-19 1 to 7 days before hospitalization. Hospitalized patients were prohibited from going out and were to avoid the use of lounges or restaurants in the hospital. When fever and respiratory symptoms occurred in patients or hospital workers, active consultation with the Department of Infectious Diseases to arrange for COVID-19 testing was undertaken. All COVID-19 tests were conducted at the outdoor COVID-19 clinic or emergency department, thoroughly separated from the existing laboratory. If emergency hospitalization was required, dedicated quarantine beds were used until the COVID-19 test result was confirmed. During this period, COVID-19 testing was conducted using a real-time PCR assay (Allplex™ 2019-nCoV Assay, Seegene, Seoul, Korea; or STANDARD M nCoV Real-Time Detection kit, SD biosensor, Suwon, Korea). All test results were recorded within 12 hours. Those who tested positive for COVID-19 were transferred to nationally designated COVID-19 hospitals or residential treatment centers.
The primary endpoints were treatment outcomes under the countermeasures implemented during the COVID-19 pandemic. Treatment outcomes were evaluated as the result of the treatment response evaluation within the corresponding period for each group. If the treatment response evaluation was performed more than twice within the corresponding period for the group, it was summarized based on the best response. The treatment response evaluation for lymphoma and multiple myeloma was assessed according to the Lugano classification [15] and International Myeloma Working Group response criteria [16,17], respectively.
The secondary endpoints were the incidence and duration of neutropenia and neutropenic fever, delays in chemotherapy, mortality during chemotherapy, and the rate of positive COVID-19 results among the tested patients. To evaluate any adverse events, all events that occurred within the corresponding period in each group were counted. Neutropenia was defined as the absolute neutrophil counts (ANCs) < 1,000/μL after chemotherapy. Neutropenic fever and its duration was defined as the time period during which the following conditions were met: (1) development of fever after chemotherapy; (2) an ANC < 500/μL or an ANC < 1,000/μL with a predicted decline to ≤ 500/μL within the next 48 hours; and (3) a temperature of > 38.3°C once, or > 38.0°C sustained over a 1 hour period [18]. A delay in chemotherapy was defined as a delay of ≥ 3 days compared with the scheduled chemotherapy date.
Mean values and standard deviations were reported for continuous variables, and percentages were reported for categorical values. Baseline characteristics were compared between the groups using the Mann–Whitney U test or chi-square test, according to the type of variable. Equivalence was concluded if the two-sided 90% confidence interval (CI) for the difference in clinical endpoints between the two groups was within the equivalence margin of ± 15% (two-one-sided t tests). In the existing equivalence comparison studies, the range of the equivalence margin was 10% to 20%, and a median value of 15% was determined in this study [19–22]. The median number of neutropenia or neutropenic fever episodes and their median durations were compared using the Mann–Whitney U test. IBM SPSS version 21.0 (IBM Corp., Armonk, NY, USA) was used for data analysis. A p value < 0.05 was set as significant.
Based on the national statistical data for 2019, the total area of South Korea is 100,401 km2, with a total population of 51,709,000 inhabitants. The total area of Seoul is 605 km2, accounting for only 0.6% of South Korea. However, 18.7% of the total population resides in Seoul, and the population density per km2 is 15,964 inhabitants, which is considerably higher than that in other regions and countries (Supplementary Fig. 1) [23–25]. As of January 1, 2021, the cumulative number of confirmed COVID-19 cases in South Korea was 61,769, of which 19,363 were reported in Seoul, accounting for 31.3% of the total number of confirmed cases (Supplementary Fig. 2) [26–28]. The number of newly reported COVID-19 cases and number of deaths from COVID-19 during the study period in Seoul are summarized in Supplementary Fig. 3 [26,27].
In total, 202 patients with lymphoma and 174 patients with multiple myeloma were analyzed in this study (lymphoma, group 1 [n = 97], group 2 [n = 105]; multiple myeloma, group 1 [n = 91], group 2 [n = 83]). Baseline characteristics are summarized in Tables 2 and 3. There was no difference between the groups in the median number of inpatients and outpatients per month during each period (group 1, 158 patients [range, 156 to 161], group 2, 166.5 [range, 148 to171], p = 0.343; and group 1, 1,049 patients [range, 1,035 to 1,098], group 2, 1,176.5 patients [range, 1,045 to 1,324], p = 0.114, respectively).
In patients diagnosed with lymphoma, there was no difference between the two groups in terms of age, sex, lymphoma subtype, disease stage, extranodal involvement, lactate dehydrogenase levels, Eastern Cooperative Oncology Group performance status score, International Prognostic Index score, and the proportion of patients receiving rituximab during chemotherapy. However, the rate of bone marrow involvement was higher in group 2 than in group 1 (group 1, 16/97 patients, 16.5%; group 2, 30/105 patients, 28.6%; p = 0.041).
In patients with multiple myeloma, the proportion of patients receiving pomalidomide during chemotherapy was higher in group 2 than in group 1 (group 1, 4/91 patients, 4.4%; group 2, 11/83 patients, 13.3%; p = 0.038). However, there was no difference between the two groups in terms of age, sex, type of multiple myeloma, International Stage System values, and the proportion of patients receiving bortezomib, carfilzomib, thalidomide, or lenalidomide during chemotherapy.
Among patients with lymphoma, 91 patients in group 1 (83.5%) and 91 patients in group 2 (86.7%) underwent response evaluation within the corresponding periods, and 54 patients in group 1 (55.7%) and 59 patients in group 2 (56.2%) achieved complete remission (Table 4). The 90% CIs for the estimate of the difference were within the equivalence margin (± 15%). In addition, the incidence of neutropenia (group 1, 62/97 patients, 63.9%; group 2, 68/105 patients, 64.8%), incidence of delays in chemotherapy (group 1, 8/97 patients, 8.2%; group 2, 8/105 patients, 7.6%), and number of deaths during chemotherapy (group 1, 9/97 patients, 9.3%; group 2, 4/105 patients, 3.8%) were similar between the two groups. Although the equivalence of the incidence of neutropenic fever was inconclusive, there was no difference between the two groups in the duration of neutropenia or neutropenic fever during chemotherapy (group 1, 2.4 [range, 1.0 to 23.0] vs. group 2, 2.4 [range, 1.0 to 45.0], p = 0.803; group 1, 4.0 [range, 1.0 to 15.0] vs. group 2, 4.3 [range, 2.0 to 11.0], p = 0.585, respectively). In group 1, the causes of delayed chemotherapy were as follows: infection (four cases: catheter-related bloodstream infection [CRBSI], intra-abdominal infection, candidemia, and neutropenic fever), cytopenia (two cases), and deterioration of the general condition (two cases). In group 2, the causes of delayed chemotherapy were as follows: infection (six cases: three cases of neutropenic fever, one case each of bacteremia, herpes zoster, and fungal arthritis), drug rash caused by chemotherapy (one case), and deterioration of the general condition (one case). In group 1, the causes of death were as follows: infection (five cases: two cases of neutropenic fever, two cases of pneumonia, and one case of bacteremia), bleeding at the cancer site (one case), tumor lysis syndrome (one case), and progression of lymphoma (two cases). In group 2, the cause of death was infection (four cases: two cases of pneumonia, and one case each of CRBSI and neutropenic fever).
In patients with multiple myeloma, 88 patients in group 1 (96.7%) and 82 patients in group 2 (98.8%) underwent response evaluation within the corresponding period, and 47 patients in group 1 (51.6%) and 43 patients in group 2 (42.9%) achieved complete remission or very good partial response (Table 5). The 90% CIs for the estimate of the difference were within the equivalence margin (± 15%). In addition, the incidence of neutropenia (group 1, 32/91 patients, 35.2%; group 2, 31/83 patients, 37.3%), incidence of neutropenic fever (group 1, 9/91 patients, 9.9%; group 2, 10/83 patients, 12.0%), incidence of delays in chemotherapy (group 1, 9/91 patients, 9.9%; group 2, 12/83 patients, 14.5%), and number of deaths during chemotherapy (group 1, 4/91 patients, 4.4%; group 2, 4/83 patients, 4.8%) were similar between the two groups. There were no differences between the two groups in the incidence and duration of neutropenia or neutropenic fever during chemotherapy (group 1, 1.3 [range, 1.0 to 29.0] vs. group 2, 1.3 [range, 1.0 to 22.0], p = 0.931; group 1, 2.0 [range, 1.0 to 3.0] vs. group 2, 2.8 [range, 1.0 to 5.5], p = 0.497, respectively). In group 1, the causes of delayed chemotherapy were as follows: infection (four cases: two cases of pneumonia, one case of urinary tract infection [UTI], and one case of infection of unknown origin) and deterioration of the general condition (five cases). In group 2, the causes of delayed chemotherapy were as follows: infection (five cases: three cases of pneumonia, one case of UTI, and one case of invasive aspergillosis) and deterioration of the general condition (four cases). In group 1, the causes of death were as follows: progression of multiple myeloma (two cases), intra-abdominal infection (one case), and underlying lung disease (one case). In group 2, the causes of death were progression of multiple myeloma (two cases) and infection (two cases: one case of UTI and one case of pneumonia).
There were no positive results for COVID-19 tests conducted according to the countermeasures implemented during the study period.
The population density in Seoul, South Korea, was 15,964 per km2, and the cumulative number of confirmed cases and deaths related to COVID-19 were 19,363 and 182, respectively, as of January 1, 2021. Although the total number of confirmed COVID-19 cases was relatively small compared with those of other countries, this study was conducted in a situation where the risk of transmission of COVID-19 was expected to be high due to the high population density. In this study, no difference in treatment response, incidence and duration of neutropenia or neutropenic fever, delays in chemotherapy, and deaths during chemotherapy were observed between the control (group 1) and study groups (group 2) of patients with lymphoma or multiple myeloma who were hospitalized repeatedly and received outpatient treatment (Table 1 [26], Fig. 1). In addition, during the study period, none of the patients tested positive for COVID-19, and there were no deaths from COVID-19.
Direct infection via expelled droplets and indirect infection via contact with virus-contaminated surfaces or objects are considered the main routes of transmission of SARS-CoV-2 [1,29]. Droplet transmission occurs when virus-laden droplets from an infected person are expelled during sneezing, coughing, or talking [30]. Exposure to an infected person at a distance of < 2 m for > 15 minutes increases the risk of droplet transmission, and in the case of symptomatic patients, droplet transmission can occur in a shorter time period [31]. In addition, expelled droplets can contaminate surrounding surfaces or objects. When these contaminated surfaces or objects are touched by another person who subsequently touches their eyes, nose, or mouth with their hands before washing them, SARS-CoV-2 infection can develop. The simplest ways to prevent droplet transmission of SARS-CoV-2 are to avoid visiting crowded places, to maintain social distancing, and to wear a mask covering the nose and mouth. In addition, to prevent indirect infection via contact with a virus-contaminated surface or object, it is recommended to refrain from touching your eyes, nose, or mouth with your hands and to wash your hands with soap under running water for at least 20 seconds [32].
The question remains, how should hospitals respond to situations such as the COVID-19 pandemic? For tertiary hospitals, guidance is needed to maintain the safety of patients already receiving treatment, along with appropriate responses to patients with COVID-19. In addition, countermeasures to minimize the depletion of the hospital workforce by ensuring safety are needed. Wearing a mask and washing hands are the most basic requirements for all patients, guardians, and hospital workers in hospitals. Based on data from the European Centers for Disease Control and Prevention, wearing a mask and handwashing resulted in significantly lower mortality and infection rates than did social distancing and handwashing [33]. Following countermeasures implemented in hospitals in Taiwan, establishing quarantine stations at the hospital entrance, screening for COVID-19-related risk factors and the presence of fever in all hospital visitors, and conducting training on wearing masks and handwashing may also be effective countermeasures to prevent hospital-acquired SARS-CoV-2 infection [34,35]. In this study, in addition to the standard countermeasures mentioned above, COVID-19 testing was performed in all patients requiring hospitalization and in their resident guardians, and hospitalization was only permitted once a negative result was obtained. Once admitted, leaving the premises was prohibited, and access to lounges or restaurants in the hospital was restricted. With the implementation of these countermeasures, there was no difference in treatment outcomes between patients with lymphoma or multiple myeloma who received chemotherapy before the COVID-19 pandemic and those who received chemotherapy during the COVID-19 pandemic. In addition, none of the patients tested positive for COVID-19, and there were no deaths from COVID-19 during the study period. To the best of our knowledge, there have been no studies on the treatment outcomes of patients with lymphoma or multiple myeloma who received chemotherapy in the social and medical context of the COVID-19 pandemic. This study may also be meaningful in that it presents countermeasures in an objective manner and offers learning that may be applied clinically should a similar situation occur post-COVID-19.
This study has some limitations. First, this was a retrospective study with a relatively small number of patients; thus, there were limitations intrinsic to the study design. Second, the analysis was performed using data from two tertiary hospitals. Additional studies from a wider variety of institutions and circumstances are needed to determine the appropriateness and effect of the countermeasures presented in this study. Third, the strategy presented in this study was limited in that COVID-19 testing was only routinely conducted in patients who required hospitalization and their resident guardians; however, outpatients, short-term visitors, and hospital workers were selectively tested according to their risk of exposure to COVID-19. In 50% to 80% of patients with COVID-19, there are no symptoms, and the transmission of COVID-19 can occur before symptoms develop [36–38], or even in the absence of symptoms [39–42]. If the population density, disease prevalence, or transmission rate is higher than that in this study, the countermeasures for asymptomatic patients should be modified. Finally, no positive COVID-19 results or deaths were reported among the enrolled patients with lymphoma or multiple myeloma in this study. This may be because the number of newly diagnosed patients with COVID-19 in Seoul was maintained at a level of < 500 per day, most likely due to the active COVID-19 quarantine measures implemented by the South Korean government. It may also be due to the countermeasures implemented by the hospital. However, conclusions on the effectiveness of the countermeasures that were implemented cannot be drawn from this study alone, and more studies in a variety of contexts are required to fully evaluate their efficacy and impact on treatment outcomes for patients with diseases other than COVID-19, particularly malignancies.
In conclusion, patients with lymphoma or multiple myeloma could receive chemotherapy during the COVID-19 pandemic without an adverse effect on treatment outcomes, and without infection-related adverse events, when appropriate countermeasures to COVID-19 were implemented. However, further studies are needed to determine the extent to which countermeasures can be applied, considering variations in population density, the prevalence and transmission rate of infectious diseases, and the characteristics of each institution. In addition, data on countermeasures to COVID-19 and the effects of these countermeasures should continue to be analyzed and shared to maintain optimal treatment outcomes for patients with existing diseases other than COVID-19 in the context of a shifting pandemic.
1. Patients with lymphoma or multiple myeloma could receive chemotherapy during the coronavirus disease 2019 (COVID-19) pandemic without an adverse effect on treatment outcomes, and without infection-related adverse events, when appropriate countermeasures to COVID-19 were implemented.
2. Data on countermeasures to COVID-19 and the effects of these countermeasures should continue to be analyzed and shared to maintain optimal treatment outcomes for patients with existing diseases other than COVID-19 in the context of a shifting pandemic.
Conflict of Interest
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Acknowledgments
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI) and Korea Infectious Diseases Clinical Trials Initiative, funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HE21C0001).
Figure 1
Countermeasures to the coronavirus disease 2019 (COVID-19) pandemic and the results of this study.
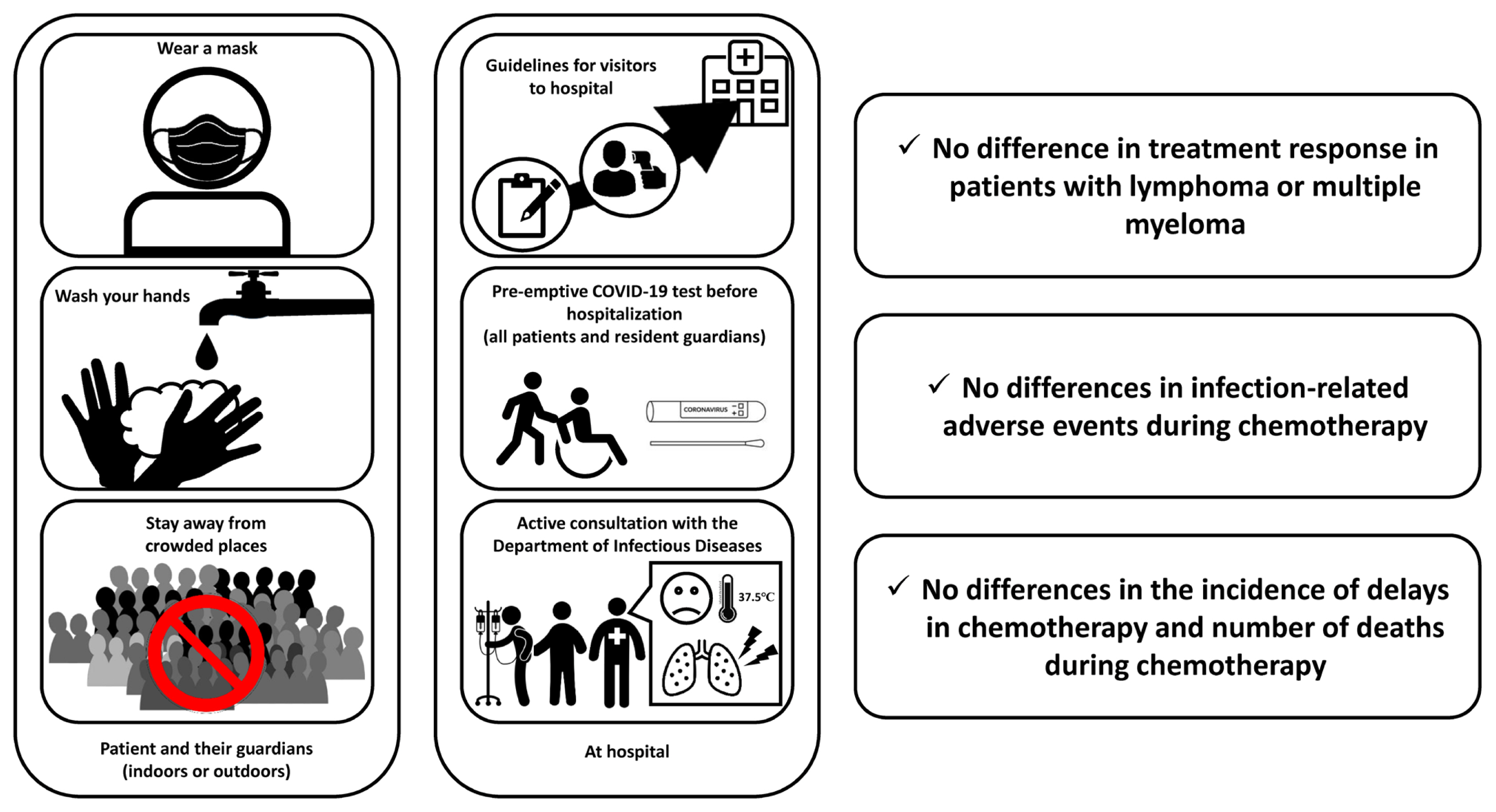
Table 1
Countermeasures to COVID-19 included in this study
| Patients and their guardians |
General principles Patients and their guardians should wear a mask, indoors or outdoors (at home or in a hospital), and should not touch their eyes, nose, or mouth with unwashed hands. In addition, patients should practice hand hygiene by washing their hands thoroughly with soap under running water. Patients and their guardians should refrain from visiting crowded places and going outside and avoid contact with people who have fever or respiratory symptoms. |
| Hospital |
For patients or visiting guardians All patients and visiting guardians are allowed to enter the hospital only after completing a questionnaire detailing any recent travel to foreign countries or areas where outbreaks of COVID-19 have occurred, as well as any COVID-19-related symptoms, and after confirming the absence of fever (temperature above 37.5°C). All patients newly admitted via emergency rooms, clinics, or other hospitals must be confirmed to be negative for COVID-19 1–7 days before their hospitalization. Resident guardians must also be confirmed to be negative for COVID-19 1–7 days before the patient’s hospitalization. Patients are prohibited from leaving the hospital during their hospitalization. Patients and their guardians should avoid unnecessary movement during hospitalization and should refrain from using lounges and restaurants in the hospital. Even if the COVID-19 test was negative before hospitalization, should patients or their guardians develop fever or respiratory symptoms during their hospitalization, the Department of Infectious Diseases should be consulted regarding the need for additional COVID-19 tests. For hospital workers All hospital workers are required to follow quarantine regulations, such as wearing a mask and practicing hand hygiene at the hospital. When COVID-19-related symptoms occur, hospital workers must cease working and undergo prompt diagnostic testing. Unnecessary events or meetings inside and outside the institution are restricted, and if unavoidable, such events or meetings must be confirmed and approved by the department in charge of COVID-19 management. At least the following conditions must be met: (1) attendee list management; (2) sufficient space between seats to ensure physical distancing between all participants; (3) no meals in conference rooms; and (4) wearing of a mask while attending. For those who test positive for COVID-19 Those who test positive for COVID-19 are transferred to nationally designated COVID-19 hospitals or residential treatment centers. |
| Government [26]a |
Infection prevention and control of foreign nationals Asymptomatic South Korean and foreign nationals on long-term visas are subject to self-quarantine of 14 days (Self-Quarantine Safety Protection App to be installed) and testing at a public health center within 3 days of arrival. Asymptomatic foreign nationals on short-term visas are subject to facility quarantine of 14 days (Self-Diagnosis App to be installed) and testing at a public health clinic within 14 days. If travelers exhibit fever or respiratory symptoms at entry screening, they are tested for COVID-19. South Korean or foreign nationals who test negative are placed under self-quarantine of 14 days (Self-Quarantine Safety Protection App to be installed) or are quarantined at a facility for 14 days (Self-Diagnosis App to be installed). Travelers who test positive for COVID-19 are transferred to nationally designated COVID-19 hospitals or residential treatment centers. Preventing the spread of the virus through epidemiological investigations and quarantine of contacts The central and local governments respond to COVID-19 cases by tracing the source of infection through prompt epidemiological investigations and quarantine of contacts. The contacts identified during the investigation are required to attend healthcare education, have their symptoms monitored, and remain in self-quarantine. Family members, housemates, and other contacts identified by epidemiological investigations on the patient’s travel and infection routes are subject to self-quarantine for the maximum incubation period (14 days) beginning from the day after the date of contact with a confirmed patient and should have their symptoms monitored. The Ministry of Interior and Safety and local governments thoroughly manage those under self-quarantine on a one-to-one basis. |
Table 2
Baseline characteristics of patients with lymphoma
Table 3
Baseline characteristics of patients with multiple myeloma
Table 4
Treatment outcomes in patients with lymphoma
Table 5
Treatment outcomes in patients with multiple myeloma
REFERENCES
1. Lotfi M, Hamblin MR, Rezaei N. COVID-19: transmission, prevention, and potential therapeutic opportunities. Clin Chim Acta 2020;508:254–266.



3. Ahn DG, Shin HJ, Kim MH, et al. Current status of epidemiology, diagnosis, therapeutics, and vaccines for novel coronavirus disease 2019 (COVID-19). J Microbiol Biotechnol 2020;30:313–324.


4. Bakouny Z, Hawley JE, Choueiri TK, et al. COVID-19 and cancer: current challenges and perspectives. Cancer Cell 2020;38:629–646.



5. Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol 2020;21:335–337.



6. Yang F, Shi S, Zhu J, Shi J, Dai K, Chen X. Clinical characteristics and outcomes of cancer patients with COVID-19. J Med Virol 2020;92:2067–2073.


7. Yu J, Ouyang W, Chua MLK, Xie C. SARS-CoV-2 transmission in patients with cancer at a tertiary care hospital in Wuhan, China. JAMA Oncol 2020;6:1108–1110.



8. Garassino MC, Whisenant JG, Huang LC, et al. COVID-19 in patients with thoracic malignancies (TERAVOLT): first results of an international, registry-based, cohort study. Lancet Oncol 2020;21:914–922.



9. Saini KS, Tagliamento M, Lambertini M, et al. Mortality in patients with cancer and coronavirus disease 2019: a systematic review and pooled analysis of 52 studies. Eur J Cancer 2020;139:43–50.



10. Kuderer NM, Choueiri TK, Shah DP, et al. Clinical impact of COVID-19 on patients with cancer (CCC19): a cohort study. Lancet 2020;395:1907–1918.


11. Westblade LF, Brar G, Pinheiro LC, et al. SARS-CoV-2 viral load predicts mortality in patients with and without cancer who are hospitalized with COVID-19. Cancer Cell 2020;38:661–671.



12. Williamson EJ, Walker AJ, Bhaskaran K, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020;584:430–436.



13. Vijenthira A, Gong IY, Fox TA, et al. Outcomes of patients with hematologic malignancies and COVID-19: a systematic review and meta-analysis of 3377 patients. Blood 2020;136:2881–2892.



14. He W, Chen L, Chen L, et al. COVID-19 in persons with haematological cancers. Leukemia 2020;34:1637–1645.


15. Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol 2014;32:3059–3068.



16. Kumar S, Paiva B, Anderson KC, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol 2016;17:e328–e346.


17. Durie BG, Harousseau JL, Miguel JS, et al. International uniform response criteria for multiple myeloma. Leukemia 2006;20:1467–1473.


18. Baden LR, Swaminathan S, Angarone M, et al. Prevention and treatment of cancer-related infections, version 2.2016, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2016;14:882–913.


19. Tunes da Silva G, Logan BR, Klein JP. Methods for equivalence and noninferiority testing. Biol Blood Marrow Transplant 2009;15(1 Suppl):120–127.


20. Sharman JP, Liberati AM, Ishizawa K, et al. A randomized, double-blind, efficacy and safety study of PF-05280586 (a Rituximab Biosimilar) compared with rituximab reference product (MabThera) in subjects with previously untreated CD20-positive, Low-Tumor-Burden Follicular Lymphoma (LTB-FL). BioDrugs 2020;34:171–181.


21. Jurczak W, Moreira I, Kanakasetty GB, et al. Rituximab biosimilar and reference rituximab in patients with previously untreated advanced follicular lymphoma (ASSIST-FL): primary results from a confirmatory phase 3, double-blind, randomised, controlled study. Lancet Haematol 2017;4:e350–e361.


22. Blackwell K, Gligorov J, Jacobs I, Twelves C. The global need for a trastuzumab biosimilar for patients with HER2-positive breast cancer. Clin Breast Cancer 2018;18:95–113.


23. Statistical Geographic Information Service. Thematic map [Internet] Daejeon (KR): SGIS, 2021. [cited 2021 Sep 22]. Available from: https://sgis.kostat.go.kr/statexp/view/index
.
24. Korean Statistical Information Service. Population and population density by region [Internet] Daejeon (KR): KOSIS, 2021. [cited 2021 Sep 22]. Available from: https://kosis.kr/index/index.do
.
25. Statistics Korea Government Official Work Conference. Population and population density by region [Internet] Daejeon (KR): Statistics Korea, 2021. [cited 2021 Sep 22]. Available from: https://index.go.kr/main.do
.
26. Korean Ministry of Health and Welfare. COVID-19 response [Internet] Sejong (KR): MOHW, 2021. [cited 2021 Sep 22]. Available from: http://ncov.mohw.go.kr
.
27. Statistics Korea. COVID-19 [Internet] Daejeon (KR): Statistics Korea, 2021. [cited 2021 Sep 22]. Available from: https://kosis.kr/covid/covid_index.do
.
28. Ministry of the Interior and Safety. Census data [Internet] Sejong (KR): MOIS, 2020. [cited 2021 Sep 22]. Available from: https://www.mois.go.kr
.
29. Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA 2020;324:782–793.


30. Shiu EYC, Leung NHL, Cowling BJ. Controversy around airborne versus droplet transmission of respiratory viruses: implication for infection prevention. Curr Opin Infect Dis 2019;32:372–379.


31. Chu DK, Akl EA, Duda S, et al. Physical distancing, face masks, and eye protection to prevent person-to-person transmission of SARS-CoV-2 and COVID-19: a systematic review and meta-analysis. Lancet 2020;395:1973–1987.



32. Guner R, Hasanoglu I, Aktas F. COVID-19: prevention and control measures in community. Turk J Med Sci 2020;50(SI-1):571–577.



33. Hu LQ, Wang J, Huang A, Wang D, Wang J. COVID-19 and improved prevention of hospital-acquired infection. Br J Anaesth 2020;125:e318–e319.


34. Juang SF, Chiang HC, Tsai MJ, Huang MK. Integrated hospital quarantine system against COVID-19. Kaohsiung J Med Sci 2020;36:380–381.



35. Lee IK, Wang CC, Lin MC, Kung CT, Lan KC, Lee CT. Effective strategies to prevent coronavirus disease-2019 (COVID-19) outbreak in hospital. J Hosp Infect 2020;105:102–103.



36. Mizumoto K, Kagaya K, Zarebski A, Chowell G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Euro Surveill 2020;25:2000180.


37. Sutton D, Fuchs K, D’Alton M, Goffman D. Universal screening for SARS-CoV-2 in women admitted for delivery. N Engl J Med 2020;382:2163–2164.


38. Day M. COVID-19: four fifths of cases are asymptomatic, China figures indicate. BMJ 2020;369:m1375.


39. Bai Y, Yao L, Wei T, et al. Presumed asymptomatic carrier transmission of COVID-19. JAMA 2020;323:1406–1407.



40. Rothe C, Schunk M, Sothmann P, et al. Transmission of 2019-nCoV infection from an asymptomatic contact in Germany. N Engl J Med 2020;382:970–971.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement 1
Supplement 1 Print
Print



