 |
 |
| Korean J Intern Med > Volume 33(6); 2018 > Article |
|
This article has been corrected. See Korean J Intern Med. 2019 Apr 30; 34(3): 685.
Abstract
Background/Aims
Vitamin D modulates innate and adaptive immune responses, and vitamin D deficiency is associated with increased mortality in hospitalized patients with pneumonia. We evaluated the prevalence of vitamin D deficiency in Korean patients with acute respiratory distress syndrome (ARDS) and its effect on the clinical outcomes of ARDS.
Methods
We retrospectively analyzed the data of 108 patients who had a measured serum level of 25-hydroxy vitamin D3 (25(OH)D3) at the time of diagnosis with ARDS. The clinical outcomes were evaluated based on 25(OH)D3 levels of 20 ng/mL and stratified by quartiles of 25(OH)D3 levels.
Results
The mean age of patients was 59.4 years old; 77 (71.3%) were male. Vitamin D deficiency was found in 103 patients (95.4%). The mean 25(OH)D3 level was 8.3 ┬▒ 7.0 ng/mL. Neither in-hospital mortality (40.0% vs. 68.0%) nor 6-month mortality (40.0% vs. 71.8%) significantly differed between groups. There were no significant differences in 25(OH)D3 level between survivors (8.1 ┬▒ 7.6 ng/mL) and non-survivors (8.5 ┬▒ 6.8 ng/mL, p = 0.765). There were no trends toward a difference in mortality among quartiles of 25(OH)D3 levels. However, 25(OH)D3 levels were inversely related with length of hospital stay and intensive care unit stay among in-hospital survivors.
Acute respiratory distress syndrome (ARDS) is a life-threatening, diffuse inflammatory lung injury that leads to increased pulmonary vascular permeability and loss of aerated lung tissue. Although many studies have reported risk factors for mortality in ARDS, such as age, disease severity, underlying cause of the ARDS, presence of multiorgan dysfunction, and fluid balance, no single factor has proven to be a greater risk factor than the others [1-7].
Vitamin D is primarily known for its important role in bone and calcium homeostasis. It is estimated that 30% to 50% of the population worldwide suffers from a form of vitamin D deficiency. In the Korea National Health and Nutrition Examination Survey, vitamin D deficiency was found in 47.3% of Korean males and 64.5% of Korean females [8]. Recently, increasing amounts of data have shown the role of vitamin D in a variety of other physiological functions, including immune responses [9-11]. Because vitamin D modulates the innate and adaptive immune responses, its deficiency appears to lead to an increased susceptibility to infection and, in a genetically susceptible host, a diathesis to autoimmunity. Many studies have demonstrated a connection between vitamin D deficiency and a number of diseases [11-16]. In addition, vitamin D deficiency is commonly found in hospitalized patients with pneumonia or sepsis and it is associated with increased mortality in pneumonia [17-20]. Recently, Dancer et al. [21] found vitamin D deficiency in all patients with ARDS and in the vast majority of patients at risk of developing ARDS. In addition, in that study it was demonstrated that vitamin D deficiency resulted in alveolar inflammation, epithelial damage, and hypoxia, using a murine model [21]. However, they did not show an association between vitamin D deficiency and clinical outcomes of ARDS.
We here evaluated the prevalence of vitamin D deficiency in Korean patients with ARDS and the effect of vitamin D deficiency on the clinical outcomes of ARDS.
We retrospectively analyzed 108 Korean patients who had a measured serum level of 25-hydroxy vitamin D3 (25(OH)D3) at the time of diagnosis of ARDS between April 2005 and March 2016 at Asan Medical Center in Republic of Korea. We identified patients meeting the Berlin ARDS criteria [22]. For each patient, the following data were collected by retrospective review of medical records: age, sex, medical history, diseases predisposing the subject to ARDS, laboratory data, acute physiology and chronic health evaluation II (APACHE II) score, lengths of stay in the intensive care unit (ICU) and hospital, and mortality. The study protocol was approved by the Institutional Review Board of Asan Medical Center, which waived the requirement for informed consent because of the retrospective nature of the analyses (IRB No. 2015-0164).
To evaluate the prevalence of vitamin D deficiency, we considered a serum level of 25(OH)D3 < 20 ng/mL to be deficient, in line with the guidelines of the Endocrine Society [23]. The baseline characteristics and clinical outcomes of patients with a status of vitamin D deficiency were compared to those in the normal range. As a large number of patients were expected to have vitamin D deficiency, we also stratified patients by quartile of 25(OH)D3 levels. Clinical outcomes such as 28-day allcause, in-hospital, 6-month, and 1-year mortalities were evaluated in each quartile. We also evaluated the lengths of stay in the ICU, those in the hospital, and ICU, hospital-free survival.
A large number of patients started continuous renal replacement therapy when they were diagnosed with ARDS. As acute kidney injury (AKI) is associated with higher mortality in patients with ARDS, we also evaluated the effects of vitamin D on the recovery of renal function [24]. We defined AKI as the abrupt loss of kidney function, resulting in dialysis, in the course of ARDS. Recovery of renal function was defined as the lack of a need for further dialysis. Serum 25(OH)D3 concentrations were measured using the DIA-source 25OH-Vit. D3-Ria-CT Kit (DIAsource ImmunoAssays SA, Louvainla-Neuve, Belgium; Cobra II Auto-╬│ Counting System, Packard Instruments, Downers Grove, IL, USA). The APACHE II score was assessed on the first day of admission to the ICU.
Categorical variables were analyzed using either a Pearson chi-square test or a Fisher exact test, and continuous variables were analyzed using either a Student t test, Mann-Whitney test, or frequency analysis. The prevalence of the vitamin D level was validated using bootstrapping, a nonparametric method that repeating chi-square analysis 1,000 times after random selection of case. Survival curves were plotted by the Kaplan-Meier method and were compared by using a log-rank test. Odds ratios for univariate survival analyses (p < 0.1) were calculated using the Cox proportional hazard model. All tests for significance were two-sided, and all variables with p < 0.05 were considered significant. All statistical analyses were performed with SPSS software version 21.0 (IBM Co., Armonk, NY, USA).
We included 108 patients with ARDS. The baseline characteristics of the included patients are presented in Table 1. The mean age of the patients was 59.4 ┬▒ 12.7 years old; 77 (71.3%) were males. Pneumonia (71/108, 65.7%) was the most common predisposing condition, followed by sepsis caused other than by pneumonia (22/108, 20.4%). The mean APACHE II score was 26.3 ┬▒ 8.5 and there were no significant differences between the two groups (p = 0.646). A total of 93 patients (86.1%) developed AKI requiring dialysis after the diagnosis of ARDS.
Of 108 patients with ARDS, 103 (95.4%) had vitamin D deficiency and five (4.6%) had a normal level of vitamin D. Only three patients satisfied the 25(OH)D3 level recommended by the Endocrine Society, of > 30 ng/mL [23]. After random sampling for 1,000 times and 10,000 times, the median frequency of vitamin D deficiency were 95.3% and 95.4%, respectively. The mean level across all patients was 8.3 ┬▒ 7.0 ng/mL, with 32.4 ┬▒ 10.2 ng/mL in the normal group and 7.2 ┬▒ 4.3 ng/mL in the deficiency group (p < 0.001). We could not find any significant differences in serum 25(OH)D3 levels and prevalence of vitamin D deficiency between in-hospital survivors and non-survivors (Table 2). In the vitamin D deficiency group, serum 25(OH)D3 levels were not significantly different between in-hospital survivors and non-survivors (p = 0.071). The p value was similar to the median p value after bootstrapping (Supplementary Table 1). The levels were consistent in 28-day, 6-month, and 1-year survivors and non-survivors. Seasonal variation in serum 25(OH)D3 levels was not observed in the study population.
The overall 28-day mortality of the study population was 42.6% (46/108). There was no significant difference in 28-day mortality between the two groups (40.0% in the normal group vs. 42.7% in the deficiency group). Neither in-hospital (40.0% vs. 68.0%) nor 6-month mortality (40.0% vs. 71.8%) was significantly different between the two groups. There were no significant differences in hospital-free and ICU-free 1-year survival between the two groups.
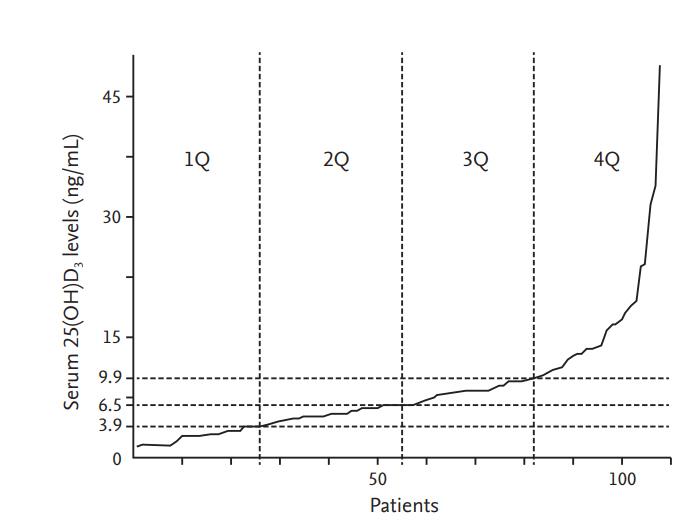
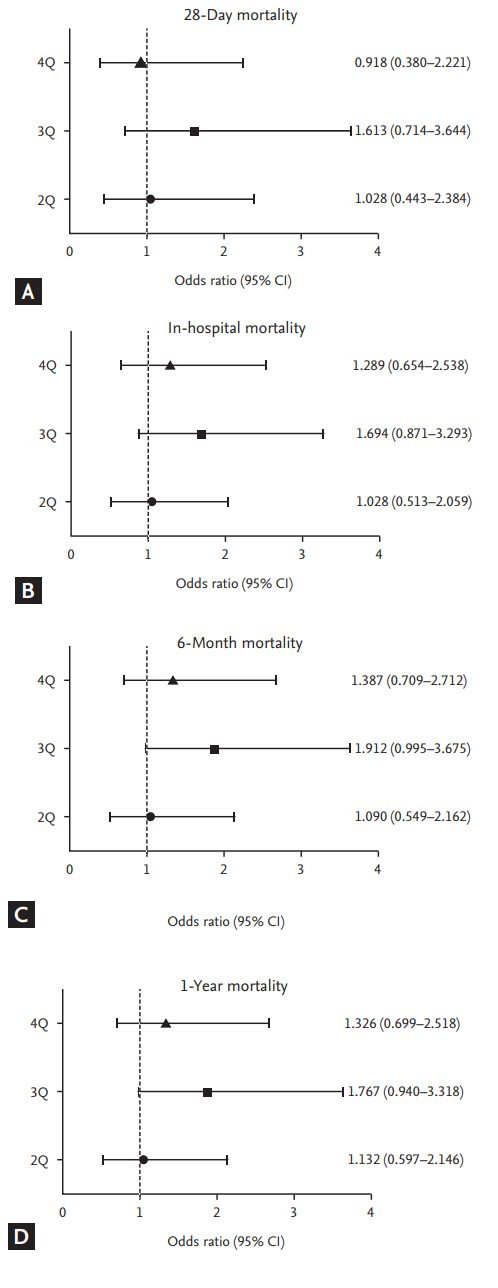
As most of patients had vitamin D deficiency, we stratified subjects by quartile of 25(OH)D3 levels. The quartile stratifications were as follows (Fig. 1); a 25(OH)D3 level less than or equal to 3.9 ng/mL, more than 3.9 to 6.5 ng/mL, more than 6.5 to 9.9 ng/mL, and more than 9.9 ng/mL. Kaplan-Meier curves did not show any significant differences of survival among the quartiles. In addition, neither ICU-free survival nor hospital-free survival was different among the quartiles. The Cox proportional hazards model was used to investigate whether the clinical features, such as age, sex, predisposing condition, APACHE ŌģĪ score, and quartiles of vitamin D level influenced on survival in study population. As a result, there was no significant association with these variables and survival. We could not find any relationship between quartiles of vitamin D levels and survival (Fig. 2).
Patients with normal vitamin D levels had longer stay in the hospital (median, 46 days vs. 31 days) and shorter stays in the ICU (14 days vs. 16 days) than those with vitamin D deficiencies. There were no trends in lengths of stay in the hospital and in the ICU. However, quartiles of serum 25(OH)D3 levels showed trends that they were inversely related with lengths of stay in the hospital and in the ICU among in-hospital survivors (Fig. 3). The median (interquartile range [IQR]) length of stay in the hospital and in the ICU of the first quartile was 52 days (IQR, 30 to 65) and 24 days (IQR, 13 to 38), respectively. Meanwhile, the median length of stay in the hospital and in the ICU of the fourth quartile was 39 days (IQR, 30 to 58) and 14 days (IQR, 10 to 24), respectively (Fig. 3C and 3D).
A total of 93 patients (93/108, 86.1%) developed AKI that required dialysis in the course of ARDS. Of these, 23 (26/93, 28.0%) recovered from AKI. Serum 25(OH)D3 levels did not differ among patients who recovered from AKI and those who did not (8.5 ┬▒ 8.2 ng/mL vs. 8.5 ┬▒ 6.6 ng/mL, p = 0.996).
In the present study, 95.4% of the patients with ARDS had a vitamin D deficiency. We did not find any relationship between serum levels of 25(OH)D3 and mortality in ARDS. Serum 25(OH)D3 levels showed trends that they were inversely related with lengths of stay in the hospital and in the ICU among in-hospital survivors.
There have been conflicting data regarding the importance of underlying comorbidities or risk factors for ARDS mortality [25-29]. As data on the role of vitamin D in immune response increases, the effects of vitamin D on chronic inflammatory and autoimmune diseases have been reevaluated. Dancer et al. [21] reported that deficiency of vitamin D was common in patients with ARDS. It is not clear that how vitamin D deficiency does damage to lung epithelial cells. Dancer et al. [21] demonstrated that vitamin D deficiency was associated with greater alveolar cellular inflammation, cytokine release, and epithelial damage using a murine model. In addition, microarray analysis revealed that vitamin D treatment had trophic effects on genes for human alveolar epithelial cells. Several studies have shown that vitamin D deficiency is associated with higher mortality in pneumonia, regardless of other confounding factors, such as age, comorbidities, and the pneumonia severity index [18,30,31]. This might suggest that vitamin D deficiency is related to severe inflammatory reactions in the lung as well as dysfunctional macrophage and antimicrobial effects [17,32].
Dancer et al. [21] reported that survivors of ARDS had significantly higher levels of vitamin D than non-survivors. However, we could not find any significant differences in serum levels of 25(OH)D3 between survivors and non-survivors. These results might suggest that levels of vitamin D are not related to mortality of ARDS. Meanwhile, in-hospital survivors in the higher quartile of 25(OH)D3 levels had shorter lengths of stay in the hospital and in the ICU than those in the lower quartile. Therefore, the heterogenous characteristics of the study subjects, with various kinds of comorbidities and predisposing conditions, might have acted as confounding factors. In addition, the results might be affected by selection bias, because 25(OH)D3 levels were not measured in all ARDS patients but principally in those who had received dialysis, a serious condition suggestive of multiorgan failure.
Vitamin D deficiency was found in 95.4% of included patients. It is a much higher prevalence than the result of medical checkup data, where it is found in 62.1% of 7,003 medical health checkup recipients at Asan Medical Center in 2012. Despite the widespread vitamin fortification of food, vitamin D deficiency has increased among city dwellers, whose exposure to sunlight has decreased due to indoor activity. Among patients of the present study, 28 (25.9%) had previously received liver or kidney transplantation before developing ARDS. In addition, 16 patients (14.8%) were receiving regular dialysis because of an underlying end-stage renal disease, which leads to a decline in circulating concentrations of calcitriol. These serious medical conditions can lead to vitamin D deficiency. However, only four patients (3.7%) had received vitamin D supplements before developing ARDS and no subject began to receive vitamin D supplements after developing ARDS. Therefore, we could not evaluate whether vitamin D supplementation can improve the clinical outcomes of ARDS.
The present study has several limitations. First, number of patients in normal range of vitamin D was too small. However, vitamin D deficiency was found in most of study subjects in the present study and all patients in the study by Dancer et al. [21]. It might be inevitable since vitamin D deficiency is highly prevalent among patients with ARDS. In addition, we performed additional analysis with stratified patients by quartile of 25(OH)D3 levels to overcome this limitation. Second, we only included subjects with measured serum 25(OH)D3 levels. This could have led to selection bias and therefore our study cohort may not be representative of all ARDS patients. In addition, the number of included patients was small, with various kinds of comorbidities and predisposing factors. Although there were no significantly different baseline characteristics between the groups, these factors might have acted as confounding factors. Therefore, a large-scale prospective study with well-controlled cohorts is needed for further evaluation. Third, the definition of vitamin D deficiency in the present study was taken from the guidelines for bone and calcium metabolism. How much vitamin D is needed for its appropriate immunologic role is not yet well defined. The reference values were different in each study in the association of vitamin D and pneumonia [17,18,30,31]. Although we could not find any tendencies in clinical outcomes according to quartile, the definition of the optimal level of vitamin D for its immunologic role remains to be completed. Ultimately, it needs to be investigated whether vitamin D supplementation can improve clinical outcomes in ARDS.
In conclusion, a vitamin D deficiency was highly prevalent in patients with ARDS. Although we did not find any relationship between 25(OH)D3 levels and mortality in ARDS cases, a large, prospective study is still needed to further evaluate the effects of vitamin D deficiency on the clinical outcomes of ARDS.
1. Vitamin D deficiency was common in patients with acute respiratory distress syndrome.
2. There were no differences in vitamin D level between survivors and non-survivors.
3. Levels of vitamin D were not related with mortality of acute respiratory distress syndrome.
4. Levels of vitamin D were inversely related with length of hospital and intensive care unit stay among in-hospital survivors.
Supplementary Materials
Supplementary┬ĀTable┬Ā1.
Range of p value in comparison of serum 25-hydroxy vitamin D3 levels between in-hospital survivors and non-survivors after bootstrapping
Figure┬Ā1.
Distribution of serum concentrations of 25-hydroxy vitamin D3 (25(OH)D3) in subjects with acute respiratory distress syndrome. Subjects were stratified by quartiles, with cutoff values of 3.9, 6.5, and 9.9 ng/mL. Q, quartile.
Figure┬Ā2.
Odds ratio of mortality among quartiles of 25-hydroxy vitamin D3 levels. No mortality trends were found (A) 28-day mortality, (B) in-hospital mortality, (C) 6-month mortality, and (D) 1-year mortality. Q, quartile; CI, confidence interval.
Figure┬Ā3.
Length of stay in the hospital (A) and intensive care unit (B) among all subjects and those among in-hospital survivors (C, D). There was a trend towards decreasing lengths of stay in the hospital and in the intensive care unit (ICU) among in-hospital survivors. IQR, interquartile range; Q, quartile; 25(OH)D3, 25-hydroxy vitamin D3.

Table┬Ā1.
Baseline characteristics of the study subjects with acute respiratory distress syndrome
Table┬Ā2.
Serum concentrations of 25-hydroxy vitamin D3 in survivors and non-survivors
REFERENCES
1. Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and outcomes of acute lung injury. N Engl J Med 2005;353:1685ŌĆō1693.


2. Bone RC, Maunder R, Slotman G, et al. An early test of survival in patients with the adult respiratory distress syndrome. The PaO2/FIo2 ratio and its differential response to conventional therapy. Prostaglandin E1 Study Group. Chest 1989;96:849ŌĆō851.


3. Nin N, Muriel A, Penuelas O, et al. Severe hypercapnia and outcome of mechanically ventilated patients with moderate or severe acute respiratory distress syndrome. Intensive Care Med 2017;43:200ŌĆō208.




4. Stapleton RD, Wang BM, Hudson LD, Rubenfeld GD, Caldwell ES, Steinberg KP. Causes and timing of death in patients with ARDS. Chest 2005;128:525ŌĆō532.


5. Doyle RL, Szaflarski N, Modin GW, Wiener-Kronish JP, Matthay MA. Identification of patients with acute lung injury. Predictors of mortality. Am J Respir Crit Care Med 1995;152(6 Pt 1):1818ŌĆō1824.


6. Bone RC, Balk R, Slotman G, et al. Adult respiratory distress syndrome. Sequence and importance of development of multiple organ failure. The Prostaglandin E1 Study Group. Chest 1992;101:320ŌĆō326.


7. Rosenberg AL, Dechert RE, Park PK, Bartlett RH; NIH NHLBI ARDS Network. Review of a large clinical series: association of cumulative fluid balance on outcome in acute lung injury: a retrospective review of the ARDSnet tidal volume study cohort. J Intensive Care Med 2009;24:35ŌĆō46.


8. Choi HS, Oh HJ, Choi H, et al. Vitamin D insufficiency in Korea: a greater threat to younger generation: the Korea National Health and Nutrition Examination Survey (KNHANES) 2008. J Clin Endocrinol Metab 2011;96:643ŌĆō651.



9. Barragan M, Good M, Kolls JK. Regulation of dendritic cell function by vitamin D. Nutrients 2015;7:8127ŌĆō8151.



10. Berraies A, Hamzaoui K, Hamzaoui A. Link between vitamin D and airway remodeling. J Asthma Allergy 2014;7:23ŌĆō30.


11. Mirzakhani H, Al-Garawi A, Weiss ST, Litonjua AA. Vitamin D and the development of allergic disease: how important is it? Clin Exp Allergy 2015;45:114ŌĆō125.



12. Parker J, Hashmi O, Dutton D, et al. Levels of vitamin D and cardiometabolic disorders: systematic review and meta-analysis. Maturitas 2010;65:225ŌĆō236.


13. Hong Q, Xu J, Xu S, Lian L, Zhang M, Ding C. Associations between serum 25-hydroxyvitamin D and disease activity, inflammatory cytokines and bone loss in patients with rheumatoid arthritis. Rheumatology (Oxford) 2014;53:1994ŌĆō2001.



14. Yawn J, Lawrence LA, Carroll WW, Mulligan JK. Vitamin D for the treatment of respiratory diseases: is it the end or just the beginning? J Steroid Biochem Mol Biol 2015;148:326ŌĆō337.


15. Kerley CP, Elnazir B, Faul J, Cormican L. Vitamin D as an adjunctive therapy in asthma. Part 2: a review of human studies. Pulm Pharmacol Ther 2015;32:75ŌĆō92.


16. Ananthakrishnan AN, Cheng SC, Cai T, et al. Association between reduced plasma 25-hydroxy vitamin D and increased risk of cancer in patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol 2014;12:821ŌĆō827.



17. Leow L, Simpson T, Cursons R, Karalus N, Hancox RJ. Vitamin D, innate immunity and outcomes in community acquired pneumonia. Respirology 2011;16:611ŌĆō616.


18. Holter JC, Ueland T, Norseth J, et al. Vitamin D status and long-term mortality in community-acquired pneumonia: secondary data analysis from a prospective cohort. PLoS One 2016;11:e0158536.



19. de Haan K, Groeneveld AB, de Geus HR, Egal M, Struijs A. Vitamin D deficiency as a risk factor for infection, sepsis and mortality in the critically ill: systematic review and meta-analysis. Crit Care 2014;18:660.




20. Moromizato T, Litonjua AA, Braun AB, Gibbons FK, Giovannucci E, Christopher KB. Association of low serum 25-hydroxyvitamin D levels and sepsis in the critically ill. Crit Care Med 2014;42:97ŌĆō107.


21. Dancer RC, Parekh D, Lax S, et al. Vitamin D deficiency contributes directly to the acute respiratory distress syndrome (ARDS). Thorax 2015;70:617ŌĆō624.



22. ARDS Definition Task Force; Ranieri VM, et al.; Rubenfeld GD. Acute respiratory distress syndrome: the Berlin definition. JAMA 2012;307:2526ŌĆō2533.

23. Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 2011;96:1911ŌĆō1930.


24. Darmon M, Clec'h C, Adrie C, et al. Acute respiratory distress syndrome and risk of AKI among critically ill patients. Clin J Am Soc Nephrol 2014;9:1347ŌĆō1353.



25. Caser EB, Zandonade E, Pereira E, Gama AM, Barbas CS. Impact of distinct definitions of acute lung injury on its incidence and outcomes in Brazilian ICUs: prospective evaluation of 7,133 patients*. Crit Care Med 2014;42:574ŌĆō582.


26. Seeley E, McAuley DF, Eisner M, Miletin M, Matthay MA, Kallet RH. Predictors of mortality in acute lung injury during the era of lung protective ventilation. Thorax 2008;63:994ŌĆō998.



27. Villar J, Blanco J, Anon JM, et al. The ALIEN study: incidence and outcome of acute respiratory distress syndrome in the era of lung protective ventilation. Intensive Care Med 2011;37:1932ŌĆō1941.


28. Yu S, Christiani DC, Thompson BT, Bajwa EK, Gong MN. Role of diabetes in the development of acute respiratory distress syndrome. Crit Care Med 2013;41:2720ŌĆō2732.



29. Gong MN, Thompson BT, Williams P, Pothier L, Boyce PD, Christiani DC. Clinical predictors of and mortality in acute respiratory distress syndrome: potential role of red cell transfusion. Crit Care Med 2005;33:1191ŌĆō1198.


30. Remmelts HH, van de Garde EM, Meijvis SC, et al. Addition of vitamin D status to prognostic scores improves the prediction of outcome in community-acquired pneumonia. Clin Infect Dis 2012;55:1488ŌĆō1494.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement 1
Supplement 1 Print
Print



