 |
 |
| Korean J Intern Med > Volume 18(4); 2003 > Article |
|
Abstract
Relapsing polychondritis (RP) is a rare multisystem disorder. Myelodysplastic syndrome (MDS) with erythroid hypoplasia/aplasia is a rare form of myelodysplasia. Several cases of RP associated with MDS have recently been described. However, RP associated with MDS with erythroid hypoplasia/aplasia has never been reported. There was only one case report of polymyalgia rheumatica associated with MDS with erythroid hypoplasia/aplasia. In this study, we report a 79-year-old patient with RP, who developed MDS subtype refractory anemia (RA) with erythroid hypoplasia/aplasia, a very characteristic subtype of MDS.
Relapsing polychondritis (RP) is a rare autoimmune disease of unknown etiology characterized by recurrent inflammatory episodes primarily affecting the auricular, nasal or laryngotracheal cartilage, as well as the ocular, audiovestibular and cardiovascular systems1,2). The myelodysplastic syndromes (MDS) are clonal disorders of hematopoiesis characterized by peripheral cytopenias and a dysplastic bone marrow that are usually hypercellular. MDSs characteristically exhibit a high number (between 5% and 50%) of erythroid precursors in the marrow3). However, a minority have merely a small amount (less than 5%) of recognizable erythroid cells in the bone marrow. Within this group, most of the patients with erythroid hypoplasia have the so-called MDS with erythroid hypoplasia/aplasia whose pathogenic mechanism remains unclear.
A review of the literature reveals only 16 well-documented cases of MDS with erythroid hypoplasia/aplasia4). There are several reports of a simultaneous or temporally close occurrence of RP and myelodysplastic/myeloproliferative conditions5ŌĆō9). Here, we report a 79-year-old patient with RP who developed MDS subtype refractory anemia (RA) with erythroid hypoplasia/aplasia. To our knowledge, this is the first case of RP associated with MDS with erythroid hypoplasia/aplasia.
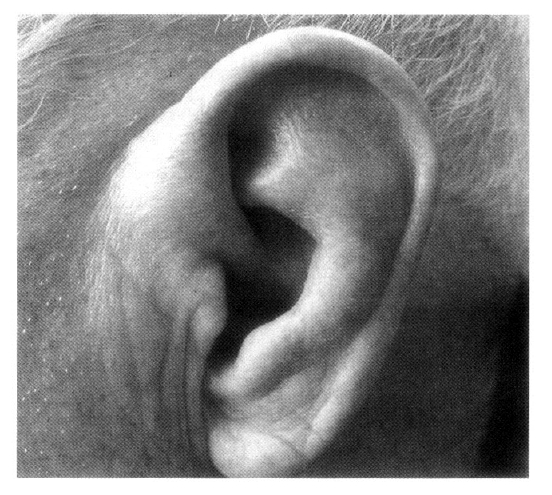
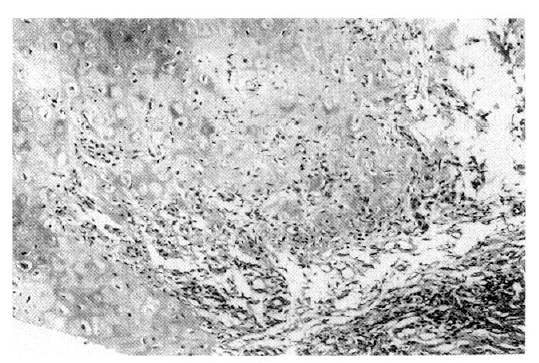
A 79-year-old male was admitted to our hospital in October 2002 because of a five-day history of fever, chills, and productive cough. He was diagnosed with RP in 1997 due to recurrent swelling and redness of both ears (Figure 1), conjunctivitis, general ache and myalgia, which was confirmed by an ear biopsy (Figure 2). Since then, he has been treated with low-dose glucocorticoid, NSAID and colchicine. Manifestations of RP were cleared under treatment with low-dose glucocorticoid over 5 years. On admission, he complained of fever, chills, and cough.
On physical examination, his blood pressure was 130/80 mmHg, pulse rate 108/min and body temperature 38.1┬░C. He had a cushingoid appearance and pale conjunctivae. Auscultation revealed rales on the right lung.
The blood count showed macrocytic anemia: hemoglobin 6.8 g/dL, hematocrit 20.6%, MCV 107 fL, and MCH 35.2 pg. Slight leukocytosis (WBC count=10,400/╬╝L) and moderate thrombocytopenia (platelet count=99,000/╬╝L) were noted. ESR was 43 mm/h and CRP was positive (36.9 mg/dL). The peripheral blood smear showed anisocytosis, polychromasia, and poikilocytosis. Biochemical tests were all within normal ranges except for slightly elevated LDH (499 IU/L) and decreased protein/albumin levels (6.0/2.7 g/dL). Serum iron level and saturation of transferrin were increased (164 ╬╝g/dL and 91%, respectively). Serum ferritin levels were (923 ng/mL). There was no vitamin B12 deficiency (serum level: 963 pg/mL; normal 200ŌĆō950 pg/mL) or folic acid deficiency (serum level: 14.5 ng/mL; normal 2.5ŌĆō20 ng/mL). The Direct Coombs test was positive, while the indirect Coombs test was negative. Serological tests for HIV and hepatitis B were negative. Tests for serum anti-nuclear antibodies, anti-neutrophil cytoplasmic antibodies and rheumatoid factor were all negative.
Chest radiographs showed air-space consolidations in the upper and lower right lobes with a small amount of right pleural effusion. Broad-spectrum antibiotics were started. Four weeks later, there was marked improvement in his chest radiographs. He no longer had a fever, and his productive cough had also improved. However, transfusion-dependent anemia (hemoglobin=7.8 g/dL) and thrombocytopenia (platelet count=24,000/╬╝L) remained.
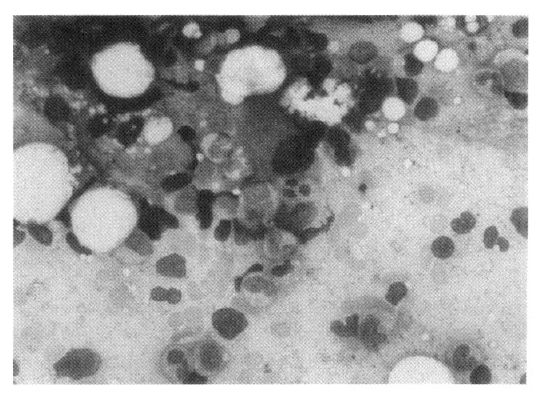
Bone marrow aspiration and biopsy were performed (Figure 3). This revealed a hypercellular marrow for his age with decreased megakaryocytes. A myeloid to erythroid ratio was markedly increased (21:1) due to the markedly decreased erythroid series. Dysplastic changes were seen in the myeloid series and megakaryocytes. Approximately 4.5% of blasts were present. Cytogenetic analysis showed a normal karyotype (46 XY). Serological tests for human parvovirus B19 were positive for IgG and negative for IgM.
A diagnosis of MDS subtype RA with erythroid hypoplasia/aplasia was made. He was treated with prednisolone 5 mg daily until discharge, and received packed red cell transfusion once a week. With low dose prednisolone therapy and intermittent transfusion, the patient has remained in a stable clinical and hematologic condition. At a follow up three months after diagnosis, his blood count showed WBC count 2,800/╬╝L, hemoglobin 6.4 g/dL and platelets 119,000/╬╝L.
RP is a rare, episodic inflammatory condition affecting cartilaginous tissues, especially the ear, nose and tracheobronchial cartilage1,2). Diagnosis is based on the characteristic clinical features. The diagnostic criteria were described by McAdam et al1). Our patient fulfilled these criteria, having both recurrent auricular chondritis and ocular inflammation. Furthermore, the diagnosis was confirmed by a biopsy of the left ear cartilage.
RP is thought to be an autoimmune disorder. Autoantibodies to type 2 collagen, association with other autoimmune disorders and response to steroids by reduction of the inflammatory response all support this view. In our patient, the manifestations of RP were cleared under treatment with low-dose glucocorticoid over 5 years.
MDSs are a group of acquired neoplastic disorders of bone marrow, characterized by quantitative and functional abnormalities of all three hematopoietic lineages caused by a defect at the stem cell stage3). MDSs characteristically exhibit a high number of erythroid precursors in the marrow. However, a minority have merely a small amount (less than 5%) of recognizable erythroid cells in the bone marrow. Within this group, most of the patients with erythroid hypoplasia have the so-called MDS with erythroid hypoplasia/aplasia4). Our patient showed 4% of erythroid precursors and markedly increased myeloid to erythroid ratio (21:1) in the marrow.
The association of RP and MDS has only occasionally been described. Michet et al. reviewed 112 cases of RP, amongst whom six patients developed hematological clonal disorders10). Van Besien et al. reported a case of RP associated with MDS subtype refractory anemia with excess blasts in transformation (RAEB-t)5). Mongin et al. reported 12 cases of RP associated with MDS, in which MDS preceded RP, occurred simultaneously, or developed subsequently11). In 1997, our patient was diagnosed with relapsing polychondritis. Initial investigations showed an anemia of hemoglobin 9.4 g/dL and normocellular marrow. Five years later, a diagnosis of MDS was made by a bone marrow aspiration. The association between MDS and RP certainly does not appear fortuitous, but the mechanism of this association continues to remain obscure7,8). However, the fact that there was anemia when he was diagnosed with RP and that RP preceded the bone marrow changes by five years add currency to the notion that RP may represent a paraneoplastic phenomenon of an underlying MDS, or alternatively, that MDS and RP may be caused by a single immunological abnormality leading to an autoimmuneŌĆÖ condition6).
Interestingly, the MDS in our case had further characteristic features. It showed a dearth of erythroid seriesŌĆÖ, and he was diagnosed with MDS with erythroid hypoplasia/aplasia, a very rare subtype of MDS. Our patient is the first case reported in the literature with RP associated with MDS with erythroid hypoplasia/aplasia. Cook reported a case of polymyalgia rheumatica concurrent with erythroid hypoplasia/aplasia associated with myeloproliferative and myelodysplastic syndrome12). In his case, the patient initially responded well to prednisolone, but relapsed after tapering off the steroid.
Although MDS with erythroid hypoplasia/aplasia is usually highly resistant to therapy, the efficacy of steroids for erythroid hypoplasia/aplasia occuring in patients with MDS is also reported in several cases4,13). Williamson et al. reported six cases of erythroid hypoplasia/aplasia occuring in patients with MDS13). They showed a diversity of clinical courses and prognoses. Three patients died within a short time of developing erythroid hypoplasia/aplasia associated with an increased blast percentage in the marrow. The other three patients showed at least a partial recovery of erythropoiesis after being treated with prednisolone. Their marrow initially showed MDS subtype RA with 1% or fewer erythroblasts. Williamson et al. suggested that the response to steroid supports an autoimmune etiology superimposed on MDS rather than an intrinsic stem cell defect13). This supports the view that those patients who have erythroid hypoplasia/aplasia and no excess of blasts may have a relatively good prognosis and should be considered for immunosuppressive treatment13).
MDS with erythroid hypoplasia/aplasia is a rare form of myelodysplasia, with only 16 well-documented cases reported in the literature. Patients with this disorder were predominantly elderly males at presentation, all requiring regular blood transfusions, and with an unfavorable prognosis because of a high risk of blastic transformation4).
The pathogenic mechanism of MDS with erythroid hypoplasia/aplasia in RP patients remains uncertain. Although, a number of autoimmune diseases including CrohnŌĆÖs disease, ulcerative colitis, BehcetŌĆÖs disease, pernicious anemia, hypothyroidism, polymyalgia rheumatica, inflammatory arthritis and RP-have been suggested to occur rather frequently in MDS, Garcia-Suarez et al. reported that most patients (15/16) with MDS with hypoplasia/aplasia did not have immunologic disorders known to be associated with autoimmune erythroid hypoplasia/aplasia4,7,14). There was only one case of polymyalgia rheumatica.
In this report, we add another immunologic disorder associated with the very rare MDS with erythroid hypoplasia/aplasia. We believe that MDS with erythroid hypoplasia/aplasia may also be related to autoimmune diseases, a feature not different from other MDS subtypes.
REFERENCES
1. McAdam LP, OŌĆÖ Hanlan MA, Bluestone R, Pearson CM. Relapsing polychondritis: prospective study of 23 patients and a review of the literature. Medicine (Baltimore) 55:193ŌĆō2151976.


2. Damiani JM, Levine HL. Relapsing polychondritis: report of ten cases. Laryngoscope 89:929ŌĆō9461979.


3. Kouides PA, Bennett JM. Morphology and classification of myelodysplastic syndromes. Hematology/Oncology clinics of North America 6:485ŌĆō4991992.

4. Garcia-Suarez J, Pascual T, Munoz MA, Herrero B, Pardo A. Myelodysplastic syndrome with erythroid hypoplasia/aplasia: a case report and review of the literature. Am J Hematol 58:319ŌĆō3251998.


5. Van Besien K, Tricot G, Hoffman R. Relapsing polychondritis: a paraneoplastic syndrome associated with myelodysplastic syndromes. Am J Hematol 40:47ŌĆō501992.


6. Diebold L, Rauh G, Jager K, Lohrs U. Bone marrow pathology in relapsing polychondritis: high frequency of myelodysplastic syndromes. Br J Hematol 89:820ŌĆō8301995.

7. Hebbar M, Brouillard M, Wattel E, Decoulx M, Hatron PY, Devulder B, Fenaux P. Association of myelodysplastic syndrome and relapsing polychondritis: further evidence. Leukemia 9:731ŌĆō7331995.

8. Myers B, Gould J, Dolan G. Relapsing polychondritis and myelodysplasia: a report of two cases and review of the current literature. Clin Lab Haematol 22:45ŌĆō482000.


9. Wright MJ, BelŌĆÖeed K, Sellars L, Richmond I. Relapsing polychondritis and myelodysplasia. Nephrol Dial Transplant 12:1704ŌĆō17071997.


10. Michet CJ, McKenna CH, Luthra HS, OŌĆÖ Fallon WM. Relapsing polychondritis: survival and predictive role of early disease manifestations. Ann intern Med 104:74ŌĆō781986.


11. Mongin M, Gabriel B, Weiller PJ. La polychondrite atrophiante et ses associations pathologiques. Bulletin Academic National Medicine 172:109ŌĆō1131988.
12. Cook MK. Red cell hypoplasia associated with myeloproliferative and myelodysplastic syndrome. J Clin Pathol 42:890ŌĆō8911989.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



