 |
 |
| Korean J Intern Med > Volume 12(2); 1997 > Article |
|
Abstract
Objectives
Hepatitis B virus (HBV) with a stop mutation at precore codon 28 (TGGŌåÆTAG, tryptophanŌåÆstop) was investigated to clarify if such a mutant virus might play a role in hepatocarcinogenesis.
Methods
A total of 73 patients with HBV-related hepatocellular carcinoma were included in this study. Polymerase chain reaction (PCR) was performed in DNA samples extracted from 73 sera to amplify a HBV-DNA segment involving the precore and proximal core regions, and sequences of PCR products were analyzed to see the presence of the mutations at precore codon 28 by a direct sequencing method.
Results
HBV-DNA was detectable in 64 (88%) patients by PCR. The stop mutation at precore codon 28 was identified in 50 of 58 PCR products (86%), in which direct sequencing was performed. Among patients with this mutant HBV, 21/50 (42%) patients were co-infected with wild-type HBV. The mutant virus was found in 23/28 (82%) patients with hepatitis B e antigen (HBeAg) and 27/30 (90%) patients without HBeAg. The mutant HBV alone was found in 10/28 (36%) patients with HBeAg and 19/30 (63%) without HBeAg. Among those patients on whom laparoscopy was performed, 22/24 (92%) with the precore codon 28 stop mutant alone had cirrhosis, compared to 12/19 (63%) co-infected by both the mutant and the wiId-type (p<0.05). The association of this mutant virus with both the presence and absence of HBeAg, and its association with cirrhosis when there is no co-infection with wild-type HBV, suggests an evolving pattern of liver pathology.
Hepatocellular carcinoma (HCC) is frequently associated with hepatitis B virus (HBV) infection1,2). Aspects of HBV infection that possibly may contribute to the malignant transformation of hepatocytes include integration of HBV in the host genome3,4) and the production by HBV of transactivating proteins, such as the protein products of the X gene5ŌĆō7) and a part of the pre-S2/S gene8,9). However, no specific pattern of HBV-DNA integration has been detected so far10,11) and no transforming oncogene has been discovered within the HBV genome12). Thus, the molecular mechanism of HBV-associated hepatocarcinogenesis is still poorly understood.
HBV containing a stop mutation of precore codon 28 has been isolated frequently from the serum of some HBV chronic carriers13ŌĆō16). This mutant cannot produce HBeAg but retains its replicative capacity17). It has been reported often in association with the development of fulminant hepatitis14,18) and it has been found in some HCC patients19,20). It is possible that this mutant virus could contribute to the development of HCC. In the present study, HBV from the serum of HCC patients was analyzed to detect a stop mutation at precore codon 28 in an attempt to define further the possible role of such a mutation in hepatocarcinogenesis.
Seventy-three HCC patients with hepatitis B surface antigen (HBsAg) and 19 controls with HBsAg (without HCC) were seen at the Department of Internal Medicine, Kangnam St. MaryŌĆÖs Hospital, Catholic University Medical College, Seoul, Korea, between 1990 and 1993 (Table 1). All were negative for antibody to the hepatitis C virus (anti-HCV). Of the 73 HCC patients, the diagnosis of HCC was made histologically in 61 and was based on elevated levels of alpha-fetoprotein (AFP) (>400ng/ml) and CT scan in 12. Cirrhosis was visualized at laparoscopy in 46/61 (75%) HCC patients. Serum samples were collected at the time of the initial hospital admission and were stored at ŌłÆ20┬░C until tested in the present study. The 19 controls had histologic evidence of chronic active hepatitis and included 9 with HBeAg and 10 with antibody to HBeAg (anti-HBe) (HBeAg-negative).
For patients and controls, sera were tested for HBsAg, antibody to HBsAg, HBeAg, anti-HBe and antibody to the hepatitis B core antigen using commercially available radioimmunoassays (Abbott Laboratories, North Chicago, IL, USA), and antibody to the hepatitis C virus (anti-HCV) using commercially available enzyme immunoassays (Abbott Laboratories).
One hundred microliters of each serum sample were mixed with 200ng of proteinase K solution (20mg/ml; Boehringer Mannheim, Mannheim, Germany) in 100 ╬╝l of 100mM NaCl, 10 mM Tris-HCl (pH 8.0), 25mM ethylenediaminetetraacetic acid (pH 8.0), and 0.5% sodium dodecyl sulfate, and incubated at 56┬░C for 2 hours. This was sequentially extracted with an equal volume of phenol, with an equal volume of a solution of phenol: chloroform: isoamyl alcohol (25:24:1), and with an equal volume of chloroform. The extracted sample was mixed with 3 ╬╝l glycogen and one-tenth volume of 3M sodium acetate (pH 5.2) and precipitated overnight with three volumes of cold 100% ethanol. The precipitated DNA was washed with 70% ethanol, air dried, and dissolved in 100 ╬╝l TE buffer (10mmol/L Tris-HCl and 0.1mmol/L EDTA, pH 8.0).
The reaction mixture (100 ╬╝l) contained 50 ╬╝l of prepared DNA template, 10mmol/L Tris (pH 8.3), 50mmol/L KCl, 1.5mmol/L MgCl2, 1% (w/v) gelatin, 0.5 units of Taq polymerase (Perkin-Elmer; Norwalk, CT, USA), 200mmol/L each of the four deoxynucleoside triphosphates and 1 mmol/L each of sense primer (nucleotides 1689ŌĆō1708; 5ŌĆ▓-ACCTTGAGGCATACTTCAAA) and antisense primer (nucleotides 2058ŌĆō2078; 5ŌĆ▓-CAGAATAGCTTGCCTGAGTGC) for a DNA fragment of 390 bp, which includes the precore and proximal core regions of the HBV genome. The reaction mixture was overlaid with a drop of mineral oil, heated at 95┬░C for 5 minutes, and amplified with a programmable thermal cycler (Perkin-Elmer). Thirty-five amplification cycles were performed, each consisting of denaturation of template DNA at 95┬░C for 1 min, primer annealing at 55┬░C for 1 minute, and polymerization at 72┬░C for 2 minutes. After the last cycle, the temperature was maintained at 72┬░C for 10 minutes for complete polymerization. Ten microliters of each polymerase chain reaction (PCR) product were separated by electrophoresis in a 2% agarose gel and stained with ethidium bromide to visualize the amplified DNA fragment.
For sera with no detectable HBV DNA by the PCR process described above, DNA was re-extracted from 250 ╬╝l of serum and two PCR amplifications were performed. In the first step, amplification conditions were the same as above; in the second amplification step, 5 ╬╝l of the PCR product were amplified for 30 cycles using the same primers with primer annealing conditions at 60┬░C for 1 minute.
Care was taken to avoid cross-contamination between different steps in the procedure; preparation of the DNA template and preparation of the PCR mixture were conducted in a separate room from the amplification, gel electrophoresis, and purification of PCR products. All samples and solutions were handled with aerosol-resistant pipette tips, and fresh aliquots of reagent solutions were used for each experiment. Positive and negative controls were run with each amplification.
Sequencing of DNA amplified by PCR was performed according to the Sanger dideoxy chain termination method using a commercially available sequencing kit (ŌĆ£fmolŌĆØ; Promega, Madison, WI, USA). The PCR product was purified using a microconcentrator (microcon-100; Amicon, Danvers, MA, USA). Ten pmol of a sequencing primer (nucleotide 1776ŌĆō1797; 5ŌĆ▓-GGCTGTAGGCATAAATTGGTC) for the sense strand of the precore and proximal core regions were end-labeled with 10 pmol of [╬│-32P]-adenosine triphosphate (Amersham, Arlington Heights, IL, USA). The sequencing product was electrophoresed with 6% polyacrylamide/7 M urea gel, dried at 80┬░C for 1 hour with a gel drier, and autoradiographed at ŌłÆ70┬░C for 2ŌĆō24 hours.
Among those cases whose sequencing results were sufficient for analysis, 50/58 (86%) of the HCC patients and 12/16 (69%) of the controls with chronic hepatitis B had an HBV with a mutation at the 83rd nucleotide of the precore region (TGG ŌåÆTAG) that changes codon 28 from tryptophan to a stop codon (Table 3).
Among HCC patients, the precore codon 28 stop mutant was detected in 23/28 (82%) HBeAg-positive patients and 27/30 (90%) HBeAg-negative patients. Among controls with chronic hepatitis B, this mutation was detected in 5/9 (56%) HBeAg-positive controls and 7/7 (100%) HBeAg-negative controls (Table 4).
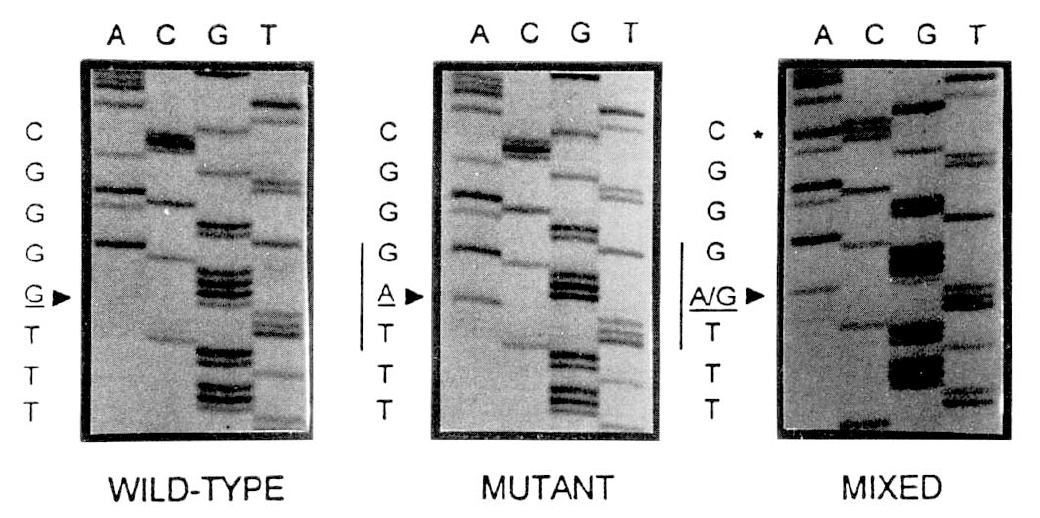
Of the 28 HBeAg-positive HCC patients, 5 (18%) had wild-type HBV alone, 13 (46%) had a mixed infection with both the wild-type and mutant HBV in their serum, since two bands could be detected at the second nucleotide of the precore codon 28 (Fig. 1), and 10 (36%) were infected with the mutant-type alone. Of the 9 HBeAg-positive controls, 4 (44%) had the wild-type alone and 5 (56%) had a mixture of both strains (Table 4).
Of the 30 HBeAg-negative HCC patients, 3 (10%) had wild-type HBV alone, 8 (27%) had a mixed infection with both strains and 19 (63%) were infected with the mutant-type alone. Of the 7 HBeAg-negative controls, 4 (57%) had a mixed infection of both strains and 3 (43%) were infected with the mutant-type alone (Table 4).
Sequencing results from HCC patients were available for 50 of the 61 patients on whom laparoscopic examination was performed. Among those, cirrhosis was observed in 34/43 (79%) patients with the precore codon 28 stop mutant, including 22/24 (92%) with the mutant-type alone, compared to 12/19 (63%) HCC patients with a mixture of wild- and mutant-type HBV (p<0.03) (Table 5).
The 19 anti-HBe-positive HCC patients with mutant HBV alone were older (median age 53 years; range 37ŌĆō64 years), compared to the 17 HBeAg-positive HCC patients with co-infection of both wild-type and mutant HBV (median age 43 years; range 23ŌĆō66 years) (p<0.05). They also had lower serum aspartate aminotransferase levels (82┬▒3IU/L vs. 192┬▒75IU/L) (p<0.03) and lower serum gamma-glutamyl transpeptidase levels (107┬▒2IU/L vs. 214┬▒35IU/L) (p<0.03).
Among HCC patients with HBV-DNA, HBV with the precore codon 28 stop mutation was detected in 82% of HBeAg-positive and 90% of HBeAg-negative patients. Among chronic hepatitis B controls, 56% of the HBeAg-positive cases and 100% of the HBeAg-negative cases had the mutant virus. These results indicate that this mutant virus infection is frequently found both in patients with HBeAg and in patients who are HBeAg-negative.
A mixed infection with both the wild-type and the mutant HBV has been reported in 30ŌĆō79% of HBeAg-positive patients with chronic hepatitis B21,22). Among HBeAg-positive patients in the present study, 46% of the HCC patients and 56% of the chronic hepatitis B controls had such a pattern, but the total prevalence of this mutant virus in the HBeAg-positive HCC patients (alone or with wild-type HBV, 82%) was much greater than that of the controls (56%). These observations suggest that HBV with the precore codon 28 stop mutation may appear in the early stages of HBV infection and may coexist with the wild-type virus prior to the development of HCC, although the actual order of events cannot be determined from the data reported here.
The data in this study suggest the presence of HBV with a stop mutation at precore codon 28 in HCC patients may not be closely correlated with the absence of HBeAg. Among HBeAg-positive HCC patients in the present study, 36% had only the precore codon 28 stop mutant without wild-type HBV; among HBeAg-negative HCC patients, 63% had mutant HBV alone. In contrast, 0/9 HBeAg-positive controls with chronic hepatitis B and 3/7 (43%) HBeAg-negative controls had the mutant type HBV alone.
In the present study, HCC patients with the mutant virus alone (frequently HBeAg-negative) were older in age and had lower serum levels of AST and GGT, compared to patients with a mixed infection of both the wild-type and the mutant HBV (frequently HBeAg-positive). Furthermore, patients with the mutant alone more often had cirrhosis than did patients with mixed infections. These clinical features support the suggestion that the HBV infection evolves from infection with the wild-type virus, to a mixed viral population of wild-type and mutant-type, and eventually to a population consisting of the mutant HBV alone in some patients22). This progression may possibly occur in association with worsening liver damage22), as in the association with cirrhosis in the present study. It is possible that the prolonged presence of this mutant replicative virus could play an active role in carcinogenesis.
Fig.┬Ā1.
Sequencing gels of three serum samples from hepatocellular carcinoma patients. The figure shows three different patterns of HBV-DNA sequences at the precore codon 28 (underlined nucleolides): wild type, mutant type and mixed infection with both wild- and mutant-type HBV.
Table┬Ā1.
Clinical and Laboratory Features of Study Population
| Hepatocellular carcinoma | Chronic Hepatitis B controls | |
|---|---|---|
| No. | 73 | 19 |
| Age(in years) | 51 (18 to 66) | 39 (15 to 52) |
| Male | 63 (86%) | 15 (79%) |
| Female | 10 (14%) | 4 (21%) |
| HBeAg-positive | 29 (40%) | 9 (47%) |
| HBeAg-negative | 44 (60%) | 10 (53%) |
| Hemoglobin (g/dl) | 13┬▒2 | 14┬▒1.2 |
| Wbc (/mm3) | 6800┬▒3300 | 5400┬▒1000 |
| Platelet (/mm3) | 171,000┬▒105,000 | 178,000┬▒50,000 |
| Albumin (g/dl) | 3.7┬▒0.5 | 4.0┬▒0.3 |
| Aspartate aminotransferase (IU/L) | 114┬▒98 | 158┬▒130 |
| Alanine aminotransferase (IU/L) | 85┬▒96 | 237┬▒158 |
| Total bilirubin (mg/dl; normal<1.2) | 1.3┬▒1.2 | 0.8┬▒0.3 |
| Alkaline phosphatase (IU/L; normal<250)) | 442┬▒277 | 296┬▒154 |
| Gamma-glutamyl transpeptidase (IU/L; normal├ż60) | 151┬▒107 | N/A |
| Alpha-fetoprotein (ng/ml; normal<20) | 641┬▒528 | 39┬▒25 |
| Cirrhosis | 46/61 (75%)ŌĆĀ | None |
Table┬Ā2.
The Prevalence of HBV-DNA Detected by Polymerase Chain Reaction
| Diagnosis | HBeAg | No. of Cases |
HBV-DNA detected by PCR
|
|
|---|---|---|---|---|
| No. | (%) | |||
| Hepatocellular carcinoma | + | 29 | 29 | (100) |
| ŌłÆ | 44 | 35 | (80) | |
| Chronic hepatitis B controls | + | 9 | 9 | (100) |
| ŌłÆ | 10 | 10 | (100) | |
Table┬Ā3.
The Prevalence of a Mutant-Type Hepatitis B Virus with a Stop Codon at Precore Codon 28 (TGGŌåÆTAG)
| Diagnosis | HBeAg | No. of Cases | Mutant-type HBV | |
|---|---|---|---|---|
| No. | (%) | |||
| Hepatocellular carcinoma (n=58)a | + | 28 | 23 | (82) |
| ŌłÆ | 30 | 27 | (90) | |
| Chronic hepatitis B controls (n=16)a | + | 9 | 5 | (56) |
| ŌłÆ | 7 | 7 | (100) | |
Table┬Ā4.
Infection Patterns of Hepatitis B Viruses; Wild-type Alone, Mutant-type Alone and Mixture of Both Wild- and Mutant-type Hepatitis B Viruses
| Diagnosis | HBeAg | No. of Cases | Mutant-typeb | Mixed patternb | Wild-typeb |
|---|---|---|---|---|---|
| No. (%) | No. (%) | No. (%) | |||
| Hepatocellular carcinomaa | + | 28 | 10 (36) | 13 (46) | 5 (18) |
| ŌłÆ | 30 | 19 (63) | 8 (27) | 3 (10) | |
| Chronic Hepatitis B controlsa | + | 9 | 0 | 4 (44) | 5 (56) |
| ŌłÆ | 7 | 3 (10) | 4 (57) | 0 |
Table┬Ā5.
Comparison of the Prevalence of Cirrhosis Visualized at Laparoscopy Between Patients with Hepatocellular Carcinoma Infected by Mutant Hepatitis B Virus with Stop Mutation at Precore Codon 28 and Those with Mixture of Wild- and Mutant-Type Hepatitis B Viruses
| HCC Patients Infected by | No. of Casesa | Total Cases with cirrhosis | |
|---|---|---|---|
| No. | (%) | ||
| Mutant-type HBV alone | 24 | 22 | (92)c |
| Mixture of wild- and mutant-type 6 | 19 | 12 | (63)c |
|
|
|||
| Total Number of Cases | 43 | 34 | (79) |
REFERENCES
1. Beasley RP, Hwang LY, Lin CC, Chien CS. Hepatocellular carcinoma and hepatitis B virus. A prospective study of 22,707 men in Taiwan. Lancet 1981;ii:1129ŌĆō1133.

2. Tabor E. Strongly supported features of the association between hepatitis B virus and hepatocellular carcinoma. In: Tabor E, Di Bisceglie AM, Purcell RH, eds. Etiology, Pathology, and Treatment of Hepatocellular Carcinoma in North America. Portfolio, The Woodlands, TX: 1991;107ŌĆō117.
3. Br├®chot C, Pourcel C, Louise A, Rain B, Tiollais P. Presence of integrated hepatitis B virus DNA sequences in cellular DNA in human hepatocellular carcinoma. Nature 1980;286:533ŌĆō535.


4. Shafritz DA, Shouval D, Sherman HI, Hadziyannis SJ, Kew MC. Integration of hepatitis B virus DNA into the genome of liver cells in chronic liver disease and hepatocellular carcinoma: Studies in percutaneous liver biopsies and post mortem tissue specimens. N Engl J Med 1981;305:1067ŌĆō1073.


5. Twu JS, Schloemer RH. Transcriptional trans-activating function of hepatitis B virus. J Virol 1987;61:3448ŌĆō3453.



6. Zahm P, Hofschneider PH, Koshy R. The hepatitis B virus X-ORF encodes a transactivator: a potential factor in viral hepatocarcinogenesis. Oncogene 1988;3:169ŌĆō177.

7. Kim C-M, Koike K, Saito I, Miyamura T, Jay G. HBx gene of hepatitis B virus induces liver cancer in transgenic mice. Nature 1991;351:317ŌĆō320.


8. Kekut├® A, Lauer U, Meyer M, Caselmann W, Hofschneider P, Koshy R. The preS2/S region of integrated hepatitis B virus encodes a transcriptional transactivator. Nature 1990;343:457ŌĆō461.


9. Natoli G, Avantaggiati M, Balsano C, De Marzio E, Collepardo D, Elfassi E, Levrero M. Characterization of the hepatitis B virus preS/S region encoded transcriptional transactivator. Virology 1992;187:663ŌĆō670.


10. Yaginuma K, Kobayashi H, Kobayashi M, Morishima T, Matsuyama K, Koike K. Multiple integration site of hepatitis B virus DNA in hepatocellular carcinoma and chronic active hepatitis tissues from children. J Virol 1987;61:1808ŌĆō1813.



11. Nagaya T, Nakamura T, Tokino T, Tsurimoto T, Imai M, Mayumi T, Kamino K, Yamamura K, Matsubara K. The mode of hepatitis B virus DNA integration into chromosomes of human hepatocellular carcinoma. Genes Dev 1987;1:773ŌĆō782.


12. Ganem D, Varmus H. The molecular biology of the hepatitis B viruses. Annu Rev Biochem 1987;56:651ŌĆō694.


13. Carman WF, Jacyna MR, Hadziyannis S, Karayiannis P, McGarvey MJ, Makris A, Thomas HC. Mutation preventing formation of hepatitis B antigen in patients with chronic hepatitis B infection. Lancet 1989;ii:588ŌĆō591.

14. Omata M, Ehata T, Yokosuka O, Hosoda K, Ohto M. Mutations in the precore region of hepatitis B virus DNA in patients with fulminant and severe hepatitis. N Engl J Med 1991;324:1699ŌĆō1704.


15. Kosaka Y, Takase K, Kojima M, Shimizu M, Inoue K, Yoshiba M, Tanaka S, Akahane Y, Okamoto H, Tsuda F, Miyakawa Y, Mayumi M. Fulminant hepatitis B: induction by hepatitis B virus mutants defective in the precore region and incapable of encoding e antigen. Gastroenterology 1991;100:1087ŌĆō1094.


16. Santantonio T, Jung M-C, Miska S, Pastore G, Pape GR, Will H. Prevalence and type of pre-C HBV mutants in anti-HBe positive carriers with chronic liver disease in a highly endemic area. Virology 1991;183:840ŌĆō844.


17. Tong S, Diot C, Gripon P, Li J, Vitritski L, Tr├®po C, Guguen-Guillouzo C. In vitro replication competence of a cloned hepatitis B virus variant with a nonsense mutation in the distal pre-C region. Virology 1991;181:733ŌĆō737.


18. Liang TJ, Hasegawa K, Rimon N, Wands JR, Ben-Porath E. A hepatitis B virus mutant associated with an epidemic of fulminant hepatitis. N Engl J Med 1991;324:1705ŌĆō1709.


19. Raimondo G, Campo S, Smedile V, Rodino G, Sardo MA, Brancatelli S, Villari D, Pemice M, Longo G, Squadrito G. Hepatitis B virus variant, with a deletion in the preS2 and two translational stop codons in the precore regions, in a patient with hepatocellular carcinoma. J Hepatol 1991;13(Suppl 4):S74ŌĆōS77.


20. Manzin A, Menzo S, Bagnarelli P, Varaldo P, Bearzi I, Carloni G, Galibert F, Clementi M. Sequence analysis of the hepatitis B virus pre-C region in hepatocellular carcinoma (HCC) and nontumoral liver tissues from HCC patients. Virology 1992;188:890ŌĆō895.


-
METRICS

- Related articles
-
Immune thrombocytopenic purpura in a patient with renal cell carcinoma2014 September;29(5)
Dermatomyositis associated with hepatitis B virus-related hepatocellular carcinoma2014 March;29(2)
Long-Term Survival in a Patient With Ruptured Hepatocellular Carcinoma2009 March;24(1)
EGFR and KRAS Mutations in Patients With Adenocarcinoma of the Lung2009 March;24(1)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


