INTRODUCTION
The regeneration of hepatocyte is cellular proliferation occurred after injury induced by chemicals or viruses, and after partial hepatectomy. The pattern and degree of the regeneration in various forms of hepatitis B may be different according to a variety of biological stimuli. Especially, the lesion with poor regenerative activity in itself may show different expression compared with that of other types of hepatitis. However, little data are available. Severe chronic hepatitis is characterized by degeneration of hepatocytes that assume an acinar arrangement and are associated with perihepatocellular inflammation progressing to conspicuous fibrosis and eventual collapse1). They are usually associated with transition to cirrhosis and also have the worst prognosis among the conventional types of chronic hepatitis1). According to the author’s follow-up study1, 2), such severe lobular changes occur following severe confluent necrosis with poor regeneration, as in severe chronic hepatitis3). Hepatic rosettes are formed from proliferation of an hepatocyte surrounded by perihepatocellular fibrosis. Acinus-forming hepatocytes (AFH) show almost hydrophic swelling and are thought to be in the stage of regenerated or degenerated process4). Proliferating cell nuclear antigen (PCNA) has been proven to be an auxiliary protein of DNA polymerase delta, which increases in late G1 and through the S-phase of the cell cycle15). Recently, nuclear APE2 revealed that it was partly associated with proliferating cell nuclear antigen16). It plays a critical role in the initiation of cell proliferation. It was confirmed by immunohistochemical staining that PCNA could be used as a reliable marker of proliferating hepatocytes and hepatocellular carcinoma cells. Electron microscopic study can be used to clarify whether the hepatocytes in chronic hepatitis B are in regenerative or degenerative process. The authors studied immunohistochemically and electron microscopically to document the proliferative and the morphologic features of hepatocytes in various forms of hepatitis B, especially the acinus-forming hepatocytes (AFH) which are believed to be a state of impaired regeneration.
MATERIALS AND METHODS
Study population
The liver specimens employed in this study were obtained from 83 patients with liver disease diagnosed by clinical tests and histologic examination who were admitted to St. Mary’s Hospital from 1986 to 1994. They consisted of eleven patients with acute viral hepatitis, twenty four patients with mild chronic hepatitis, thirty four with severe chronic hepatitis with early cirrhosis and fourteen with severe chronic hepatitis. The patients composed of 67 males and 16 females with median age of 35.4 years (range; 15 to 69 years).
Histologic diagnosis
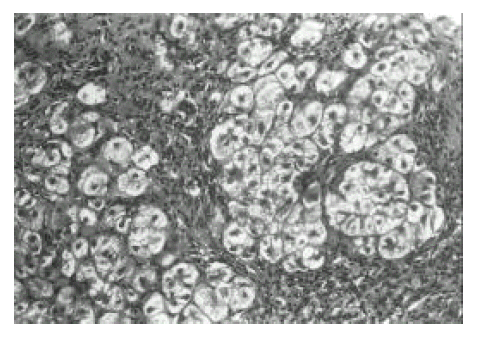
The patients were all HBs Ag positive by RIA (Abbott Lab, III, USA). The lesion with diffuse spotty necrosis in liver parenchyme were classified to acute viral hepatits and mild chronic hepatitis by histologic examination. Acute viral hepatitis was defined to those cases clinically and biochemically recovered within six months after illness and mild chronic hepatis to those persisted over six months. Histologically acute viral hepatits showed more pleomorphism of liver cells, lobular disarray and spotty necrosis with degenerative and regenerative features in the lobules than those in mild chronic hepatitis. Mild chronic hepatitis showed rather prominent sinusoidal cell activation than in acute viral hepatitis. Severe chronic hepatits with early cirrhosis, referred to as chronic active hepatitis with increased inflammatory fibrosis and regenerative nodules, was classified by conventional method. Severe chronic hepatitis was defined as a perihepatocellular fibrosis acinar arrangement of hepatocytes and localized parenchymal necroinflammation in a lobular multilobules1, 2). Severe chronic hepatitis contained AFH in almost 30–100% of liver specimens examined.
Immunohistochemical staining for PCNA-LI
The liver specimens were fixed in 10% formaldehyde and embedded in paraffin. Then 5μ-thick sections were prepared from the paraffin blocks. After deparaffinization through graded ethanol, the sections were washed in tris buffered saline (TBS). For the immunohistochemical staining for PCNA, the streptavidin-biotin immunoperoxidase (ABC method) using commercially available PCNA Kit (Novocastra Lab, U.K.) was performed7, 8). After deparaffinization, the slides were incubated in 1.5% hydrogen peroxide in methyl alcohol for 10 minutes to inhibit endogenous tissue enzyme activity, then rinsed with PBS five times for 5 minutes each time and normal rabbit serum was dropped on tissue sections. The sections were treated with about 100 μL of mouse monoclonal antibody at room temperature for 1 hour, rinsed with TBS (pH 7.6) two times for 5 minutes. One hundred μL biotinylated rabbit anti-mouse IgG antibody (1:500 in TBS) were dropped on the tissue sections and allowed to react at room temperature for 30 minutes. After rinsing with TBS two times for 5 minutes, the tissue sections were incubated with avidin-biotin-peroxide complex for 30 minutes. Peroxidase activity was demonstrated with 0.025% 3.3′-diaminobenzidine tetrahydrochloride (DAB) and 0.01% H2O2 in TBS, pH 7.6, as the substrate. The reaction time was 3 to 5 minutes. Then the sections were lightly counterstained with hematoxylin, dehydrated in ethanol and mounted with mounting medium. Cells positively stained for PCNA were stained brown. The nuclei were scored as positive or negative without regard for staining intensity or location within the hepatic lobule. The expression rate of hepatocytes positive for PCNA was calculated as positive percent per number of total cases. Nuclear labelling indices for PCNA (positive nuclei / total number of counted nuclei) were determined by random evaluation of at least 600 hepatocyte nuclei.
Transmission electron microscopy
The liver biopsy specimens were cut into small pieces, each about 1 mm3 dimension, and were immediately fixed in 2% phosphate-buffered glutaraldehyde solution (pH 7.4) for 2 hours and later post-fixed in 1% buffered osmium tetroxide for 2 hours at 4°C. After processing through a graded series of ethanol and propylene oxide, the tissue was embedded in Epon. Semithin (1 μm) section in thickness was cut and stained with uranyl acetate and lead citrate and examined under a JEOL-100 electron microscope.
RESULTS
Immunohistochemical staining for PCNA-LI
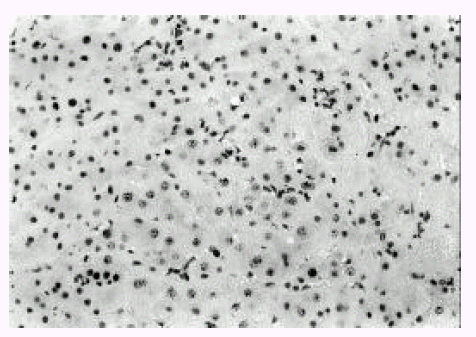
Proliferating cell nuclear antigen was detected in the nucleus, which was stained in dark brown and exhibited a granular or uniform pattern. The positive expression rate for PCNA was 27.3% in acute viral hepatitis, 62.5% in mild chronic hepatits and 47.1% in severe chronic hepatitis with early cirrhosis, respectively. The labelling index of PCNA was 5.3±0.9% in acute viral hepatitis, 22.9±31.7% in mild chronic hepatitis and 14.1±24.2% in severe chronic hepatitis with early cirrhosis but, in the specimens obtained from fourteen patients with severe chronic hepatitis, no expression was seen for PCNA (all nuclei stained green) in liver parenchyme as well as AFH (Figure 1). In some biopsy specimens obtained from patients with acute viral hepatitis, two cell thick plates showing weak PCNA immunoreactive nucleus were occasionally observed. In contrast with acute viral hepatitis, in specimens from patients with mild chronic hepatitis, hepatocytes were prominently arranged in multiple thick plates (Figure 2) and also showed strong positive immunoreactivity for PCNA (Figure 4). The PCNA expression rate and labelling index in mild chronic hepatitis tended to increase remarkably compared with those of acute viral hepatits (Figure 1). In specimens from patients with severe chronic hepatits with early cirrhosis, there was positivity for PCNA in multiple thick plates or early stage of regenerative nodules. The expression rate and labelling index of severe chronic hepatitis with early cirrhosis were in the midst of three groups. However, there was no PCNA expression in fully developed regenerative nodule without sinusoidal activation. Two patients with fulminant hepatitis on first liver biopsy showed severe chronic hepatitis on follow-up biopsy three months later. In the specimens obtained from two patients with fulminant hepatitis, acute massive necrosis of parenchyme without pleomorphism of hepatocytes was observed. The remaining hepatocytes were nearly necrotic. There was no evidence of regenerating process of hepatocytes, such as thick cell plates. Liver specimens obtained three months after first biopsy showed severe chronic hepatitis with formation of acinus. Serial biopsy confirmed that severe chronic hepatitis was originated from acute flare-up of necroinfammation and massive parenchymal necrosis. In the specimens obtained from patients with severe chronic hepatitis after repeated acinar hepatic necrosis, severe chronic hepatitis was mainly located in zone 1 and/or zone 3 of Rappaport. Severe chronic hepatitis is approximated to portal area and separated from uninvolved intralobular parenchyme showing sharp border. Another case of severe chronic hepatitis showed hepatocytes attached to central vein. Collapsed fibrotic thickening was noted around the central vein. Lesion of severe chronic hepatitis near central vein was separated from uninvolved intralobular parenchyme by irregular border. These AFH showed no positive immunostaining for PCNA.
Transmission electron microscopy
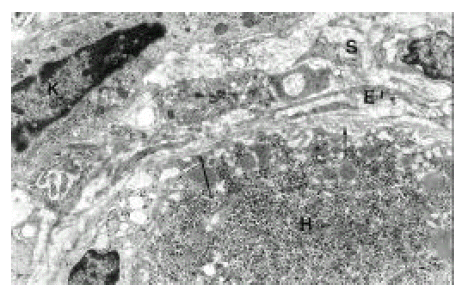
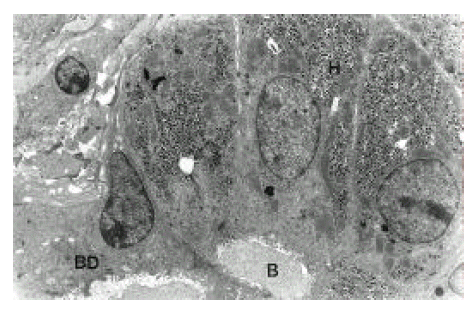
Electron microscopy focused on ultrastructural changes of hepatic acinus in the liver of severe chronic hepatitis. Hepatocytes forming a rosette showed marked degenerative process in the stage of necrosis. The nucleus has pseudoinclusion and irregular margin and was somewhat shrunken. Markedly dilated rough endoplasmic reticulum and dense mitochondria were seen. Bile canaliculus is also dilated (Figure 7, 8). In necrotic area near the hepatic acinus, mesenchymal cell reactions such as an increase of collagen fibers, fibroblast, hepatic stellate cells, angiogenesis and proliferation of bile ductules were observed (Figure 6).
AFH in another area of severe chronic hepatitis appeared to be markedly dense and shrunken, and its cytoplasmic organelles were hardly identified. Bile canaliculi in the center are widened and had prominent microvilli in the lumen. Double layered, basement membrane-like material was running along hepatocytic membrane (capillarization of the sinusoid) which showed complete loss of microvilli (Figure 9). These findings may suggest degenerative process with poor proliferative activity rather than regeneration.
DISCUSSION
The most striking morphologic features in viral hepatitis B are hepatocyte necrosis and regeneration, accompanied by lymphocyte and histiocytic inflammation. Among them, proliferative activity of hepatocyte reflects regenerative state of hepatocytes. In a decade, PCNA immunohistochemical methods have been developed to identify proliferating cells7, 8). Hepatocytes nuclei with positive immunostaining for PCNA means a state of hepatic regeneration. In order to elucidate the proliferative state of hepatocytes in various pathologic conditions by hepatitis B virus infection, PCNA was tested by means of immunohistochemical staining using PCNA commercial kit. Liver regeneration induced by massive hepatic necrosis is associated with proliferative activity of hepatocytes. PCNA was immunohistochemically stained for use as an indicator of proliferative activity of cancer cells11, 16). Our labelling index in acute hepatitis was 5.2%, while 43.0% in acute hepatitis by murine monoclonal anti-PCNA IgM Ab. The labelling index in chronic active hepatitis was 4.2%, 1.96% in active cirrhosis10). But our labelling index was 22.9% in mild chronic hepatitis, 14.1% in severe chronic hepatitis with early cirrhosis. These results are partly because of diagnostic criteria for patient selection and sensitivity of PCNA test. The positive immunostaining for PCNA was noted in massive hepatic necrosis. But we could not find the positive immunostaining for PCNA near necrotic foci of hepatocytes in acute hepatitis. The PCNA immunoreactive cells frequently appeared in near regenerating nodules, especially fibrotic area, in liver cirrhosis. But we could not find any PCNA immunoreactive cells in fully developed regenerating nodules except immature regenerating nodules. These results may suggest that our PCNA method is not sensitive. We suggest that PCNA can be used as a differentiation marker between severe chronic hepatitis and mild chronic hepatitis because of remarkable changes in PCNA-LI. The characteristic findings of severe chronic hepatitis is AFH which suggest massive hepatic necrosis and deficiency of regenerating activity. Lesions of severe chronic hepatitis located near central vein were separated from uninvolved intralobular parenchyme by irregular border. Hepatocyte forming a rosette showed double layered, basement membranelike material was running along hepatocytic membrane in electron microscopy. These findings may suggest degenerative process with poor proliferative activity rather than regeneration. The AFH had a regenerative activity in severe chronic hepatitis, but there was no PCNA expression. We speculated that it was due to resting stage of mitosis, incomplete blood supply by fibrous septa. The PCNA expression rate is high in zone 1, but low in zone 3 by Rappaport classification7). PCNA expression was usually seen in many cell thick plates. The plates were characterized by a “cobble stone” hepatocellular change in continuous regeneration. Therefore, expression rate and labelling index for PCNA was predominantly higher in patients with mild chronic hepatitis than acute viral hepatitis, while the intralobular pathologic features are similar to each other. By contrast, no detectable PCNA expression was noted at all in acinar hepatocytes and the remaining nonacinar cells of severe chronic hepatitis. These are probably due to longer duration of disease and continued regeneration in mild chronic hepatitis than acute viral hepatitis. In our follow-up study, severe chronic hepatitis containing AFH was developed from severe hepatic necrosis, including massive necrosis which is considered to be a state of impairied regeneration. Accordingly, regeneration of AFH seems to be impaired. On the other hand, degeneration is progressive. Therefore, severe chronic hepatitis is a distinct entity not only in the clinical and morphological points of view, but also in the biological growing process. AFH in severe chronic hepatitis was poor in proliferating activity in comparison with the hepatocytic cells of other types of hepatitis B. The false proliferative activity of AFH may be influenced by the lack in regenerating activity of precursor lesion undergoing massive hepatic cell necrosis which is considered to be a state of impaired regeneration. Electron microscopic study confirmed that hepatocytes forming a rosette showed marked degenerative process in the stage of necrosis. The nucleus has pseudoinclusion and irregular margin and was somewhat shrunken. Markedly dilated rough endoplasmic reticulum and dense mitochondria were seen. Bile canaliculus is also dilated (Figure 6). In necrotic area near the hepatic acinus, mesenchymal cell reactions, such as an increase of collagen fibers, fibroblast, hepatic stellate cells, angiogenesis and proliferation of bile ductules were also observed (Figure 7). AFH in another area of severe chronic hepatitis appeared to be markedly dense and shrunken and its cytoplasmic organelles were hardly identified. Bile canaliculus in the center are widened and had prominent microvilli in the lumen. Double layered, basement membrane-like material was running along hepatocytic membrane (capillarization of the sinusoid) which showed complete loss of microvilli (Figure 8). These findings may suggest degenerative process with poor proliferative activity rather than regeneration. Acinar hepatocyte in severe chronic hepatitis originate from an episodic acute bout of massive parenchymal necrosis which is considered to be a state of impaired regeneration. Our results suggest that AHF in severe chronic hepatitis is associated with alteration in cell cycle-related proteins and that the expression of those proteins is responsible for hepatocyte regeneration in damaged liver and may be involved in liver cirrhosis14). In conclusion, our results suggested that PCNA-LI reflects the liver functional reserve and has prognostic significance for patient survival in chronic hepatitis15). Future studies, including many enrolled cases, will be needed for certifying our results.








 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





