 |
 |
| Korean J Intern Med > Volume 34(2); 2019 > Article |
|
Abstract
Background/Aims
Morphological changes due to lung disease in patients with cystic fibrosis (CF) were evaluated using high resolution computed tomography (HRCT), and the HRCT scores obtained using the Bhalla scoring system were correlated with those obtained using clinical and laboratory indicators.
Methods
Medical records of 28 children with CF who underwent chest CT in Department of Pediatric Allergy and Immunology, Cukurova University Balcali Hospital between March 2011 and January 2016 were retrospectively reviewed. Demographic data and physical examination, respiratory cultures, pulmonary function tests, and chest HRCT findings were evaluated. Patients were divided into the following two groups according to their forced expiratory volume in the first second (FEV1) values: normal FEV1 (Ōēź 80% of predicted values) and low FEV1 (< 80% of predicted values). Deep throat or sputum cultures were evaluated for the presence of Pseudomonas aeruginosa (PsA) and other bacteria. HRCT scans were scored using the Bhalla scoring system.
Results
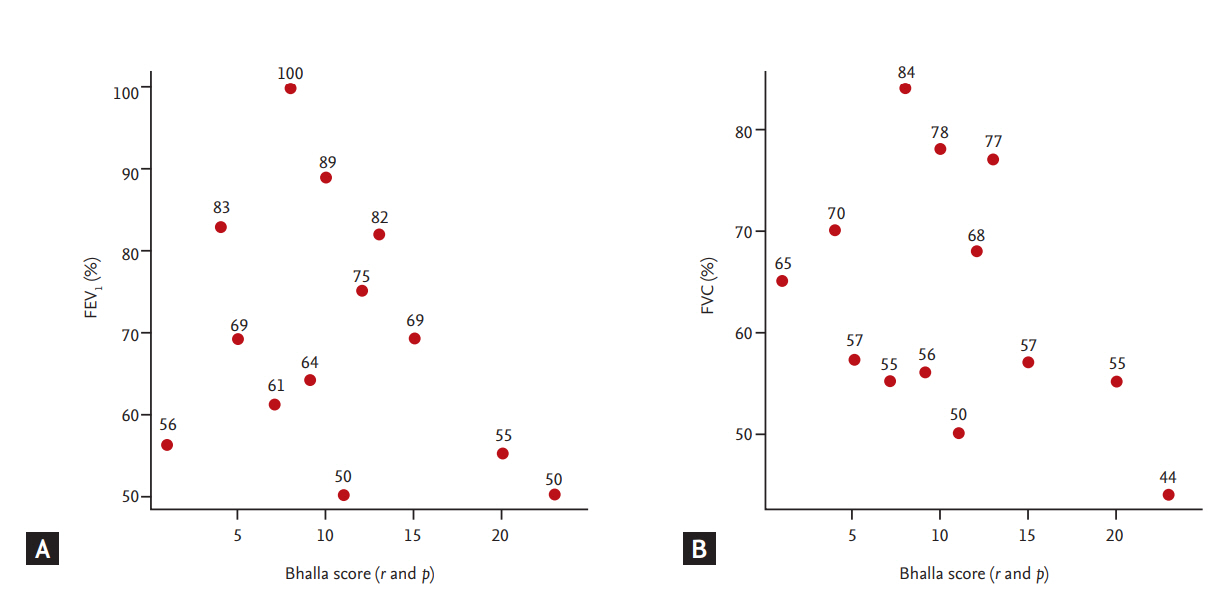
No significant correlation was found between the Bhalla scores and sex, age group, or height percentiles. Significant relationships were found between the Bhalla score and weight (p = 0.036) and body mass index (BMI) (p = 0.032) percentiles below the third percentile, bacterial growth in the sputum/ deep throat cultures (p = 0.009), and presence of PsA (p = 0.004). Moreover, a significant correlation was found between the Bhalla score and FEV1 (r = ŌłÆ0.315, p = 0.0272), forced vital capacity (FVC; r = ŌłÆ0.381, p = 0.0178), forced expiratory flow between 25% and 75% of FVC (r = ŌłÆ0.229, p = 0.0431), and BMI (r = ŌłÆ3.368, p = 0.050).
Cystic fibrosis (CF), caused by a mutation in the cystic fibrosis transmembrane conductance regulator (CFTR) gene, is an inherited, chronic, progressive, and fatal disease [1]. Deletion in CFTR leads to dehydration of the airway surface liquid layer, reduction in mucociliary clearance, and chronic bronchopulmonary infection in the lungs [2]. Detection of early CF lung disease is essential for understanding disease progression and providing endpoints for clinical trials. However, this is difficult because of the lack of sensitive noninvasive outcome measures [3]. Pulmonary function tests (PFTs) and chest radiographs have been used to monitor lung structure in patients with CF. However, PFTs require active participation from patients to perform the required respiratory maneuvers, and forced expiratory volume in the first second (FEV1) has poor sensitivity in evaluating peripheral airway disease [4]. Similarly, chest radiographs are also not sensitive for detecting early disease [5]. High resolution computed tomography (HRCT) is well-established and is the current ŌĆ£gold standardŌĆØ method for monitoring lung anatomical changes in patients with CF [6]. To facilitate evaluation and comparison of quantitative data, investigators have developed quantitative scoring systems based on CT findings. The Bhalla scoring system [7], widely used particularly in pediatric and adult patients with CF, is a scoring system that has been demonstrated to correlate HRCT findings with clinical and physiological characteristics [8].
Studies have reported a good correlation between Bhalla scores and spirometry values in children [3,5]. The aim of this study was to evaluate the morphological changes due to CF lung disease in patients according to HRCT findings and to correlate the HRCT scores obtained using the Bhalla scoring system with clinical and laboratory indicators.
This retrospective study consisted of a review of all the medical records of children with a confirmed diagnosis of CF who were followed up at Department of Pediatric Allergy and Immunology, Cukurova University Balcali Hospital between March 2011 and January 2016. Patients who underwent chest HRCT because of persistent wheezing or collapse and who underwent PFTs within a month of HRCT were included in the study. CF was diagnosed by standard sweat electrolyte testing and chromosomal analysis in all patients [9-11]. CFTR whole-genome sequencing was performed. Clinical data, including demographic characteristics, physical examination findings, age at application, follow-up period, deep throat and sputum cultures, PFTs, and chest HRCT findings, were obtained from the medical records of the patients. Weight, height, and body mass index (BMI) were used for clinical evaluation. The weight and height of the patients were compared with the mean weight and height of healthy Turkish children of the same age and sex [12]. The patients were divided into the following two groups depending on the weight and height percentiles: (1) < 3rd percentile and (2) Ōēź 3rd percentile. Deep throat or sputum cultures were evaluated for the presence of bacteria including Pseudomonas aeruginosa (PsA).
Spirometry was performed within 1 month of HRCT in patients aged Ōēź 5 years. All patients were clinically stable at the time the tests were conducted. FEV1, forced vital capacity (FVC), and forced expiratory flow between 25% and 75% of FVC (FEF25ŌĆō75) were expressed as the percentages of predicted values [13]. The patients were divided into two groups according to their FEV1 value: (1) normal FEV1 (Ōēź 80% of predicted values) and (2) low FEV1 (< 80% of predicted values). Spirometry was performed according to the American Thoracic Society/European Respiratory Society guidelines [14].
HRCT was performed with a TSX-012B scanner (Toshiba, Tokyo, Japan) using a high-resolution technique with 2-mm slice thickness, 120 kVp, and 300 mA at 10-mm intervals, extending from the lung apices to the level of costophrenic angles. All HRCTs were performed by the same radiologist who was unaware of the clinical condition of the patients. If more than one chest HRCT had been performed for a patient, only the last scan was scored.
The Bhalla scoring system is used to evaluate CT findings (Table 1) [7,15]. The scoring system proposed by Bhalla et al. [7] for CT evaluates the following: (1) severity of bronchiectasis, (2) extent of bronchiectasis, (3) peribronchial thickening, (4) extent of mucus plugs, (5) abscesses/sacculation, (6) generalities of the bronchial division involved, (7) number of bubbles, (8) emphysema, and (9) collapse/consolidation. Scores from 0 to 3 were assigned to each of the first seven categories (severity of bronchiectasis, peribronchial thickening, extent of bronchiectasis, extent of mucus plugging, sacculations, generations of bronchi involved, and number of bubbles) and from 0 to 2 for the last two categories (emphysema and collapse/consolidation). The total score has a maximum value of 25. The total score was calculated by adding individual scores for each item, and the scores ranged from 0 (normal) to 25 (severe lung structure abnormalities).
Quantitative variables were expressed as mean ┬▒ standard deviation. Frequencies were used for categorical variables. Correlation of the Bhalla score and its categories with other variables including sex, age at diagnosis, and bacteria/PsA growth in the sputum/deep throat culture was analyzed using the KruskalŌĆōWallis test. SpearmanŌĆÖs rank correlation (r) was used to assess the correlation between the Bhalla scores (and its categories) and spirometry data and BMI. The results were considered significant at a p value of < 0.05.
The Institutional Review Board of Cukurova University Hospital reviewed and approved the study (IRB date: 10.03.2017 file No. 61/49).
The clinical records of 80 patients were reviewed. Of these, 28 patients (nine females) who underwent chest HRCT and PFT within 1 month of the HRCT were included in the study.
The patients underwent HRCT at a median age of 6.8 years (interquartile range [IQR], 0.9 to 17). The mean age at the time of diagnosis was 2 years 4 months (0.08 to 12 years). Twenty children (71.4%) were aged < 2 years at the time of diagnosis. The mean follow-up period was 5.57 years (range, 0.4 to 15.83).
The clinical characteristics of the studied population are presented in Table 2. There was no significant correlation between the Bhalla score and sex, age group, or height percentiles. However, a significant relationship was found between the Bhalla score and weight (p = 0.036) and BMI (p = 0.032). Moreover, a significant correlation was found between the Bhalla score and BMI (r = ŌłÆ3.368, p = 0.050).
For 11 patients (39.3%), deep throat or sputum cultures were done when chest HRCT was performed. PsA, detected in the cultures of seven patients (25%), was the most commonly identified bacteria. Patients with cultures positive for PsA (p = 0.004) or other bacteria (p = 0.009) showed significantly greater Bhalla scores than those whose cultures did not test positive for bacteria.
PFT data were available for 14 patients (50%); the others were too young to undergo PFTs. All 14 children had bronchiectasis, including four with FEV1 > 80% of predicted values; three among them also had normal FEF25ŌĆō75 values. PFT results are presented in Table 2. There were significant correlations between the Bhalla score and FEV1, FVC, and FEF25ŌĆō75 values.
The median age at the time of chest HRCT was 6.07 ┬▒ 4.66 years. The median Bhalla score was 7.5 (IQR, 1 to 24).
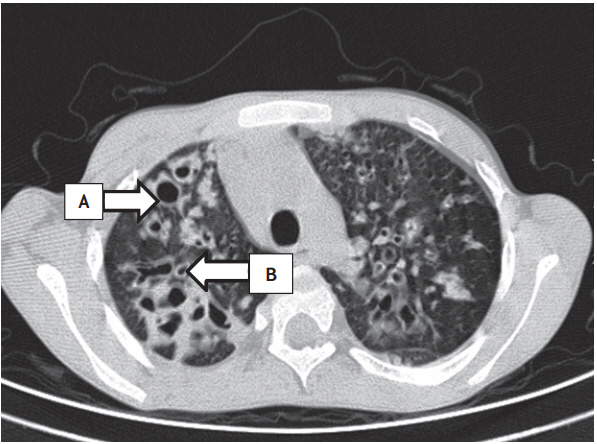
Severe peribronchial thickening, severe bronchiectasis, and sacculations were demonstrated in Fig. 1. Peribronchial thickening (96.4%), bronchiectasis (75%), emphysema (64.3%), mucus plugs (50%), and collapse/consolidation (32.1%) were the most common CT abnormalities in the 28 patients. Sacculations and bullous disease were present in 10.7% and 7.1% patients, respectively. Bronchiectasis was detected in HRCT of 21 patients (75%). Bronchiectasis was mild in 15 patients (53.6%), moderate in three (10.7%), and severe in three (10.7%). Bronchiectasis was found in 1 to 5 segments in seven patients (25%), in 6 to 9 segments in five (17.9%), and in > 9 segments in nine (32.1%). Three patients had sacculations secondary to bronchiectasis involving 1 to 5 segments in one patient and > 9 segments in two.
There was peribronchial thickening in 27 patients (96.4%). Of these, 21 patients (75%) had mild, three (14.3%) had moderate, and two (7.1%) had severe peribronchial thickening.
Mucus plugs were detected in 14 patients (50%), including 1 to 5 segments in five patients (17.9%), 5 to 9 segments in seven (25%), and > 9 segments in two (7.1%). Collapse/consolidation was detected in nine patients (32.1%), including subsegmental collapse in five patients and segmental collapse in four. Emphysema was identified in 18 patients, with 1 to 5 segments in nine patients (32.1%) and > 5 segments in nine (32.1%). Correlations between the Bhalla score and PFTs are shown in Table 3 and Fig. 2. Bhalla sub-scores and their relationship with the presence of PsA are shown in Table 4.
The main cause of mortality in patients with CF is lung disease. For this reason, it is critical to improve pulmonary diagnosis in patients to better predict adverse events and monitor therapies [5]. Currently, different variables, including chest radiography, PFTs, sputum cultures, and clinical examinations, are used to assess pulmonary changes in patients with CF [16]. Spirometry is a fast and noninvasive way to measure airflow obstruction in medium-to-large airways, but is less sensitive to small airway function [17,18]. Spirometry also requires significant respiratory effort and is unsuitable for children aged < 6 years [5]. Chest radiography typically show minimal and nonspecific abnormalities [3], are less sensitive than HRCT for the detection of CF lung disease [19], and are relatively insensitive during pulmonary exacerbations [20]. Chest radiography cannot accurately define bronchiectasis, especially in young patients [21]. For pediatric patients, it is essential to monitor the lungs as early as possible because morphological changes (i.e., mucus plugging and airway inflammation) occur during the first few months of life and are usually not detected via functional measures, e.g., FEV1, even up to 4 years of age [22].
The advent of HRCT has enabled early detection of pathological changes, including mild degrees of bronchiectasis and mucoid impaction [23].
Because of the accepted superiority of CT over chest radiography in the evaluation of pulmonary morphology, many investigators have developed CT scoring systems for patients with CF [7,24-26]. In 1991, two independent publications by Nathanson et al. [27] and Bhalla et al. [7] proposed two distinct scoring systems for the evaluation of CF lung disease in children and adults. In a study comprising 16 patients aged < 12 years, Marchant et al. [21] compared these two scoring systems and showed that the Bhalla method was superior to the Nathanson method in young children with CF. Furthermore, they found a significant correlation between the final Bhalla score and FEV1 (r = ŌłÆ0.65, p = 0.012) and FVC (r = ŌłÆ0.57, p = 0.032). In a study using the Bhalla score, Pereira et al. [28] evaluated CT findings of 23 patients with a mean age of 17.45 ┬▒ 5.75 years. A good correlation was found between the Bhalla score and FVC (r = 0.71, p < 0.001), FEV1 (r = 0.83, p < 0.001), and FEF25ŌĆō75 (r = 0.78, p < 0.001). On the other hand, Helbich et al. [16] evaluated the CT findings of 107 patients with an average age of 14.5 ┬▒ 7.3 years at four different time points. There was a weak correlation (r = 0.25, p < 0.0001) between the CT and PFT results. Consistent with these findings, the present study also identified a weak correlation between the Bhalla scores and FVC (r = ŌłÆ0.381, p < 0.05), FEV1 (r = ŌłÆ0.315, p < 0.05), and FEF25ŌĆō75 (r = ŌłÆ0,229, p < 0.05).
Other studies of HRCT scoring systems have demonstrated the sensitivity of HRCT in detecting CF lung disease. de Jong et al. [29] evaluated two sets of HRCT findings using five scoring systems (Castile, Brody, Helbich, Santamaria, and Bhalla). HRCTs were obtained from children with CF over a period of 2 years. Two simultaneous PFTs were also conducted. A striking finding of this study was that structural abnormalities as scored on HRCT increased significantly, independent of the scoring system, while PFT parameters remained unchanged or even improved. This finding suggests that HRCT is more sensitive than PFTs in the detection of early and progressive lung disease and may be useful in the follow-up of children with CF and as an outcome measure in studies that evaluate therapies being developed to reduce lung damage. Similar results were published in a study by Carpio et al. [5] that evaluated HRCT findings of 31 patients with CF in whom spirometry tests were performed 4 years later. The authors found that Bhalla scoring of the HRCT findings detected the progression of structural lung disease in patients with CF despite stable spirometric findings.
PsA infection is considered a risk factor for the development of bronchiectasis in children with CF [30]. All the patients in this study who were positive for PsA had bronchiectasis. Four patients (50%) had mild bronchiectasis, one (12.5%) had moderate bronchiectasis, and three (37.5%) had severe bronchiectasis. The results of this study also found that the Bhalla scores correlated with respiratory cultures. The presence of bacteria in the deep throat or sputum cultures correlated with the severity of bronchiectasis (p = 0.028) and the extent of mucus plugs (p = 0.013). Martinez-Garcia et al. [31] found that bronchiectasis is related to pathological bacterial colonization, mainly by Pseudomonas species, and that this may be a predictor of exacerbations. These results further support the notion that it may be advantageous to include HRCT in the standard follow-up of patients with CF. Correlation between Bhalla scores and PsA status would reflect clinically meaningful disease progression [32]. Robinson et al. [32] used the modified Bhalla CF HRCT scoring system and found that CF HRCT scores highly correlate with the presence of PsA.
The Cystic Fibrosis Foundation recommends that nutritional status be monitored because optimal nutritional status is associated with better clinical outcomes. Nutritional status may be assessed by weight, height, and BMI. Weight-for-age percentile at 4 years of age has been associated with improved height and growth velocity as well as improved survival and fewer pulmonary exacerbations at 18 years of age [33]. A decrease in the weight-for-height percentage is associated with an increase in mortality rate [18]. Results from this study suggest that BMI is correlated with the Bhalla score and morbidity. According to the Bhalla scores, weight percentile was significantly correlated with the severity of bronchiectasis (p = 0.010), extent of bronchiectasis (p = 0.029), presence of abscess or sacculation (p = 0.028), and collapse/consolidation (p = 0.023). Height percentile was correlated only with the presence of abscess or sacculation (p = 0.018). BMI was correlated with severity of bronchiectasis (p = 0.013), peribronchial thickening (p = 0.001), and sacculations (p = 0.00).
Although the risk of radiation-induced cancer in patients with CF undergoing annual lung CT is < 0.5% [6,34], HRCT results in significant exposure to X-rays. It must be noted that the cumulative radiation dose in multiple chest radiographies may have unknown adverse effects [35,36]. Low-dose HRCT presents a significant reduction in the dose of radiation received by the patient (72% reduction when using 50 mAs and 80% reduction when using 34 mAs) compared HRCT with the usual dose (180 mAs) [37]. Magnetic resonance imaging (MRI) offers the advantage of no ionizing radiation. Intravenous contrast-enhanced lung MRI (IV-CEMRI) with gadolinium-based agents is the most common MRI of the lung [6]. To determine if MRI is sensitive enough to detect early symptoms of CF, Wielputz et al. [38] compared structural and perfusion abnormalities in healthy people (n = 26) and infants and preschool children with CF (n = 50). Abnormal lung structure and perfusion defects were clearly visible in MRI in the CF group [37]. In addition to MRI protocol standardization, there is also a need for MRI scoring systems to detect lung damage in patients with CF. IV-CEMRI has limitations, with the primary issue being insufficient sensitivity because of low proton density in lung air spaces [6]. If protocol standardization, scoring systems, and sensitivity are improved, MRI may someday become a useful tool for measuring loss of lung function in patients with CF.
One of the limitations of this study is the small size and the studyŌĆÖs retrospective design. A prospective and larger study is needed to confirm these results and would indeed help in further understanding CF progression and disease management. Another limitation of the study is the relatively small number of different clinical parameters measured. For instance, the number of exacerbations and quality of life were not evaluated in this study. Future studies are needed to assess the relationships between these variables.
In conclusion, this study showed the usefulness of chest HRCT scoring system in the pulmonary evaluation of children with CF. The usefulness of methods that do not employ ionizing radiation in diagnosing and monitoring lung disease in CF patients should be compared with HRCT. If it is demonstrated that these methods perform as well or better than HRCT, radiation-free scanning methods should be utilized in place of HRCT.
1. High resolution computed tomography (HRCT) is well-established and is the current ŌĆ£gold standardŌĆØ method for monitoring lung anatomical changes in patients with cystic fibrosis (CF).
2. To facilitate evaluation and comparison of quantitative data, investigators have developed quantitative scoring systems based on computed tomography findings.
3. Bhalla chest HRCT scoring system is useful for pulmonary evaluation of children with CF.
Figure┬Ā1.
A is severe bronchiectasis and sacculations. B is peribronchial thickening, in high resolution computed tomography scan.
Figure┬Ā2.
(A) Dot graph for correlation between Bhalla score and forced expiratory volume in the first second (FEV1). (B) Dot graph for correlation between Bhalla score and forced vital capacity (FVC).
Table┬Ā1.
Bhalla score
Table┬Ā2.
Characteristics of the population studied
| Variable | No. (%) | Mean ┬▒ SD | MinŌĆōMax |
|---|---|---|---|
| Female sex | 9 (32) | ||
| Age, yr | 7.83 ┬▒ 4.79 | 0.9ŌĆō17 | |
| Application age, yr | 2.16 ┬▒ 3.61 | 0.08ŌĆō16 | |
| Follow up period, yr | 5.57 ┬▒ 4.28 | 0.4ŌĆō15.83 | |
| Height per | |||
| ŌĆāŌēż 3 | 3 (10.7) | ||
| ŌĆā3ŌĆō10 | 4 (14.3) | ||
| ŌĆā10ŌĆō25 | 6 (21.4) | ||
| ŌĆā25ŌĆō50 | 7 (25) | ||
| ŌĆā50ŌĆō75 | 4 (14.3) | ||
| ŌĆā75ŌĆō90 | 2 (7.1) | ||
| ŌĆā90ŌĆō97 | 2 (7.1) | ||
| Weight per | |||
| ŌĆāŌēż 3 | 2 (7.1) | ||
| ŌĆā3ŌĆō10 | 4 (14.3) | ||
| ŌĆā10ŌĆō25 | 11 (39.3) | ||
| ŌĆā25ŌĆō50 | 5 (17.9) | ||
| ŌĆā50ŌĆō75 | 4 (14.3) | ||
| ŌĆā75ŌĆō90 | 2 (7.1) | ||
| Spirometry | |||
| ŌĆāFEV1, % | 14 (50) | 68 ┬▒ 15.99 | 49ŌĆō100 |
| ŌĆāFEV1 < 80% | 10 (28.5) | ||
| ŌĆāFVC | 14 (50) | 61.79 ┬▒ 12.07 | 44ŌĆō84 |
| ŌĆāFEF 25%ŌĆō75% | 14 (50) | 78.4 ┬▒ 32.73 | 24ŌĆō140 |
| Culture | |||
| ŌĆāPseudomonas aeruginosa | 7 (25) | ||
| ŌĆāOthera | 4 (14.3) | ||
| Genotype | |||
| ŌĆāŌłåF508homozygos | 4 (14.3) | ||
| ŌĆāŌłåF508heterozygos | 4 (14.3) | ||
| ŌĆāOther | 20 (71.4) |
Table┬Ā3.
Correlation between the Bhalla score and pulmonary function tests
| Characteristic |
Bhalla score |
|
|---|---|---|
| r | p value | |
| FVC, % | ŌĆō0.381 | 0.0178 |
| FEV1, % | ŌĆō0.315 | 0.0272 |
| FEF 25%ŌĆō75% | ŌĆō0.229 | 0.0431 |
Table┬Ā4.
Bhalla subscore: presence of Pseudomonas aeruginosa (%)
REFERENCES
1. National Heart, Lung and Blood Institute. Facts about Cystic Fibrosis. Bethesda (MD): National Institutes of Health, 1995.
2. Bradbury NA. CFTR and cystic fibrosis: a need for personalized medicine. In: Hamilton KL, Devor DC, eds. Ion Channels and Transporters of Epithelia in Health and Disease. New York: Springer, 2016;773ŌĆō802.
3. Davis SD, Fordham LA, Brody AS, et al. Computed tomography reflects lower airway inflammation and tracks changes in early cystic fibrosis. Am J Respir Crit Care Med 2007;175:943ŌĆō950.


4. Gappa M, Ranganathan SC, Stocks J. Lung function testing in infants with cystic fibrosis: lessons from the past and future directions. Pediatr Pulmonol 2001;32:228ŌĆō245.


5. Carpio C, Albi G, Rayon-Aledo JC, et al. Changes in structural lung disease in cystic fibrosis children over 4 years as evaluated by high-resolution computed tomography. Eur Radiol 2015;25:3577ŌĆō3585.


6. Kolodziej M, de Veer MJ, Cholewa M, Egan GF, Thompson BR. Lung function imaging methods in cystic fibrosis pulmonary disease. Respir Res 2017;18:96.




7. Bhalla M, Turcios N, Aponte V, et al. Cystic fibrosis: scoring system with thin-section CT. Radiology 1991;179:783ŌĆō788.


8. Judge EP, Dodd JD, Masterson JB, Gallagher CG. Pulmonary abnormalities on high-resolution CT demonstrate more rapid decline than FEV1 in adults with cystic fibrosis. Chest 2006;130:1424ŌĆō1432.


11. Gibson LE, Cooke RE. A test for concentration of electrolytes in sweat in cystic fibrosis of the pancreas utilizing pilocarpine by iontophoresis. Pediatrics 1959;23:545ŌĆō549.



12. Neyzi O, Binyildiz P, Alp H. Pediyatri. Istanbul Tip Fak Mecmuasi 1978;41(Suppl):74.
13. Demirkazik FB, Ariyurek OM, Ozcelik U, Gocmen A, Hassanabad HK, Kiper N. High resolution CT in children with cystic fibrosis: correlation with pulmonary functions and radiographic scores. Eur J Radiol 2001;37:54ŌĆō59.


14. Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J 2005;26:319ŌĆō338.


15. Brody AS, Kosorok MR, Li Z, et al. Reproducibility of a scoring system for computed tomography scanning in cystic fibrosis. J Thorac Imaging 2006;21:14ŌĆō21.


16. Helbich TH, Heinz-Peer G, Eichler I, et al. Cystic fibrosis: CT assessment of lung involvement in children and adults. Radiology 1999;213:537ŌĆō544.


17. Stanojevic S, Bilton D, McDonald A, et al. Global lung function initiative equations improve interpretation of FEV1 decline among patients with cystic fibrosis. Eur Respir J 2015;46:262ŌĆō264.


18. Kerem E, Conway S, Elborn S, Heijerman H, Consensus Committee. Standards of care for patients with cystic fibrosis: a European consensus. J Cyst Fibros 2005;4:7ŌĆō26.

19. Jacobsen LE, Houston CS, Habbick BF, Genereux GP, Howie JL. Cystic fibrosis: a comparison of computed tomography and plain chest radiographs. Can Assoc Radiol J 1986;37:17ŌĆō21.

20. Greene KE, Takasugi JE, Godwin JD, Richardson ML, Burke W, Aitken ML. Radiographic changes in acute exacerbations of cystic fibrosis in adults: a pilot study. AJR Am J Roentgenol 1994;163:557ŌĆō562.


21. Marchant JM, Masel JP, Dickinson FL, Masters IB, Chang AB. Application of chest high-resolution computer tomography in young children with cystic fibrosis. Pediatr Pulmonol 2001;31:24ŌĆō29.


22. Aurora P, Stanojevic S, Wade A, et al. Lung clearance index at 4 years predicts subsequent lung function in children with cystic fibrosis. Am J Respir Crit Care Med 2011;183:752ŌĆō758.


23. Santamaria F, Grillo G, Guidi G, et al. Cystic fibrosis: when should high-resolution computed tomography of the chest be obtained? Pediatrics 1998;101:908ŌĆō913.


24. Stiglbauer R, Schurawitzki H, Eichler I, Vergesslich KA, Gotz M. High resolution CT in children with cystic fibrosis. Acta Radiol 1992;33:548ŌĆō553.


25. Shah RM, Sexauer W, Ostrum BJ, Fiel SB, Friedman AC. High-resolution CT in the acute exacerbation of cystic fibrosis: evaluation of acute findings, reversibility of those findings, and clinical correlation. AJR Am J Roentgenol 1997;169:375ŌĆō380.


26. Maffessanti M, Candusso M, Brizzi F, Piovesana F. Cystic fibrosis in children: HRCT findings and distribution of disease. J Thorac Imaging 1996;11:27ŌĆō38.


27. Nathanson I, Conboy K, Murphy S, Afshani E, Kuhn JP. Ultrafast computerized tomography of the chest in cystic fibrosis: a new scoring system. Pediatr Pulmonol 1991;11:81ŌĆō86.


28. Pereira FF, Ibiapina Cda C, Alvim CG, Camargos PA, Figueiredo R, Pedrosa JF. Correlation between Bhalla score and spirometry in children and adolescents with cystic fibrosis. Rev Assoc Med Bras (1992) 2014;60:216ŌĆō221.



29. de Jong PA, Nakano Y, Lequin MH, et al. Progressive damage on high resolution computed tomography despite stable lung function in cystic fibrosis. Eur Respir J 2004;23:93ŌĆō97.


30. Stick SM, Brennan S, Murray C, et al. Bronchiectasis in infants and preschool children diagnosed with cystic fibrosis after newborn screening. J Pediatr 2009;155:623ŌĆō628.


31. Martinez-Garcia MA, Soler-Cataluna JJ, Donat Sanz Y, et al. Factors associated with bronchiectasis in patients with COPD. Chest 2011;140:1130ŌĆō1137.


32. Robinson TE, Leung AN, Chen X, Moss RB, Emond MJ. Cystic fibrosis HRCT scores correlate strongly with Pseudomonas infection. Pediatr Pulmonol 2009;44:1107ŌĆō1117.


33. Sullivan JS, Mascarenhas MR. Nutrition: prevention and management of nutritional failure in cystic fibrosis. J Cyst Fibros 2017;16 Suppl 2:S87ŌĆōS93.


34. Spycher BD, Lupatsch JE, Zwahlen M, et al. Background ionizing radiation and the risk of childhood cancer: a census-based nationwide cohort study. Environ Health Perspect 2015;123:622ŌĆō628.



35. Kuo W, Ciet P, Tiddens HA, Zhang W, Guillerman RP, van Straten M. Monitoring cystic fibrosis lung disease by computed tomography. Radiation risk in perspective. Am J Respir Crit Care Med 2014;189:1328ŌĆō1336.


36. de Gonzalez AB, Kim KP, Samet JM. Radiation-induced cancer risk from annual computed tomography for patients with cystic fibrosis. Am J Respir Crit Care Med 2007;176:970ŌĆō973.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



