 |
 |
| Korean J Intern Med > Volume 33(4); 2018 > Article |
|
Abstract
Background/Aims
Pulmonary sarcomatoid carcinoma (PSC) is a poorly differentiated non-small cell lung cancer (NSCLC) that contains components of spindle or giant cells. Owing to its low prevalence, there are insufficient data regarding its clinical features, therapeutic strategies and prognosis.
Methods
The medical records of 26 patients diagnosed with PSC from January 2009 to June 2015 were reviewed and analyzed for clinicopathological characteristics, treatment modality, and outcomes.
Results
The median age was 69.5 years. Twenty-three patients (88%) were male. Twenty-four patients (92%) were smokers. The median time from symptom onset to diagnosis was one month. Eighteen patients (69%) were diagnosed at an advanced stage. Pleomorphic carcinoma was the most common subtype, and epidermal growth factor receptor (EGFR) mutation was positive in two of 11 patients. Among 13 patients tested for programmed death ligand 1 (PD-L1) immunohistochemistry assay, eight showed high expression of PD-L1. The median overall survival (OS) of all patients was 9.5 months. In total, 12 patients were treated with chemotherapy: nine with platinum-based doublet therapy, two with tyrosine kinase inhibitor, and one with docetaxel. Seven patients showed partial response or stable disease. The median OS and progression-free survival of patients who received chemotherapy were 8.7 and 2.8 months, respectively.
Pulmonary sarcomatoid carcinoma (PSC) is a group of non-small cell lung cancers (NSCLC) that contain components of spindle or giant cells. According to the 2015 World Health Organization (WHO) classification, it can be classified into five subgroups: pleomorphic carcinoma, spindle cell carcinoma, giant cell carcinoma, carcinosarcoma, and pulmonary blastoma [1]. The prevalence of PSC is very low. Recent population-based studies in the United States revealed that 0.4% of patients with lung cancer had PSC, and in Korea, 2.35% of patients with NSCLC who underwent curative surgery had PSC [2,3]. Because of its rarity, there are limited data regarding its clinical manifestation, treatment, and prognosis of PSC. Unfavorable prognosis is anticipated owing to its pathological feature of poorly differentiated tumors. Some authors reported that PSC patients had worse clinical outcomes than ordinary lung cancer patients [2,4]; however, others found that there was no significant survival difference between PSC and ordinary NSCLC [5,6]. Currently, tyrosine kinase inhibitor (TKI) is recommended in treatment of NSCLC patients who have epidermal growth factor receptor (EGFR) mutation, and novel immune checkpoint inhibitors are also recommended as first-line therapy in selected patients [7]; however, their efficacy has not been well studied in PSC. This study aimed to investigate the clinicopathological characteristics, treatment modalities and responses, and prognostic factors of PSC in Korea.
We identified patients who presented with the disease codes for both malignant neoplasm of the bronchus/lung and sarcomatoid carcinoma (Korean Classification of Disease, 6th edition and International Classification of Disease for Oncology, 3rd edition, respectively) at two academic hospitals from January 2009 to June 2015. Experienced pathologists at each institute reviewed the specimens, verified the diagnoses, and classified them into one of five subgroups in accordance with the 2015 WHO classification: pleomorphic carcinoma, spindle cell carcinoma, giant cell carcinoma, carcinosarcoma, and pulmonary blastoma [1].
We retrospectively collected baseline demographics, macroscopic and microscopic tumor features, initial treatment type, and clinical outcomes from patientsŌĆÖ electronic medical records. Baseline demographics included age, sex, presenting symptoms, smoking profiles, and underlying diseases. The images of radiology and nuclear medicine were reviewed with their formal readings to record macroscopic features of tumor such as stage, involved lobe, and location. We used the definition suggested by Brooks et al. [8] to determine whether the tumor location was central or peripheral. Central tumors were defined as those whose center of mass was within the hilar structures, and peripheral tumors as those whose center of mass was within the parenchyma and with no or minimal contact with hilar structures. Overall survival (OS) was defined as the time from the date of diagnosis to the date of death from any cause, and was calculated for every patient. OS was also calculated according to the primary treatment modality. Progression-free survival (PFS) was defined as the time from the date of beginning of chemotherapy to the date of progression or death from any cause, and was calculated for patients who received chemotherapy. The treatment response of those patients was also analyzed. At the researcherŌĆÖs institutes, assessment of treatment response to chemotherapy follows the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 [9].
Programmed death ligand 1 (PD-L1) expression was examined to measure its prevalence and understand the clinical implications of PSC. In contrast to tests for EGFR mutation that were performed at the attending physicianŌĆÖs discretion, tests for PD-L1 expression had not been conducted at all at the time of diagnosis because their utility was only recently implicated. Among the retrospectively collected PSC patients, those whose archival tissues were available were tested for PD-L1 expression. PD-L1 expression was assessed in formalin-fixed tumor samples with the use of the commercially available PD-L1 IHC22C3 pharmDx assay (Dako North America, Carpinteria, CA, USA) [10]. The degree of PD-L1 expression was presented as tumor proportion score (TPS), which is the percentage of viable tumor cells showing membrane PD-L1 staining relative to all viable tumor cells.
The Mann-Whitney U test and the chi-square test or Fisher exact test were used to analyze the continuous and categorical variables, respectively. The continuous variables were expressed as a median with the interquartile range or mean ┬▒ standard deviation, and the categorical variables were expressed as number (%). The Kaplan-Meier method was used to calculate survival curves. The Cox regression model was used to find variables affecting the survival. Variables with a p value less than 0.1 in univariate analysis were employed in multivariate analysis. A p < 0.05 was considered statistically significant. All statistical analyses were two tailed and performed using SPSS version 20.0 (IBM Co., Armonk, NY, USA).
During the study period, 27 patients were found to have sarcomatoid carcinoma of the lung. Of the 27 patients, one was excluded because the primary tumor site was the skin. Twenty-three patients were male, and three were female. The median age at the diagnosis was 69.5 years. Twenty-four patients were current or former smokers, and the median smoking dose was 35 pack-years. The most common presenting symptoms were respiratory symptoms such as cough, hemoptysis, or dyspnea. Six patients were asymptomatic but presented with abnormal chest radiographs during routine examination. Others had cancer-related pain or constitutional symptoms. Median time from onset of symptoms to diagnosis was 1 month (Table 1).
The primary lesion was located peripherally in 18 patients, and centrally in eight patients. The primary lesion was located more frequently in the upper lobe than in the lower lobe, and more frequently in the right lobe than in the left lobe. The dominant lobe could not be determined in one patient because the primary lesion was consolidation in both upper lobes. Most patients were diagnosed at an advanced stage: 18 patients were stage IV at the time of diagnosis; four were stage III; three were stage II; and one had stage I disease. Subgroup classification of the tumor was conducted in 19 patients. Six patients underwent surgical resection, with three patients showing a subgroup of pleomorphic carcinoma. Specimens from the core needle biopsy were used for subgroup classification in the remaining 16 patients. Six patients showed pleomorphic carcinoma, seven showed spindle cell carcinoma, and two showed giant cell carcinoma. Subgroup was not determined in one patient. EGFR mutation was examined in 11 patients, and two patients were positive for EGFR mutation. PD-L1 expression was tested in 13 patients by using archival tissues. Eight patients (61.5%) showed PD-L1 high expression (TPS Ōēź 50%). Four patients showed low expression (1 Ōēż TPS < 50%), and the other one patient showed no expression (TPS < 1%) (Table 2).
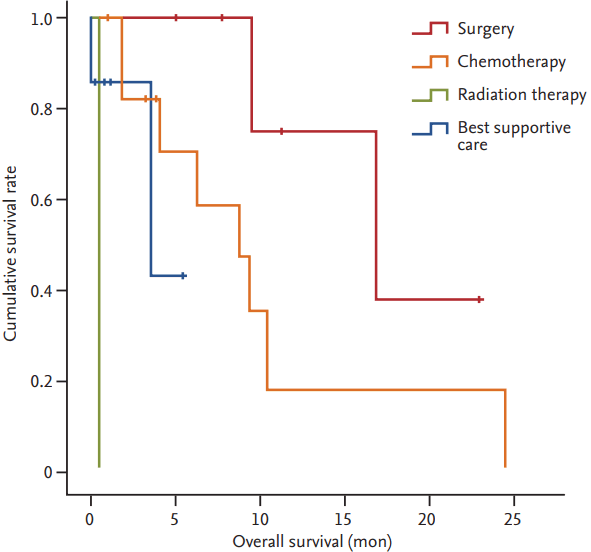
Surgical resection was the primary treatment in six patients. One patient had stage IB disease, three had stage IIB and one had stage IIIA. One patient with stage IV disease had a single metastasis at the left adrenal gland, and was treated with curative right upper lobectomy and adrenalectomy. Radiation therapy was the primary treatment in one patient with stage IV, for palliation of cancer-related pain. Chemotherapy was the primary treatment in 12 patients, all but one of whom had stage IV disease. In the one patient with stage IIIA, there was no surgical operability because the tumor had invaded the pulmonary vein and right atrium. The median OS for all patients was 9.5 months. The median OS of patients with surgical resection was 16.9 months, while chemotherapy was 8.7 months, and best supportive care was 3.6 months (Fig. 1). The median OS of radiation therapy was 13 days; however, it should be kept in mind that radiation therapy was performed in only one patient for palliative purpose (Table 3).
Among the patients who had received chemotherapy, the median PFS was 2.8 months. Because of limited number, median survival could not be calculated in patients receiving TKI or docetaxel. The mean PFS of platinum-based doublet regimen was 2.8 ┬▒ 2.3 months. In one patient receiving TKI, PFS was 7.3 months. PFS was not available in the patient receiving docetaxel. The mean OS of platinum-based doublet regimen, TKI and docetaxel was 6.2 ┬▒ 3.6, 13.2 ┬▒ 16.1, and 3.3 months, respectively. Although it was not possible to find any statistically meaningful results owing to the small number of cases, patients treated with TKI showed longer survival than patients treated with cytotoxic chemotherapy. Treatment response could be assessed in nine patients. Seven patients showed partial response (PR) or stable disease (Table 4). We conducted Cox regression analyses to find variables associated with PFS and OS. There were no significant variables related to PFS. Age, time from symptom onset to diagnosis, TNM (tumor, node, metastasis) stages and TPS of PD-L1 expression were meaningful (p < 0.1) in univariate Cox regression for OS. Multivariate Cox regression with forward method selection was performed. The odds ratio (OR) of age for OS was 1.106 (95% confidence interval [CI], 1.005 to 1.218). The OR of time from symptom onset to diagnosis was 2.002 (95% CI, 1.089 to 3.681). TNM stage and PD-L1 expression were not significant (Table 5).
In this study, we examined clinical characteristics, macro- and microscopic features, treatment response, and prognostic factors of PSC. With regard to microscopic features, we paid special attention to EGFR mutation and PD-L1 expression because TKI and immune check point inhibitor have recently replaced conventional cytotoxic chemotherapy. In patients tested, the proportions of EGFR mutation and high PD-L1 expression were 18.2% and 61.5%, respectively. Approximately 46% of patients were primarily treated with chemotherapeutic agents, and they showed fair treatment response. Although age and time from symptom onset to diagnosis were statistically significant, their clinical relevance was unclear.
The baseline demographics were similar to the findings from previous studies. PSC was more prevalent in males, smokers, and the elderly [11]. Respiratory symptoms were the most common chief complaint [12]. However, tumors were detected incidentally in about onefourths of our patients, reflecting the popularity and usefulness of routine health examinations.
The primary lesion typically presented in the right lobe and the upper lobe. It is well known that PSC presents more frequently in upper lobes [11]. However, we could identify neither the clinical significance nor any underlying mechanism. Interestingly, none of the patients had a primary lesion in the right middle lobe despite the result of right lobe dominancy. The primary lesion was located peripherally more than twice as frequently as being centrally located. In regards to centricity, there have been mixed results regarding the predominant location of primary lesions. Fishback et al. [13] reported that 60% of lesions were peripherally located. In contrast, Koss et al. [14] reported that 38% of lesions were peripherally located. It is important to note that both studies did not provide the definition of central or peripheral lesion. The proportion of central or peripheral lesion may change per the selected definition used in the particular study.
Pleomorphic carcinoma was the most common subtype, followed by spindle cell carcinoma. Other studies also demonstrated that pleomorphic carcinoma was most common. These results may be due to a pathologic definition of pleomorphic carcinoma that encompasses the widest range of cellular components. For a similar reason, spindle or giant cell carcinomas were rare because they consist almost entirely of spindle cells or giant cells [1,3,13]. Interestingly, spindle cell carcinoma presented in more than one-thirds of patients in our study. In contrast to other studies that analyzed surgical specimens only, our study included needle biopsy specimens. This explains why spindle cell carcinomas were relatively common. All three surgical specimens were pleomorphic carcinoma.
It is well known that PSC has a worse prognosis than does ordinary NSCLC, and this was partly true in our study, but studies present contradictory outcomes for PSC and ordinary NSCLC. Pelosi et al. [5] demonstrated that PFS and OS of pleomorphic carcinoma were not significantly different from those of ordinary NSCLC. Nakajima et al. [6] also reported that there was no statistically significant difference in prognosis between PSC and ordinary NSCLC. However, these results should be interpreted with caution; the former included patients with stage I and pleomorphic carcinoma subtype only; the latter encompassed various stages and subtypes of PSC, but did not provide exact data such as 5-year survival rate or median OS. In our study, the median OS of the patients who underwent surgical resection was about 17 months, which was very similar to the data presented by Martin et al. [15]. These authors compared the survival time of PSC and ordinary NSCLC by propensity score matching, and demonstrated that PSC patients had shortened survival times with more rapid disease recurrence. Previous studies usually focused on patients who had been initially treated with surgical resection. Notably, our study collected more patients who had received palliative chemotherapy as their primary treatment rather than receiving surgery. Outcomes of chemotherapy against PSC is also known to be unfavorable. Vieira et al. [4] demonstrated that 69% of patients showed progressive disease (PD), and only 16.5% of patients showed PR. Median PFS and OS were 2.0 and 6.3 months, respectively. Bae et al. [16] reported that median OS was only 5 months and no patients responded to first-line chemotherapy. In our study, 25% of all patients receiving chemotherapy showed PR, and only 16.7% showed PD. The median PFS and OS were about 2.8 and 8.6 months, respectively. These results were better than findings from previous reports even though median PFS and OS of NSCLC were about 5.0 and 11.0 months in recent studies [17].
The occurrence of EGFR mutation was 18.2% in the patients who had been tested for, and Chang et al. [18] published a similar result in 2011. However, the actual proportion was difficult to estimate in our study because 15 patients had not been tested. Patients with EGFR mutation had been initially treated with EGFR TKI; mean OS in these patients was longer than in patients treated with platinum-based doublet therapy. However, their outcomes were at extreme ends of the spectrum: one died 54 days after diagnosis, and the other, showing PR, died 736 days after diagnosis. Efficacy of EGFR TKI for PSC was described in previous studies with promising results. In one patient harboring EGFR mutation who received TKI as first line treatment, complete response was maintained for 35 months [19]. EGFR TKI was also effective in a patient possessing EGFR mutation whose primary lesion was wild type and recurring [20]. EGFR mutation test should be performed in all PSC patients considering the frequent occurrence of EGFR mutation and the efficacy of EGFR TKIs.
Half of patientsŌĆÖ specimens were available for examination of PD-L1 expression. 61.5% of patients showed high expression based on TPS. Earlier studies demonstrated that PSC had higher expression of PD-L1 than ordinary NSCLC [21,22]. Although the reason why PD-L1 expression is higher in PSC is not understood, some authors hypothesized that epithelial-mesenchymal transition may have a role [23,24], and others suggested associations with K-ras mutation [22,25]. The association between K-ras mutation and PD-L1 expression could be explained by heavier smoking in PSC patients, a conclusion supported by Dix Junqueira Pinto et al. [26], who showed that patients with smoking history were more likely to express PD-L1 than patients who never smoked. However, there was no significant correlation between PD-L1 expression and smoking dose (p = 0.713) or PD-L1 expression and smoking status (p = 0.308) in our study. PD-L1 expression had a positive prognostic value on OS in univariate analysis but was insignificant in multivariate analysis. This result is consistent with other studies regarding PSC [22,23]. The PD-L1 expression seems to have limitations in predicting prognosis of PSC. Despite this, examination of PD-L1 expression is recommended because PSC patients generally show higher PD-L1 expression, and it could affect treatment planning and strategy.
There are some limitations in our study. This study was retrospective and with only a small number of patients. Therefore, our results may not be appropriate for generalization. However, these problems are inevitable to some degree because of the rarity of PSC. A multicenter collaborative project should be undertaken to overcome these issues. Additionally, it should be noted that the shortage of surgical specimens likely contributes to the high proportion of spindle cell carcinoma cases and this presents a risk of bias by distorting the ratio of PSC case components.
PSC is a rare tumor with discouraging prognosis even after complete resection. Moreover, it is known to be refractory to chemotherapy. However, our study suggests that PSC patients had a comparatively fair response to chemotherapy. The prevalence of EGFR mutation was not infrequent, and the outcome of EGFR TKI was noticeable. The introduction of PD-1/PD-L1 pathway blockade and high PD-L1 expression in PSC would provide broader treatment options. Extensive workups of biomarkers on target agents, as well as active treatment with cytotoxic chemotherapy, EGFR TKIs, or immune checkpoint inhibitors are necessary to achieve better results and more data regarding PSC management.
1. Patients who have sarcomatoid carcinoma and good performance status need active treatment strategies because they might have good therapeutic response with cytotoxic chemotherapy.
2. In some sarcomatoid patients, epidermal growth factor receptor (EGFR) mutation is positive and may show good EGFR tyrosine kinase inhibitor response.
3. In some sarcomatoid patients, the expression of programmed death ligand 1 is high and it may response well to immune checkpoint inhibitors.
Figure┬Ā1.
Kaplan-Meier survival curve according to treatment modality. The median overall survival (OS) of patients with surgical resection was 16.9 months, chemotherapy was 8.7 months, and best supportive care was 3.6 months. The median OS of radiation therapy was 13 days.
Table┬Ā1.
Baseline demographics
Table┬Ā2.
Macroscopic and microscopic features
| Variable | No. (%) |
|---|---|
| Macroscopic features | |
| ŌĆāTNM stage | |
| ŌĆāŌĆāIB | 1 (3.8) |
| ŌĆāŌĆāIIB | 3 (11.5) |
| ŌĆāŌĆāIIIA | 2 (7.7) |
| ŌĆāŌĆāIIIB | 2 (7.7) |
| ŌĆāŌĆāIV | 18 (69.2) |
| ŌĆāLocationa | |
| ŌĆāŌĆāLeft lower lobe | 3 (11.5) |
| ŌĆāŌĆāLeft upper lobe | 6 (23.1) |
| ŌĆāŌĆāRight lower lobe | 4 (15.4) |
| ŌĆāŌĆāRight upper lobe | 12 (46.2) |
| ŌĆāŌĆāCentral | 8 (30.8) |
| ŌĆāŌĆāPeripheral | 18 (69.2) |
| Microscopic features | |
| ŌĆāSubgroupb | |
| ŌĆāŌĆāPleomorphic carcinoma | 9 (47.4) |
| ŌĆāŌĆāSpindle cell carcinoma | 7 (36.8) |
| ŌĆāŌĆāGiant cell carcinoma | 2 (10.5) |
| ŌĆāŌĆāCarcinosarcoma | 0 |
| ŌĆāŌĆāPulmonary blastoma | 0 |
| ŌĆāŌĆāUndetermined | 1 (5.3) |
| ŌĆāEGFR mutationc | |
| ŌĆāŌĆāPositive | 2 (18.2) |
| ŌĆāŌĆāNegative | 9 (81.8) |
| ŌĆāPD-L1 expressiond | |
| ŌĆāŌĆāHigh expression (TPS Ōēź 50%) | 8 (61.5) |
| ŌĆāŌĆāLow expression (1 Ōēż TPS < 50%) | 4 (30.8) |
| ŌĆāŌĆāNegative (TPS < 1%) | 1 (7.7) |
Table┬Ā3.
Outcomes of each treatment modality
| Primary treatment | Patients | Overall survival, mon |
|---|---|---|
| Surgical resection | 6 (23.1) | 16.9 (5.9ŌĆō28.0) |
| Chemotherapy | 12 (46.2) | 8.7 (4.3ŌĆō13.0) |
| Radiation therapya | 1 (3.8) | 0.4 |
| Best supportive care | 7 (26.9) | 3.6 (0.0ŌĆō8.7) |
| All patients | 26 (100) | 9.5 (8.1ŌĆō11.0) |
Table┬Ā4.
Detailed matters about chemotherapy
| Characteristic | Value |
|---|---|
| Type of first line chemotherapy | |
| ŌĆāPlatinum-based doublet | 9 (75) |
| ŌĆāTKI | 2 (16.7) |
| ŌĆāDocetaxel | 1 (8.3) |
| Response to chemotherapya | |
| ŌĆāComplete response | 0 |
| ŌĆāPartial response | 3 (25) |
| ŌĆāStable disease | 4 (33.3) |
| ŌĆāProgressive disease | 2 (16.7) |
| ŌĆāAssessment not available | 3 (25) |
| ŌĆāProgression-free survival, monb | 2.8 (1.2ŌĆō4.5) |
Table┬Ā5.
Cox regression for overall survival
REFERENCES
1. Travis WD, Brambilla E, Nicholson AG, et al. The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol 2015;10:1243ŌĆō1260.


2. Yendamuri S, Caty L, Pine M, et al. Outcomes of sarcomatoid carcinoma of the lung: a Surveillance, Epidemiology, and End Results Database analysis. Surgery 2012;152:397ŌĆō402.


3. Park JS, Lee Y, Han J, et al. Clinicopathologic outcomes of curative resection for sarcomatoid carcinoma of the lung. Oncology 2011;81:206ŌĆō213.


4. Vieira T, Girard N, Ung M, et al. Efficacy of first-line chemotherapy in patients with advanced lung sarcomatoid carcinoma. J Thorac Oncol 2013;8:1574ŌĆō1577.


5. Pelosi G, Fraggetta F, Nappi O, et al. Pleomorphic carcinomas of the lung show a selective distribution of gene products involved in cell differentiation, cell cycle control, tumor growth, and tumor cell motility: a clinicopathologic and immunohistochemical study of 31 cases. Am J Surg Pathol 2003;27:1203ŌĆō1215.


6. Nakajima M, Kasai T, Hashimoto H, Iwata Y, Manabe H. Sarcomatoid carcinoma of the lung: a clinicopathologic study of 37 cases. Cancer 1999;86:608ŌĆō616.


7. Reck M, Rodriguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 2016;375:1823ŌĆō1833.


8. Brooks DR, Austin JH, Heelan RT, et al. Influence of type of cigarette on peripheral versus central lung cancer. Cancer Epidemiol Biomarkers Prev 2005;14:576ŌĆō581.


9. Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228ŌĆō247.


10. Roach C, Zhang N, Corigliano E, et al. Development of a companion diagnostic PD-L1 immunohistochemistry assay for pembrolizumab therapy in non-smallcell lung cancer. Appl Immunohistochem Mol Morphol 2016;24:392ŌĆō397.



11. Pelosi G, Sonzogni A, De Pas T, et al. Review article: pulmonary sarcomatoid carcinomas: a practical overview. Int J Surg Pathol 2010;18:103ŌĆō120.


12. Rossi G, Cavazza A, Sturm N, et al. Pulmonary carcinomas with pleomorphic, sarcomatoid, or sarcomatous elements: a clinicopathologic and immunohistochemical study of 75 cases. Am J Surg Pathol 2003;27:311ŌĆō324.


13. Fishback NF, Travis WD, Moran CA, Guinee DG Jr, Mc-Carthy WF, Koss MN. Pleomorphic (spindle/giant cell) carcinoma of the lung. A clinicopathologic correlation of 78 cases. Cancer 1994;73:2936ŌĆō2945.


14. Koss MN, Hochholzer L, Frommelt RA. Carcinosarcomas of the lung: a clinicopathologic study of 66 patients. Am J Surg Pathol 1999;23:1514ŌĆō1526.


15. Martin LW, Correa AM, Ordonez NG, et al. Sarcomatoid carcinoma of the lung: a predictor of poor prognosis. Ann Thorac Surg 2007;84:973ŌĆō980.


16. Bae HM, Min HS, Lee SH, et al. Palliative chemotherapy for pulmonary pleomorphic carcinoma. Lung Cancer 2007;58:112ŌĆō115.


17. Soo RA, Loh M, Mok TS, et al. Ethnic differences in survival outcome in patients with advanced stage non-small cell lung cancer: results of a meta-analysis of randomized controlled trials. J Thorac Oncol 2011;6:1030ŌĆō1038.


18. Chang YL, Wu CT, Shih JY, Lee YC. EGFR and p53 status of pulmonary pleomorphic carcinoma: implications for EGFR tyrosine kinase inhibitors therapy of an aggressive lung malignancy. Ann Surg Oncol 2011;18:2952ŌĆō2960.


19. Tamura Y, Fujiwara Y, Yamamoto N, et al. Retrospective analysis of the efficacy of chemotherapy and molecular targeted therapy for advanced pulmonary pleomorphic carcinoma. BMC Res Notes 2015;8:800.



20. Zou F, Xie G, Ma JA, Zhou DA, Jiang YI, Zheng JY. Epidermal growth factor receptor mutation heterogeneity analysis of pulmonary sarcomatoid carcinoma successfully treated with erlotinib: a case report. Oncol Lett 2015;9:2239ŌĆō2243.



21. Velcheti V, Rimm DL, Schalper KA. Sarcomatoid lung carcinomas show high levels of programmed death ligand-1 (PD-L1). J Thorac Oncol 2013;8:803ŌĆō805.



22. Vieira T, Antoine M, Hamard C, et al. Sarcomatoid lung carcinomas show high levels of programmed death ligand-1 (PD-L1) and strong immune-cell infiltration by TCD3 cells and macrophages. Lung Cancer 2016;98:51ŌĆō58.


23. Kim S, Kim MY, Koh J, et al. Programmed death-1 ligand 1 and 2 are highly expressed in pleomorphic carcinomas of the lung: comparison of sarcomatous and carcinomatous areas. Eur J Cancer 2015;51:2698ŌĆō2707.


24. Shimoji M, Shimizu S, Sato K, et al. Clinical and pathologic features of lung cancer expressing programmed cell death ligand 1 (PD-L1). Lung Cancer 2016;98:69ŌĆō75.


-
METRICS

- Related articles
-
Clinical Characteristics of Pulmonary Embolism with Underlying Malignancy2010 March;25(1)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


