 |
 |
| Korean J Intern Med > Volume 34(2); 2019 > Article |
|
Abstract
Background/Aims
Fibromyalgia (FM) is a common rheumatologic disease characterized by chronic widespread pain, along with various clinical manifestations including atypical autoimmune characteristics. Despite its high prevalence, there remain no approved laboratory tests to identify specific manifestations of FM, or to rule out FM from other rheumatic diseases. Anti-dense fine speckled 70 (anti-DFS70) antibodies were initially identified as a form of anti-nuclear antibodies in a patient with interstitial cystitis. Anti-DFS70 antibodies are found in ≤ 10% of healthy individuals, but have suggestive negative association with autoimmune diseases; however, the clinical significance of these autoantibodies in FM patients remains poorly understood.
Methods
We examined 39 patients with FM, along with 17 patients with systemic lupus erythematosus (SLE), and 19 healthy individuals (HI). Patients were compared based on physical measurements, disease duration, tender point counts, FM Impact Questionnaire (FIQ) scores, visual analog scale (VAS) for pain, somatic symptoms, and anti-DFS70 antibodies.
Results
Levels of anti-DFS70 antibodies were significantly higher in the FM and HI groups than in those with SLE. Both anti-DFS70 antibodies and VAS scores were positively correlated with FM. Within the FM group, patients with arthralgia had higher anti-DFS70 antibody values compared to those without arthralgia (p = 0.024); antibody levels were also higher in patients with sleep disturbances relative to those without sleep issues (p = 0.024). In contrast, there were no correlations between anti-DFS70 antibodies and age, body mass index, disease duration, tender point counts, FIQ, short-form health survey results, or other clinical manifestations.
Conclusions
Anti-DFS70 antibodies may represent a useful biomarker for differentiating between FM and other autoimmune diseases. The levels of anti-DFS70 antibodies were also significantly higher among patients with arthralgia and sleep disturbances. Further investigations are necessary to evaluate the relationships between anti-DFS70 antibodies and other cytokines as a predictive marker for pain.
Fibromyalgia (FM) is not uncommon rheumatologic disease characterized by chronic widespread pain, although the causes underlying this condition remain poorly understood. Recent evidence suggests that malfunctions in the central nervous system resulting in amplification of pain transmission, in combination with environmental factors, may play a role in the etiopathology of FM. Current guidelines for the diagnosis of FM, such as those put forth by the American College of Rheumatology (ACR), are based entirely on clinical features, with no known serological markers available to either confirm or rule out other diseases [1,2]. Variability in the clinical presentation of FM, along with the absence of diagnostic biomarkers, can often result in misdiagnosis, with important consequences on the disease course [3]. Significant overlap is seen between FM symptoms and those of other autoimmune diseases [4]. Among patients with systemic lupus erythematosus (SLE), up to 65% have been shown to meet the ACR criteria for FM [5], which may substantially affect quality of life [6]. Similarly, 57% of patients with rheumatoid arthritis (RA) and 24% of those with psoriatic arthritis exhibit symptoms of FM [7]. Taken together, these studies highlight the difficultly faced by clinicians in trying to differentiate between FM and other autoimmune diseases.
Indirect immunofluorescence (IIF) is one of the most commonly used tests for detection of antinuclear antibody (ANA) on human epithelial type-2 cell (HEp-2) cells, and has recently been recommended as a screening test of autoimmune diseases [8]. In recent years, the IIF assay has been replaced in many laboratories by high throughput and economical screening immunoassays, which incorporate the key autoantibody target antigens into a single assay, on platforms such as enzyme-linked immunosorbent assay (ELISA) and multiplex assays based on addressable laser bead technology [9]. Recognition of nuclear dense fine speckled (DFS) pattern by IIF was initially identified as an atypical ANA pattern in a patient with interstitial cystitis, but have since been linked to a much wider array of conditions [10,11], including chronic inflammatory conditions, cancer, and even certain healthy individuals [10,12,13]. As with other patterns, the typical DFS pattern can vary depending on the manufacturer source of the HEp-2 slides used as substrate [14]. Since a 70-kDa protein was recognized by immunoblotting, the antigen was initially termed dense fine speckled 70 (DFS70) but eventually the primary target autoantigen was identified as the DNA binding transcription coactivator p75 [15]. Anti-DFS70 antibodies can be detected by various technologies including IIF, immunoblot, and ELISA [15].
Although the clinical significance of anti-DFS70 antibodies has been investigated in various autoimmune diseases including RA, SLE, and Sjögren’s syndrome, its prevalence in FM has not been examined [16,17]. Here, we evaluated the clinical significance of anti-DFS70 antibodies in FM, and its association with clinical characteristics.
Enrollment in this study was limited to FM patients over the age of 20 years who visited the rheumatology clinic of a university hospital between August 2008 and January 2011, and were willing to undergo anthropometric measurements and blood tests, and complete a study-related questionnaire, resulting in a final cohort of 39 patients. Two control groups were also included: a disease control group consisting of 17 SLE patients, and a healthy control group consisting of 19 healthy individuals (HI) with no history of rheumatic diseases, diabetes, hypertension, dyslipidemia, stroke, cardiovascular diseases, malignancy, or recent infection. All patients provided written informed consent prior to enrollment. The Chosun University Hospital Ethic Committees of the Institutional Review Board approved the study protocol and all participants provided written informed consent prior to participation in the study (IRB 11 S-197). The study was conducted in accordance with the principles of the current version of the Declaration of Helsinki.
A trained rheumatologist aided by research nurses conducted physical examinations; they assessed disease duration, made tender point counts, applied the visual analog scale (VAS) for pain, completed the Fibromyalgia Impact Questionnaire (FIQ) and short-form 36 (SF-36) health survey, and assessed somatic symptoms through one-on-one interviews. Levels of anti-DFS70 antibodies were assessed using an ELISA method.
Clinical diagnosis of FM was performed in accordance with the 1990 ACR criteria for FM [2].
The patients were weighed standing upright, barefoot, in a thin gown using a body composition analyzer (Zeus 9.9, Jawon Medical Co., Seoul, Korea); height was measured to the nearest 0.1 cm using a manual height analyzer. Body mass index (BMI) was calculated based on these measurements.
Digital palpation with an approximate force of 4 kg was applied to 18 sites on the right and left sides of each patient’s body using the thumb. All measurements were collected by a single physician to eliminate inter-observer bias.
The FIQ was first published in 1991, and has become the industry standard index of disease activity and therapeutic efficacy. We used Korean fibromyalgia impact questionnaire (KFIQ), a well-established adaptation of the original questionnaire culturally adapted for use in Korea [18]. The KFIQ consists of 20 questions that assess the physical ability, mood, ability to work, pain, fatigue, relaxation, stiffness, depression, and anxiety of patients. The questionnaire is scored in the following manner: questions 1 to 11 are directly related to physical function, with ability to complete physical tasks classified as ‘always, mostly, sometimes, or never.’ Question 12 is an assessment of the patient’s general well-being, with the answer consisting of the number of days they felt well. Question 13 is the number of days a patient was unable to work (including housework) because of FM symptoms. Questions 14 to 20 ask patients to rate the severity of disease symptoms including pain, fatigue, anxiety, and depression on a scale of 0 to 10.
Investigators showed patients a 10-cm ruler, which was defined as ranging from no pain to the worst imaginable pain possible on the right. Respondents were asked to mark the point along the ruler that represented their pain intensity. Scores were determined by measuring the distance between the 0 cm and the patient’s mark, providing a range of scores from 0 to 10.
To analyze the strength and significance of anti-DFS70 responses, we compared their levels among the groups (FM, SLE, and HI controls) using ANOVA for parametric method or Kruskal-Wallis test for nonparametric method. For comparison with two different groups, we used post hoc analysis including the Dunnett and Mann-Whitney U test test and Bonferroni correction was performed to solve the multiple testing problems. We compared the differences among antibody levels, patient characteristics such as age, height, weight, and BMI, and clinical outcomes such as disease duration, tender point counts, VAS, KFIQ, and somatic symptoms in FM group.
All data are presented as the median and interquartile range (IQR), which is driven from the distribution of each item, unless otherwise noted. Kruskal-Wallis tests were used to detect differences in anti-DFS70 antibody responses among groups (FM, SLE, and healthy controls). Student t test was performed to assess differences in height, weight, and BMI; Fisher exact test used for sex. Spearman correlation analysis with a Bonferroni correction was used to examine differences in height, weight, BMI, disease duration, tender point counts, KFIQ, somatic symptoms, and VAS. Statistical analyses were performed using SPSS version 14.0 (SPSS Inc., Chicago, IL, USA); R v3.1.2 (https://cran.r-project.org/bin/windows/base/old/3.1.2/) was used to plot data. All tests were two-tailed, and p < 0.05 were considered statistically significant.
A total of 75 patients (39 FM, 17 SLE, and 19 HI controls) were enrolled; demographics of the study population are shown in Table 1. The average age was 51.87 ± 11.36 years in the FM group, 37.24 ± 12.13 years in the SLE group, and 48.63 ± 9.42 years in controls (p < 0.001). There were 36 females (92%) in the FM group, 16 (94%) in the SLE group, and 10 (53%) in the control group (p < 0.001). BMI and disease duration were significantly different between the FM and SLE groups.
Anti-DFS70 antibodies were detected at a level of 6.30 U/mL (IQR, 4.722 to 8.435) in the FM group, 4.148 U/mL (IQR, 3.574 to 6.443) in the SLE group, and 5.152 U/mL (IQR, 4.148 to 7.878) in HI controls. These levels were significantly different between the FM and SLE groups (p = 0.008), but not between the SLE group and HI controls (Fig. 1).
No correlation was seen between KFIQ outcomes and anti-DFS70 antibody status (Spearman correlation coefficient r = 0.182, p = 0.268), or between anti-DFS70 antibody status and SF-36 outcomes (r = 0.264, p = 0.104) within the FM group. However, a positive correlation was seen between anti-DFS70 antibody status and VAS (r = 0.589, p < 0.001) (Table 2).
Within the FM group, patients with arthralgia had higher anti-DFS70 antibody values compared to those without arthralgia (p = 0.024); antibody levels were also higher in patients with sleep disturbances relative to those without sleep issues (p = 0.024). Other symptoms, including dry eye, dry mouth, irritable bowel syndrome, Raynaud’s phenomenon, coldness, numbness, depression, and headache were not significantly associated with antibody values (Table 3).
It is now generally accepted that FM is a complex disorder with most experts recommending a multidimensional diagnostic approach including somatic complaints, psychosocial stressors, and psychological factors [19]. Despite improvements to longstanding classification criteria, the modified version of the ACR 2010 for FM is still unable to completely resolve all controversies regarding the diagnosis of FM, due in part to the lack of a reliable biomarker for positive diagnosis or excluding other diseases [20]. Given the variability among methods used to rate severity of FM symptoms, many studies now focus on developing screening tools to enable a definitive diagnose of FM, along with assessments tools that can better rate the severity of FM [21]. Moreover, differential diagnostic tools, including identification of objective biomarkers, are necessary to definitively exclude other autoimmune diseases.
The screening for ANA and other specific autoantibodies has been the standard of the serologic diagnosis for autoimmune diseases. These connective tissue diseases are also called ANA-associated rheumatic diseases (AARD) because of their strong association with ANA [22]. The diagnosis of SLE, Sjögren’s syndrome and systemic sclerosis is highly unlikely in a negative ANA. However, due to a perceived high prevalence of “false negative or positive” results and other technologies for ANA detection continue to evolve the atypical ANA type. In a recent study, the DFS IIF pattern was found in 33.1% of ANA positive HI compared to 0.0% of ANA positive autoimmune diseases (p < 0.0001) [23].
The typical IIF staining pattern against DFS is recognized as fine, uniformly distributed speckles throughout the interphase nucleus and on metaphase chromatin [10,11,24]. As this autoantibody was first detected as a 70 kDa protein by immunoblotting, the antigen was initially termed DFS70, most commonly known as the primary target autoantigen was identified as lens epithelium- derived growth factor or DNA-binding transcription coactivator p75 [10,25]. The anti-DFS70 antibodies have since been detected in a variety of conditions [10,11]. The highest prevalence of anti-DFS70 antibodies has been reported in patients with Vogt-Koyanagi-Harada syndrome (66.7%) and atopic dermatitis (30%), followed by apparently healthy individuals (≤ 10%), while prevalence among patients with autoimmune diseases is significantly lower (≤ 2% to 3%) [10-13,23-29]. An evaluation of more than 10,000 ANA-positive samples by ELISA and immunoblot found that anti-DFS70 antibodies were common among ANA-positive individuals with no evidence of autoimmune disease [27]. Regarding the prognostic and long-term outcomes of HI with positive anti-DFS70 antibodies, the 0 of 40 developed an autoimmune disease over an average of 4 years of follow-up [23]. Therefore, the presence of isolated anti-DFS70 antibodies may be taken as strong evidence against the diagnosis of an autoimmune disease, such as SLE. In our study, anti-DFS70 antibody levels were significantly higher in the HI group than in SLE patients.
In a previous study, upwards of 14% of FM patients were shown to be positive for ANAs, of which 30% had a history of Raynaud’s phenomenon, and 18% had symptoms of Sjögren’s syndrome such as dry eye and dry mouth, but none of these patients progressed to a classic connective tissue disease [30]. FM patients have also been shown to exhibit the same rate of ANA positivity as that of osteoarthritis patients (8.8% and 8.9%, respectively) [31]. Taken together, these studies show that ANAs look as not a good predictor for the differentiation with other connective tissue diseases. The prevalence of anti-DFS70 antibodies and the specificity of these antibodies in FM patients have not been examined. In our study, anti-DFS70 antibody levels were significantly higher in the FM group than in SLE patients, and these antibodies look helpful for differentiating between FM and autoimmune disease.
FM is sometimes related to arthralgia; however, symptoms of arthralgia in FM patients are different from that of arthralgia seen in association with arthritis. In FM patients, pain often appears to be coming from the joints, with many patients reporting symptoms of joint stiffness. However, when the joints of an FM patient are examined by X-ray or other imaging tools, no such damage is evident. While the bones of FM patients are not affected, the soft tissues play an important role in moving a joint, resulting in pain signals from the surrounding muscles and tendons. This type of arthralgic pain must therefore be treated differently than arthralgia due to arthritis. In some FM patients, medications can be directly injected into a painful, stiff muscle to help ease joint pain. Certain pain medications, including those designed to block nerve pain, and antidepressants, also seem to have a positive effect on FM pain. Nonpharmacological interventions, such as lifestyle changes and cognitive-behavioral therapy, including stretching exercises and massage therapy, have also been shown to ease FM arthralgia symptoms.
Insomnia is a common sleep disorder in FM patients. The sleep abnormalities reported in FM patients mainly include a reduction of total sleep time, particularly a decrease in the percentage of slow-wave sleep, along with increased awakenings or arousal. Studies of the microstructure of sleep in FM patients have shown an anomalous intrusion of the alpha rhythm in the slow delta activity, which characterizes deeper sleep stages; a larger number of oxygen desaturations per hour of sleep have also been observed. Furthermore, the Sleep and Pain Diathesis model suggests that sleeping problems may play an important role in the etiology of FM and the persistence of many disease symptoms. Several studies have observed an inverse relationship between sleep disorders and pain thresholds, with those having trouble sleeping more likely to experience widespread pain, fatigue, and negative mood. Given the nature of these symptoms, numerous psychological treatments have been investigated in FM, with several showing improvements in clinical outcomes [32]. In our study, among FM patients, anti-DFS70 antibody levels were positively correlated with VAS scores and were significantly higher in patients reporting symptoms of arthralgia and sleep disturbances. These results suggest that anti-DFS70 antibodies could be correlated with certain cytokine for predicting pain in FM patients. Therefore, anti-DFS70 antibodies may represent an important diagnostic marker for FM without AARD, and be useful for evaluating pain severity in these patients.
As with all research, this study had several limitations. First, the single-center nature of this study limits the extent to which we can generalize these results to other clinics. Second, the small number of FM patients and SLE patients were evaluated without comparisons to other autoimmune diseases such as RA or Sjögren’s syndrome. Further investigation into the role of anti-DFS70 antibodies in more bigger size of FM are necessary to determine the efficacy of using anti-DFS70 antibody levels to predict the severity of FM.
1. Fibromyalgia (FM) is a common rheumatologic disease characterized by various clinical manifestations including atypical autoimmune characteristics. There is no specific biomarker to diagnose FM.
2. Anti-dense fine speckled 70 (anti-DFS70) antibodies may represent a useful biomarker for differentiating between FM and other autoimmune diseases.
3. The levels of anti-DFS70 antibodies were also significantly higher among patients with arthralgia and sleep disturbances.
Figure 1.
We checked the differences in the levels of anti-dense fine speckled 70 (anti-DFS70) antibodies between three groups by Kruskal-Wallis test. In the pairwise post hoc analysis, we used the Mann-Whitney U test and p value adjusted by Bofferoni correction. FM, fibromyalgia; SLE, systemic lupus erythematosus; HI, health individuals. ap < 0.05.
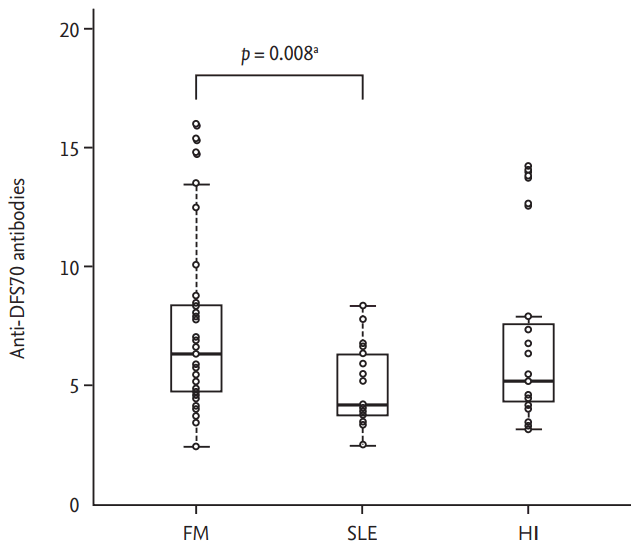
Table 1.
Demographic characteristics of fibromyalgia, systemic lupus erythematosus, and healthy individuals
| Characteristic | Fibromyalgia (n = 39) | Systemic lupus erythematosus (n = 17) | Healthy individuals (n = 19) | p value |
|---|---|---|---|---|
| Female sex | 36 (92)a | 16 (94) | 10 (53) | 0.0009 |
| Age, yr | 51.87 ± 11.36b | 37.24 ± 12.13 | 48.63 ± 9.42 | 0.0007 |
| Body mass index, kg/m2 | 22.52 ± 2.08 | 20.64 ± 2.76 | - | - |
| Disease duration, mon | 32 (30–35)c | 61 (42–144) | - | - |
Table 2.
It shows the Spearman-correlation between antidense fine speckled 70 antibodies and clinical parameters of fibromyalgia
| Variable | r | p value |
|---|---|---|
| Age | 0.294 | 0.859 |
| Body mass index | –0.085 | 0.606 |
| Disease duration | –0.038 | 0.816 |
| Tender point 18 | 0.073 | 0.658 |
| VAS of pain | 0.589 | < 0.001a |
| KFIQ | 0.182 | 0.269 |
| Q1 (physical impairment) | 0.044 | 0.790 |
| Q2 (feel good) | 0.270 | 0.096 |
| Q3 (work missed) | –0.032 | 0.849 |
| Q4 (do work) | 0.188 | 0.252 |
| Q5 (pain) | 0.372 | 0.020 |
| Q6 (fatigue) | 0.332 | 0.039 |
| Q7 (rested) | 0.017 | 0.920 |
| Q8 (feeling stiffness) | 0.181 | 0.271 |
| Q9 (anxiety) | 0.271 | 0.095 |
| Q10 (depression) | 0.175 | 0.287 |
| SF-36 | 0.264 | 0.104 |
Table 3.
It shows the difference between clinical symptoms and anti-dense fine speckled 70 antibody in fibromyalgia group
| Variable | Existent | Nonexistent | p value |
|---|---|---|---|
| Irritable bowel syndrome | 5.08 (3.18–7.83) | 6.86 (4.79–8.44) | 0.117 |
| Raynaud’s phenomenon | 4.72 (4.15–8.45) | 6.72 (4.94–8.40) | 0.255 |
| Coldness | 7.52 (6.30–8.74) | 6.30 (4.72–8.31) | 0.529 |
| Numbness | 6.86 (4.72–7.88) | 6.30 (4.61–8.67) | 0.629 |
| Arthralgia | 7.73 (5.87–13.43) | 5.80 (4.26–7.99) | 0.024a |
| Sleep disturbance | 7.73 (5.58–11.23) | 5.37 (4.33–7.77) | 0.024a |
| Depression | 8.02 (5.44–11.59) | 6.08 (4.65–8.12) | 0.203 |
| Headache | 6.30 (5.44–8.02) | 6.44 (4.47–8.67) | 0.986 |
| Dry eye | 7.73 (5.44–8.31) | 6.30 (4.43–8.66) | 0.414 |
| Dry mouth | 6.08 (4.54–8.02) | 6.59 (4.79–9.38) | 0.414 |
REFERENCES
1. Wolfe F, Clauw DJ, Fitzcharles MA, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res (Hoboken) 2010;62:600–610.


2. Wolfe F, Smythe HA, Yunus MB, et al. The American College of Rheumatology 1990 criteria for the classification of fibromyalgia: report of the Multicenter Criteria Committee. Arthritis Rheum 1990;33:160–172.


3. Di Franco M, Iannuccelli C, Bazzichi L, et al. Misdiagnosis in fibromyalgia: a multicentre study. Clin Exp Rheumatol 2011;29:S104–S108.
4. Gran JT. The epidemiology of chronic generalized musculoskeletal pain. Best Pract Res Clin Rheumatol 2003;17:547–561.


5. Romano TJ. Seminar I: guided posters epidemiology and natural history of myofascial pain and fibromyalgia: coexistence of fibromyalgia syndrome (FS) and systemic lupus erythematosus (SLE). Scand J Rheumatol 1992;21 Suppl 94:12.
6. Abu-Shakra M, Mader R, Langevitz P, et al. Quality of life in systemic lupus erythematosus: a controlled study. J Rheumatol 1999;26:306–309.

7. Buskila D, Gladman DD, Langevitz P, Urowitz S, Smythe HA. Fibromyalgia in human immunodeficiency virus infection. J Rheumatol 1990;17:1202–1206.

8. Meroni PL, Schur PH. ANA screening: an old test with new recommendations. Ann Rheum Dis 2010;69:1420–1422.


9. Mutlu E, Eyigor M, Mutlu D, Gultekin M. Confirmation of anti-DFS70 antibodies is needed in routine clinical samples with DFS staining pattern. Cent Eur J Immunol 2016;41:6–11.



10. Ganapathy V, Casiano CA. Autoimmunity to the nuclear autoantigen DFS70 (LEDGF): what exactly are the autoantibodies trying to tell us? Arthritis Rheum 2004;50:684–688.


11. Ochs RL, Stein TW Jr, Peebles CL, Gittes RF, Tan EM. Autoantibodies in interstitial cystitis. J Urol 1994;151:587–592.


12. Daniels T, Zhang J, Gutierrez I, et al. Antinuclear autoantibodies in prostate cancer: immunity to LEDGF/p75, a survival protein highly expressed in prostate tumors and cleaved during apoptosis. Prostate 2005;62:14–26.


13. Watanabe A, Kodera M, Sugiura K, et al. Anti-DFS70 antibodies in 597 healthy hospital workers. Arthritis Rheum 2004;50:892–900.


14. Bizzaro N, Tonutti E, Villalta D. Recognizing the dense fine speckled/lens epithelium-derived growth factor/p75 pattern on HEP-2 cells: not an easy task! Comment on the article by Mariz et al. Arthritis Rheum 2011;63:4036–4037.


15. Mahler M, Fritzler MJ. The clinical significance of the dense fine speckled immunofluorescence pattern on HEp-2 cells for the diagnosis of systemic autoimmune diseases. Clin Dev Immunol 2012;2012:494356.




16. Kang SY, Lee WI. Clinical significance of dense fine speckled pattern in anti-nuclear antibody test using indirect immunofluorescence method. Korean J Lab Med 2009;29:145–151.


17. Mahler M, Hanly JG, Fritzler MJ. Importance of the dense fine speckled pattern on HEp-2 cells and anti-DFS70 antibodies for the diagnosis of systemic autoimmune diseases. Autoimmun Rev 2012;11:642–645.


18. Kim YA, Lee SS, Park K. Validation of a Korean version of the Fibromyalgia Impact Questionnaire. J Korean Med Sci 2002;17:220–224.



19. Eich W, Hauser W, Arnold B, et al. Fibromyalgia syndrome: definition, classification, clinical diagnosis and prognosis. Schmerz 2012;26:247–258.


20. Grob GN. The evolution of fibromyalgia in modern America. MD Advis 2014;7:28–33.
21. Boomershine CS. A comprehensive evaluation of standardized assessment tools in the diagnosis of fibromyalgia and in the assessment of fibromyalgia severity. Pain Res Treat 2012;2012:653714.




22. Conrad K, Rober N, Andrade LE, Mahler M. The clinical relevance of anti-DFS70 autoantibodies. Clin Rev Allergy Immunol 2017;52:202–216.



23. Mariz HA, Sato EI, Barbosa SH, Rodrigues SH, Dellavance A, Andrade LE. Pattern on the antinuclear antibody- HEp-2 test is a critical parameter for discriminating antinuclear antibody-positive healthy individuals and patients with autoimmune rheumatic diseases. Arthritis Rheum 2011;63:191–200.


24. Ochs RL, Muro Y, Si Y, Ge H, Chan EK, Tan EM. Autoantibodies to DFS 70 kd/transcription coactivator p75 in atopic dermatitis and other conditions. J Allergy Clin Immunol 2000;105:1211–1220.


25. Shinohara T, Singh DP, Chylack LT Jr. Review: age-related cataract: immunity and lens epithelium-derived growth factor (LEDGF). J Ocul Pharmacol Ther 2000;16:181–191.


26. Ayaki M, Ohoguro N, Azuma N, et al. Detection of cytotoxic anti-LEDGF autoantibodies in atopic dermatitis. Autoimmunity 2002;35:319–327.


27. Dellavance A, Viana VS, Leon EP, Bonfa ES, Andrade LE, Leser PG. The clinical spectrum of antinuclear antibodies associated with the nuclear dense fine speckled immunofluorescence pattern. J Rheumatol 2005;32:2144–2149.

28. Maertens G, Cherepanov P, Pluymers W, et al. LEDGF/p75 is essential for nuclear and chromosomal targeting of HIV-1 integrase in human cells. J Biol Chem 2003;278:33528–33539.


29. Yamada K, Senju S, Shinohara T, et al. Humoral immune response directed against LEDGF in patients with VKH. Immunol Lett 2001;78:161–168.


30. Dinerman H, Goldenberg DL, Felson DT. A prospective evaluation of 118 patients with the fibromyalgia syndrome: prevalence of Raynaud’s phenomenon, sicca symptoms, ANA, low complement, and Ig deposition at the dermal-epidermal junction. J Rheumatol 1986;13:368–373.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



