INTRODUCTION
Myocarditis is included in the differential diagnosis of patients presenting with acute chest pain. Clinical presentations vary from nonspecific systemic symptoms (e.g., fever, myalgia, palpitation, chest pain, dyspnea) to fulminant hemodynamic collapse and sudden death. Infections (viral, bacterial, fungal, protozoal, parasitic), toxins, immunological syndromes, and hypersensitivity can cause myocarditis. Among these, hypersensitivity myocarditis is very rare, and its true incidence is unknown. Lewin et al. [1] reported that seven explanted hearts from 288 heart transplants (2.4%) had histological evidence of hypersensitivity myocarditis. Unlike other forms of myocarditis, the prognosis of hypersensitivity myocarditis is excellent, usually improving after administration of the causative drug is ceased. Thus, suspicion of hypersensitivity myocarditis is important. Here, we report a case of hypersensitivity myocarditis, as confirmed by cardiac magnetic resonance imaging (CMR) and endomyocardial biopsy (EMB), which subsequently improved after cessation of the causative drug and administration of prednisolone.
CASE REPORT
A 68-year-old woman presented with constrictive chest pain lasting for 2 hours. She had a history of hypertension, transient cerebral ischemic attack, and osteoarthritis in both knees. Her blood pressure was 91/53 mmHg, pulse rate was 63 beats per minute, respiration rate was 20 breaths per minute, and body temperature was 37℃. Upon physical examination, heart sounds were normal without murmur. Pulmonary auscultation revealed coarse vesicular sounds with crackles on both lower lung fields. Multiple itchy erythematous lesions were present on the hands, buttocks, abdomen, and trunk (Fig. 1).
The results of laboratory examinations were as follows: white blood cell count, 9,140/mm3; eosinophil count, 1,380/mm3; hemoglobin, 10.3 g/dL; platelet count, 123,000/mm3; C-reactive protein level, 1.7 mg/dL; and erythrocyte sedimentation rate, 72 mm/sec. The levels of brain natriuretic peptide, creatine kinase-MB (CK-MB), and troponin-I were 944 pg/mL, 56.73 ng/mL, and 38.85 ng/mL, respectively.
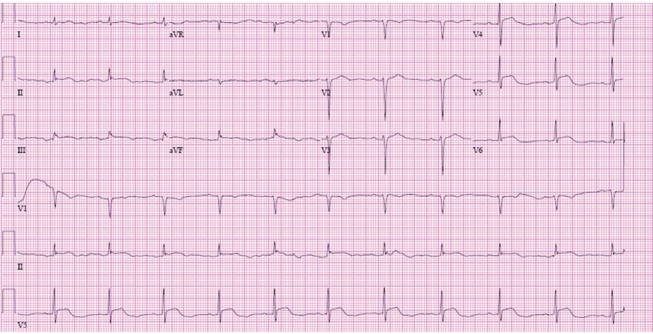
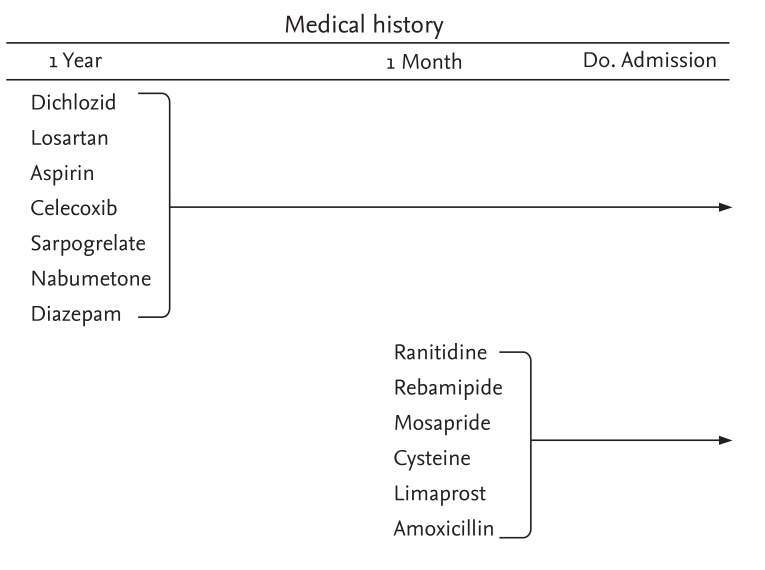
Initial electrocardiography showed ST segment elevation ≥ 1 mm in leads II, III, aVF, V5, and V6 (Fig. 2). Emergency coronary angiography was performed under the assumption of acute ST segment elevation myocardial infarction at the inferolateral wall, but coronary atherosclerosis was not evident. Echocardiography showed the "sparkling" appearance of the left ventricular (LV) myocardium, concentric LV hypertrophy, and mild hypokinesia on the anterior, anterolateral, and posterior walls from the mid-LV to the apex with 50% LV ejection fraction. Peripheral blood eosinophilia, skin rash, and a history of taking multiple medications suggested hypersensitivity myocarditis. The medications taken recently by the patient are shown in Fig. 3. Management of heart failure using aldosterone and furosemide was initiated promptly.
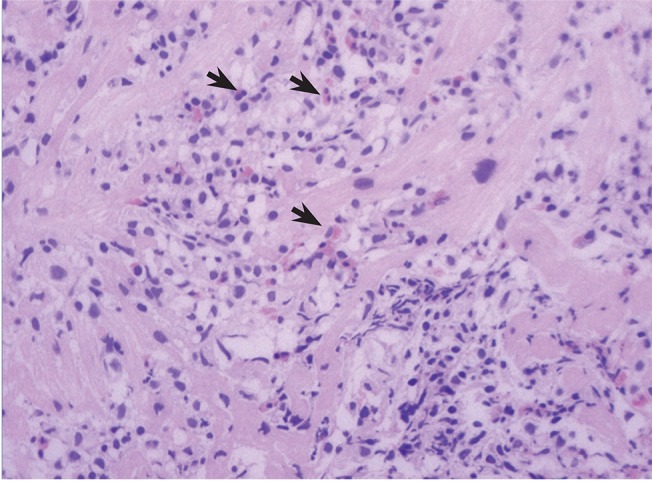
On the second day of hospital admission, however, dyspnea worsened, and newly developed bilateral pulmonary congestion was noted on chest radiography. The level of troponin I peaked at > 50.00 ng/mL. Therefore, CMR and EMB were carried out to confirm the diagnosis of hypersensitivity myocarditis. Prednisolone (60 mg, orally) treatment was initiated. Contrast-enhanced CMR (CE-CMR) showed high signal intensities in circular and nodular patterns mainly in the epicardium and septum of the left ventricle (Fig. 4A). These patterns suggested infiltrative myocarditis, such as amyloidosis, sarcoidosis, or eosinophilic myocarditis. EMB obtained from the septum of the right ventricle revealed lymphocytic and eosinophilic infiltration in the interstitium and perivascular area, findings that were consistent with a diagnosis of hypersensitivity myocarditis (Fig. 5).
The dyspnea improved upon treatment with prednisolone and diuretics. Levels of CK-MB and troponin-I were normalized on days 6 and 9, respectively. The peripheral eosinophil count decreased from 1,380/mm3 to 620/mm3 on day 2 and was normalized (50/mm3) on day 4. The skin rash disappeared almost completely after use of 5 mg prednisolone, and the patient was discharged from the hospital on day 12. Echocardiography at the 6-month follow-up showed normalized regional wall motion abnormalities and improved LV systolic function from 50% to 70% of the ejection fraction. At the 6-month follow-up, CE-CMR showed that previously high signal intensities in circular patterns in the epicardium of the left ventricle had disappeared and the oval-shaped area of high signal intensity in the LV septum had decreased in size (Fig. 4B). The patient remained asymptomatic without the need for prednisolone or diuretics.
DISCUSSION
Eosinophilic myocarditis can be classified according to cause and includes those types associated with systemic disease (e.g., hypereosinophilic syndrome, Churg-Strauss syndrome, malignancies), parasitic infections (e.g., Toxocara canis), and drugs or vaccines (hypersensitivity myocarditis) [2,3]. Eosinophilic myocarditis associated with hypereosinophilic syndrome is usually underpinned by a history of peripheral eosinophilia (> 1.5 × 109/L) lasting > 6 months and is related to systemic involvement (e.g., central nervous system, gastrointestinal tract, heart, skin, or lung) [4]. In the present case, hypereosinophilic syndrome and parasitic infection were excluded because the patient did not have a history of prolonged eosinophilia, other organ involvement, or ingestion of raw meat. Instead, she had a history of taking multiple medications, urticaria, and peripheral eosinophilia. Therefore, we promptly made a diagnosis of hypersensitivity myocarditis and initiated treatment.
Drug-related myocarditis can be classified into five types: 1) hypersensitivity myocarditis; 2) toxic myocarditis; 3) endocardial fibrosis (e.g., ergotamine tartrate, methysergide, phentermine, fenfluramine); 4) drug-induced cardiomyopathy (e.g., anthracycline, chloroquine); and 5) giant cell myocarditis [3]. Hypersensitivity myocarditis is the most common form of acute drug-related myocardial injury. Numerous medications, including clozapine, sulfonamide, penicillin antibiotics, methyldopa, and some antiseizure drugs, have been reported to be associated with hypersensitivity myocarditis [2,3,5,6]. Delayed hypersensitivity is the main pathogenesis and occurs from several days to months after administration of the causative agent [7]. Hypersensitivity myocarditis is not easy to recognize because the clinical features suggestive of drug hypersensitivity reactions (e.g., urticaria, angioedema, malaise, fever, eosinophilia) do not occur uniformly [8]. Most cases of hypersensitivity myocarditis improve after cessation of administration of the putative agent. However, if tissue necrosis or deterioration of clinical status is observed, corticosteroid and cytotoxic agents can be used [5,7]. The clinical course rapidly deteriorated in our case but improved markedly after glucocorticoid administration.
EMB is the gold standard tool in the diagnosis of myocarditis. According to the Dallas criteria, acute myocarditis is defined by lymphocytic infiltrates in association with myocyte necrosis [3]. Eosinophils may be a minor component of postviral and giant cell myocarditis but are predominant in hypersensitivity and parasitic myocarditis as well as hypereosinophilic syndrome [3]. Lymphocytic and eosinophilic infiltrates with slight myocyte necrosis were observed in our case, which coincided with hypersensitivity myocarditis. However, EMB has several clinical limitations. Myocarditis is often a focal process, so sampling error may occur and increase the false-negative rate. A previous study of endomyocardial samples obtained from the hearts of patients who died of myocarditis performed at the Mayo Clinic reported a false-negative rate of 37% for the right ventricle [9]. Other limitations of EMB include the aggressiveness of the procedure as well as the delay until confirmation of the result.
CMR has become the leading modality in the noninvasive imaging of myocarditis. The Lake Louise criteria were found to have a sensitivity of 67%, specificity of 91%, accuracy of 78%, positive predictive value of 91%, and negative predictive value of 69%, when the criteria were compared with clinical or histopathological data [9]. Mahrholdt and Sechtem [10] reported recently that CMR-guided biopsy improved the sensitivity of EMB. Thus, the combined application of CMR and EMB may yield diagnostic synergy and overcome some of the limitations of CMR or EMB when applied individually. In our case, we performed CMR-guided EMB on the right ventricular septum, in which we detected regional high signal intensity on late gadolinium enhancement.
In conclusion, careful history taking (especially with regard to drugs), thorough physical examination, and clinical suspicion can help in recognizing hypersensitivity myocarditis both accurately and promptly. CMR-guided EMB is a valuable tool for the early and appropriate diagnosis for hypersensitivity myocarditis. Our patient recovered completely after cessation of administration of the causative drug and short-term administration of prednisolone for rapidly worsening hypersensitivity myocarditis.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





