 |
 |
| Korean J Intern Med > Volume 28(1); 2013 > Article |
|
Abstract
Background/Aims
The Taxus Liberte stent (Boston Scientific Co.) evolved from the Taxus Express stent, with enhanced stent deliverability and uniform drug delivery. This study was designed to compare angiographic and clinical outcomes in real-world practice between the Taxus Liberte and Taxus Express stents.
Methods
Between 2006 and 2008, 240 patients receiving the Taxus Liberte stent at three centers were registered and compared to historical control patients who had received the Taxus Express stent (n = 272). After propensity score matching, 173 patients treated with the Taxus Liberte stent and the same number of patients treated with the Taxus Express stent were selected. The primary outcome was a composite of major adverse cardiac events (MACE), including cardiac death, myocardial infarction (MI), ischemia driven target vessel revascularization (TVR), and stent thrombosis (ST) at 1 year. An additional angiographic assessment was conducted at 9 to 12 months.
Results
The study showed no significant difference between the Taxus Express and Taxus Liberte stents (death, 1.73% vs. 2.31%, p = 1.000; MI, 0% vs. 1.73%, p = 0.2478; TVR, 2.31% vs. 1.16%, p = 0.6848; and ST, 0% vs. 1.16%, p = 0.4986). The total MACE rate at 1 year did not differ between the groups (4.05% in Taxus Express vs. 4.05% in Taxus Liberte, p = 1.000). In addition, the binary restenosis rate did not differ (2.25% in Taxus Express vs. 1.80% in Taxus Liberte, p = 0.6848).
Drug-eluting stents have shown great efficacy in the reduction of restenosis as compared with bare-metal stent (BMS) by suppressing neointimal growth [1-5]. The second-generation Taxus Liberte stent (paclitaxel-eluting stent, Boston Scientific Co., Natick, MA, USA) evolved from the Taxus Express stent in order to enhance stent deliverability and uniform drug delivery.
However, there is little comparative data from real-world daily clinical practice with these stents. The aim of this study was to compare angiographic and clinical outcomes between the new Taxus Liberte stent and the old Taxus Express stent in real-world clinical practice.
The Taxus Liberte-SR stent consists of a balloon-expandable Liberte stent with a polymer coating containing 1 µg/mm2 of paclitaxel in a slow-release formulation. Drug dosing and release kinetics are identical to that of the Taxus Express-SR stent. Both stents are made from 316 L stainless steel, but the Liberte platform has a more uniform strut pattern and thinner struts (0.097 mm) than the Express platform (0.132 mm) [6].
From May 2006 to June 2008, 240 patients receiving the Taxus Liberte stent at three qualified centers in South Korea (Yeungnam University Medical Center, Keimyung University Dongsan Medical Center, Inje University Busan Paik Hospital, and Inje University Haeundae Paik Hospital) were registered and compared with Taxus Express historical control patients that were treated from January 2005 to April 2006 (n = 272). After performing propensity matching, we were able to successfully match 173 Taxus Express patients with 222 lesions to 173 Taxus Liberte patients with 222 lesions. Patients with a left main lesion and a bifurcation lesion requiring two stents in both the mother and side branch were excluded.
Percutaneous coronary intervention (PCI) was performed using standard techniques. All patients received aspirin 325 mg orally and a loading dose of 300 mg of clopidogrel before coronary angiography (CAG) or after PCI in emergency cases. After PCI, the patients were treated routinely with aspirin 100 mg/day, clopidogrel 75 mg/day, and/or cilostazol 200 mg/day at the operator's discretion. The patients were advised to maintain life-long aspirin therapy. The duration of taking clopidogrel was at the operator's discretion, which depended on the complexity of the lesion and procedure.
Intracoronary nitroglycerin (0.2 mg) was administered before and after each intervention to achieve maximal dilatation. Quantitative CAG was performed immediately before and after stenting by an experienced technician who was blinded to the type of stent deployed. Angiographic measurements included proximal and distal reference, minimum lumen diameter (MLD), percentage of lesion diameter stenosis, and lesion length. Acute gain was measured and defined as the difference between the MLD after stent deployment and baseline MLD. Late lumen loss was calculated as the difference in MLD immediately after the procedure and at angiographic follow-up. All measurements were performed for both the stented segment (in-stent) and 5-mm proximal and distal margins of the stented segment (in-segment). Quantitative coronary angiographic analysis was performed using the computer-assisted automated edge detection method (Centricity, Cardiology CA1000, GE Healthcare, Milwaukee, WI, USA) in the angiography analysis core laboratory at Yeungnam University Medical Center.
The study outcome was a composite of major adverse cardiac events (MACE), including cardiac death, myocardial infarction (MI), ischemia-driven target vessel revascularization (TVR), and stent thrombosis (ST). MI was defined as a recurrent ischemic symptom and/or ECG change with creatine kinase-myocardial band fraction elevation up to twice the upper limit of normal. Ischemia-driven TVR was defined as emergency or elective CABG or repeat PCI in the target vessel for chest pain or a positive test for ischemia [7]. ST was defined as acute (< 24 hours), sub-acute (< 30 days), or late (> 30 days) after the index procedure, and was defined as 1) definite: an acute coronary syndrome with angiographic documentation of either vessel occlusion or thrombus within or adjacent to a previously successfully stented vessel or autopsy evidence of ST; 2) probable: acute MI in the distribution of the treated vessel or unexplained death < 30 days; or 3) possible: unexplained death > 30 days [8]. Angiographic restenosis was defined as a ≥ 50% diameter stenosis within the target lesion. Death was regarded as cardiac except for those of proven non-cardiac origin.
Clinical follow-up was performed at 30 days, 3 months, 6 months, and 1 year after PCI according to our routine practice. Angiographic follow-up was recommended in all living patients and performed at 9 to 12 months after PCI. The 1-year clinical follow-up data were collected by chart review or telephone interview.
Data are expressed as means ± standard deviation (SD) for continuous variables, and as frequencies for categorical variables. Categorical data were analyzed with the chi-square test and continuous variables were evaluated with Student's t tests. To decrease the effect of selection biases and potential confounding, propensity score matching was performed on patient baseline characteristics [9]. For the entire cohort comparison, a separate propensity score for Taxus Liberte versus Taxus Express was derived. The following variables were used to calculate the propensity score: age, sex, hypertension, current smoking, diabetes mellitus, dyslipidemia, previous PCI, previous CABG, renal failure, left ventricular ejection fraction, clinical diagnosis, disease vessel number, medications, target vessel, type of lesion, bifurcation lesion, procedure on infarct related artery, procedure-related MI, stent number, mean stent diameter, total stent length, maximal inflation pressure, and post-stenting adjunctive balloon inflation. After all of the propensity score matches were performed, angiographic and clinical outcomes were compared. The p values < 0.05 were considered to indicate statistical significance. Data were analyzed using the SPSS for Windows version 12.0 (SPSS Inc., Chicago, IL, USA).
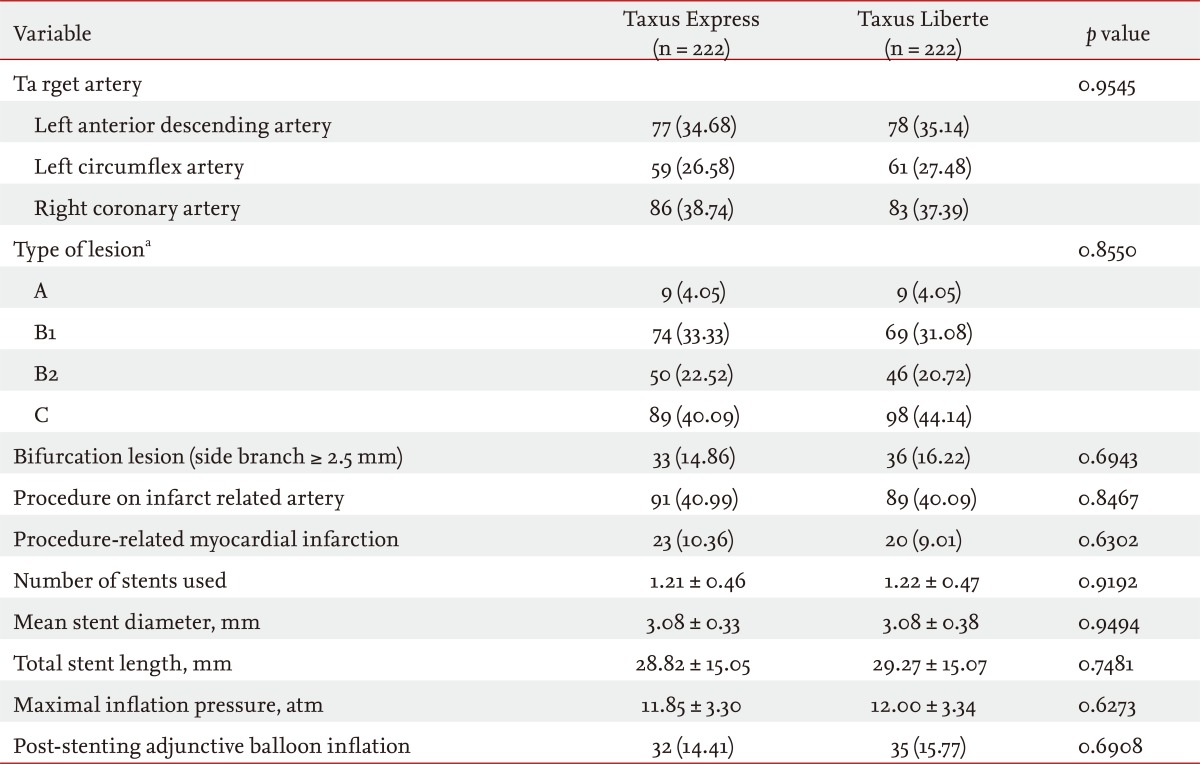
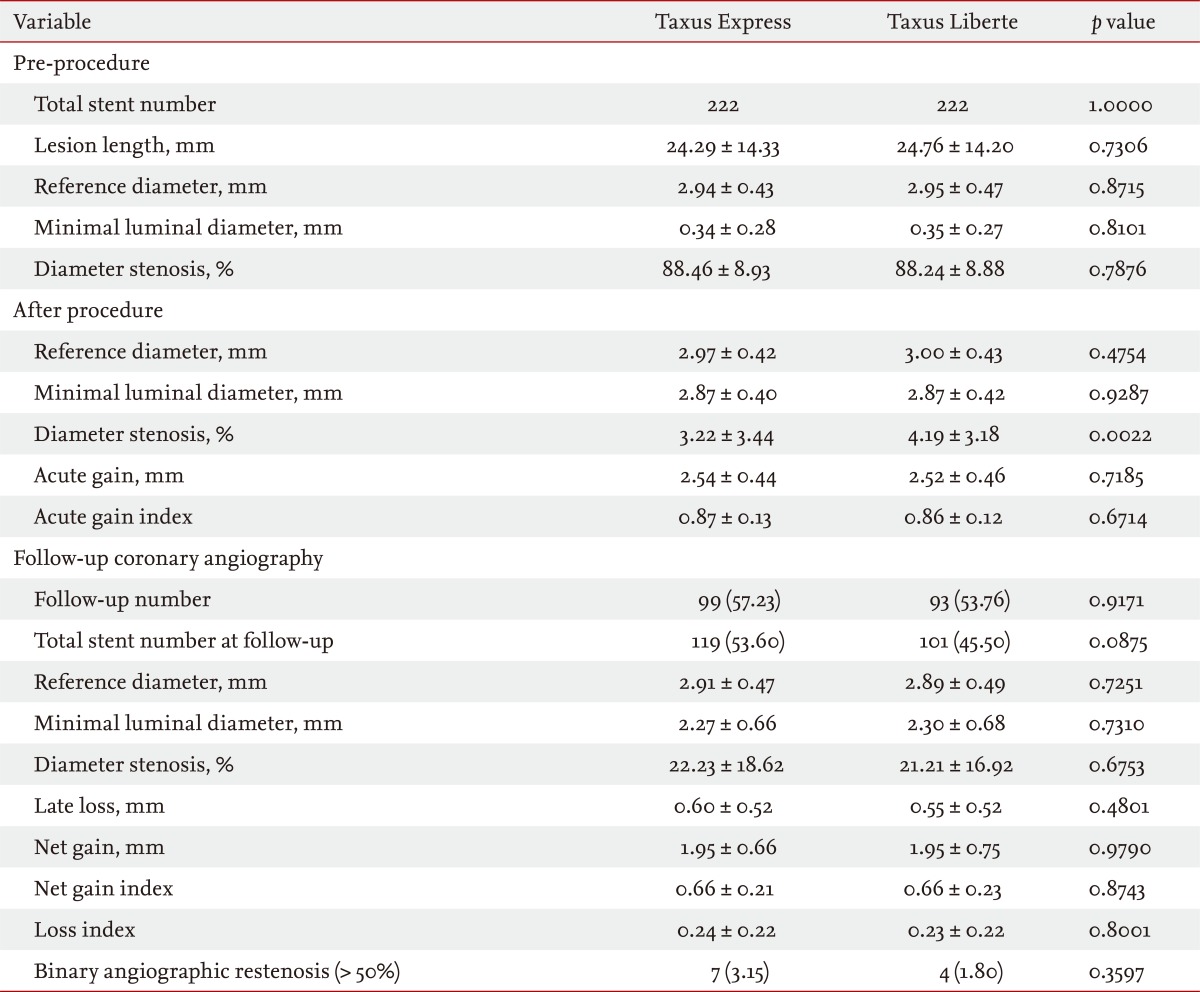
Baseline characteristics (Table 1), angiographic and procedural outcomes (Table 2), and quantitative coronary angiography outcomes in (Table 3) were well matched and comparable between groups. Baseline characteristics and angiographic findings including restenosis rate (3.15% in Taxus Express vs. 1.80% in Taxus Liberte, p = 0.3597) and the mean duration of dual antiplatelet therapy (13.2 months in Taxus Express vs. 12.7 months in Taxus Liberte, p = 0.7582) were not different between the groups.
Table 4 shows clinical outcomes of patients. In the 30-day-outcome, the frequency of MACE was not different between the groups (0% in Taxus Express group vs. 0.58% in Taxus Liberte group; p = 1.000). From 30 days to 1 year after the index PCI, the frequency of MACE also did not differ (3.47% in Taxus Express group vs. 2.89% in Taxus Liberte group; p = 0.7593). Overall cumulative MACE rate for 1 year was 4.05% for the Taxus Express group and 4.05% for the Taxus Liberte group (p = 1.000). No acute ST was observed. Subacute ST was observed in one case in the Taxus Liberte group. One late ST was observed in the Taxus Liberte group. No death occurred in the Taxus Express group and one patient died due to a cardiac cause in the Taxus Liberte group (p = 1.000). The MACE-free survival rate at 1 year is indicated in Fig. 1; there was no significant difference between the groups (p = 0.808).
The main findings of this study suggest that both Taxus stents, with the same polymer and the same drug on different stent designs, had similarly good 1-year clinical and angiographic outcomes in real-world use. The Taxus Express stent shows polymer-regulated delivery of paclitaxel from the Boston Scientific/Medinol NIR and Boston Scientific EXPRESS stent platforms [10]. The Taxus stent consists of three components: 1) the metallic stent, 2) paclitaxel, an antiproliferative agent that inhibits neointimal formation, and 3) the polymer, a drug-carrier vehicle [11,12]. The Taxus Express stent was approved in March 4, 2004 by the Food & Drug Administration for marketing and sale in the United States, and thereafter various clinical data were released. Taxus stents have been shown to be superior to BMSs in reducing restenosis rates and target lesion revascularization [4,5,13-15].
Stent design is recognized as an important determinant of stent performance [16]. Strut thickness, metal composition, and radial strength can each influence short- and long-term outcomes [17,18]. The second-generation Taxus Liberte stent was designed to provide enhanced deliverability, better conformability, uniform vessel coverage, and a lower profile as compared with the Taxus Express. Although most of the Taxus stents are similar, the struts of the Taxus Liberte are thinner (0.0038 inches vs. 0.0052 inches) and the strut distribution is more uniform than those of the Taxus Express. The continuous cell design and smaller open cell area (2.75 mm2 vs. 2.84 mm2) of the Taxus Liberte provides more uniform vessel coverage than that of the Taxus Express.
The pivotal Taxus "ATLAS" trial compared the Taxus Liberte with the Taxus Express. This study showed that the Taxus Liberte was not inferior to the Taxus Express, despite the treatment of more complex lesions with the Taxus Liberte. The authors concluded that successful transfer of the proven Taxus technology to the more advanced Taxus Liberte platform was demonstrated [19]. The latter study focused mainly on stable angina patients (52.8% in Taxus Express vs. 60.2% in Taxus Liberte; p = 0.0015); however, our study mostly included patients with unstable angina and MI (65.9% in Taxus Express vs. 68% in Taxus Liberte). The MACE rate in the Taxus ATLAS study was 12.3% in the Taxus Express group and 12.5% in the Taxus Liberte group, driven mainly by an increased TVR rate (8.9% in Taxus Express vs. 9.2% in Taxus Liberte; p = 0.833). Our study showed that the MACE rate was 4.05% in each group and TVR rate was 2.31% in Taxus Express and 1.16% in Taxus Liberte (p = 0.684). The reason for the better results is unknown. However, longer treatment duration with the dual antiplatelet regimen may be a cause. In the Taxus ATLAS study, the incidence of the dual antiplatelet regimen at 9 months was only 62.6% in the Taxus Liberte and 54.2% in the Taxus Express patients. In our study, although the duration of dual antiplatelet was at the operator's discretion, the mean duration was 12.7 months in the Liberte group and 13.2 months in the Express group.
Several previous studies have demonstrated that thinner strut stents are associated with a significant reduction of clinical and angiographic restenosis as compared with thicker-strut stents [17,18,20]. Another study comparing two Taxus stents, the Taxus ATLAS small vessel (2.25 mm stent) and long lesion (38 mm stent) multicenter studies, showed that the Taxus Liberte stent improved MACE rates as compared with the earlier Taxus Express stent in both small vessels and long lesions [6].
Although the mechanism for this improved outcome remains unclear, thinner strut thickness and the more uniform and denser distribution of struts in the Taxus Liberte may be related to reduced deep vessel injury during implantation or improved healing, beyond simply improving deliverability [17].
Our study is a real-world clinical study, comparing the new-generation Taxus Liberte stent with the Taxus Express. At first, we thought the later-developed stent (Taxus Liberte) may have better clinical and angiographic outcomes than the earlier version (Taxus Express). However, our study did not show any clinical difference at 1 year. First, this may be due to the relatively short follow-up duration and a longer follow-up might reveal differences. Second, the potential impact of reduced strut thickness on late lumen loss may be overcome by the relative impact of drug and polymer.
This study has some limitations. First, the study population was relatively small. Second, this was a retrospective, observational, non-randomized study. However, we performed propensity score matching to minimize these shortcomings. Third, after propensity score matching, 69 patients in the Liberte group and 99 patients in the Express group were excluded. Exclusion of such large numbers of patients may weaken the representativeness of the target population. Fourth, as we mentioned, there was no follow-up beyond 1 year. With further long-term follow-up data, we cannot exclude the possibility that the final results may be different. Fifth, we did not use imaging modalities, such as intravascular ultrasound, in most patients to more precisely determine differences and the underlying mechanisms between the two stents.
In conclusion, in this real-world study of experience with the two Taxus stents, both showed similarly good clinical and angiographic outcomes. A long-term follow-up study is warranted.
1. The second generation paclitaxel-eluting Taxus Liberte stent consists of same polymer and drug with the Taxus Express stent, but has different stent design and evolved to enhance stent deliverability and uniform drug delivery.
2. Stent design is recognized as an important determinant of stent performance, and previous study showed that Taxus Liberte stent improved major adverse cardiac event rates compared with the earlier Taxus Express stent in both small vessels and long lesions.
3. Although both stents showed similarly good clinical and angiographic outcomes, there was no clinical differences at 1 year in this real-world clinical study.
References
1. Morice MC, Serruys PW, Sousa JE, et al. A randomized comparison of a sirolimus-eluting stent with a standard stent for coronary revascularization. N Engl J Med 2002;346:1773–1780PMID : 12050336.


2. Moses JW, Leon MB, Popma JJ, et al. Sirolimus-eluting stents versus standard stents in patients with stenosis in a native coronary artery. N Engl J Med 2003;349:1315–1323PMID : 14523139.


3. Schofer J, Schluter M, Gershlick AH, et al. Sirolimus-eluting stents for treatment of patients with long atherosclerotic lesions in small coronary arteries: double-blind, randomised controlled trial (E-SIRIUS). Lancet 2003;362:1093–1099PMID : 14550694.


4. Colombo A, Drzewiecki J, Banning A, et al. Randomized study to assess the effectiveness of slow- and moderate-release polymer-based paclitaxel-eluting stents for coronary artery lesions. Circulation 2003;108:788–794PMID : 12900339.


5. Stone GW, Ellis SG, Cox DA, et al. A polymer-based, paclitaxel-eluting stent in patients with coronary artery disease. N Engl J Med 2004;350:221–231PMID : 14724301.


6. Turco MA, Ormiston JA, Popma JJ, et al. Reduced risk of restenosis in small vessels and reduced risk of myocardial infarction in long lesions with the new thin-strut TAXUS Liberte stent: 1-year results from the TAXUS ATLAS program. JACC Cardiovasc Interv 2008;1:699–709PMID : 19463387.


7. Kim U, Seol SH, Kim DI, et al. Clinical outcomes and the risk factors of coronary artery aneurysms that developed after drug-eluting stent implantation. Circ J 2011;75:861–867PMID : 21258162.


8. Cutlip DE, Windecker S, Mehran R, et al. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation 2007;115:2344–2351PMID : 17470709.


9. D'Agostino RB Jr. Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Stat Med 1998;17:2265–2281PMID : 9802183.


10. Halkin A, Stone GW. Polymer-based paclitaxel-eluting stents in percutaneous coronary intervention: a review of the TAXUS trials. J Interv Cardiol 2004;17:271–282PMID : 15491330.


11. Laarman GJ, Suttorp MJ, Dirksen MT, et al. Paclitaxel-eluting versus uncoated stents in primary percutaneous coronary intervention. N Engl J Med 2006;355:1105–1113PMID : 16971717.


12. Kitahara H, Kobayashi Y, Yamaguchi M, et al. Damage to polymer of paclitaxel-eluting stent. Circ J 2008;72:1907–1908PMID : 18787290.


13. Grube E, Silber S, Hauptmann KE, et al. TAXUS I: six- and twelve-month results from a randomized, double-blind trial on a slow-release paclitaxel-eluting stent for de novo coronary lesions. Circulation 2003;107:38–42PMID : 12515740.


14. Stone GW, Ellis SG, Cox DA, et al. One-year clinical results with the slow-release, polymer-based, paclitaxel-eluting TAXUS stent: the TAXUS-IV trial. Circulation 2004;109:1942–1947PMID : 15078803.


15. Schwalm JD, Ahmad M, Velianou JL, Pericak D, Natarajan MK. Long-term outcomes with paclitaxel-eluting stents versus bare metal stents in everyday practice: a Canadian experience. Can J Cardiol 2010;26:e40–e44PMID : 20151057.



16. Rogers CD. Optimal stent design for drug delivery. Rev Cardiovasc Med 2004;5(Suppl 2):S9–S15PMID : 15184829.


17. Briguori C, Sarais C, Pagnotta P, et al. In-stent restenosis in small coronary arteries: impact of strut thickness. J Am Coll Cardiol 2002;40:403–409PMID : 12142103.


18. Kastrati A, Mehilli J, Dirschinger J, et al. Intracoronary stenting and angiographic results: strut thickness effect on restenosis outcome (ISAR-STEREO) trial. Circulation 2001;103:2816–2821PMID : 11401938.


Figure 1
Major adverse cardiac events (MACE)-free survival rate at 1 year. There was no difference between the Taxus Express and Taxus Liberte groups (p = 0.808).
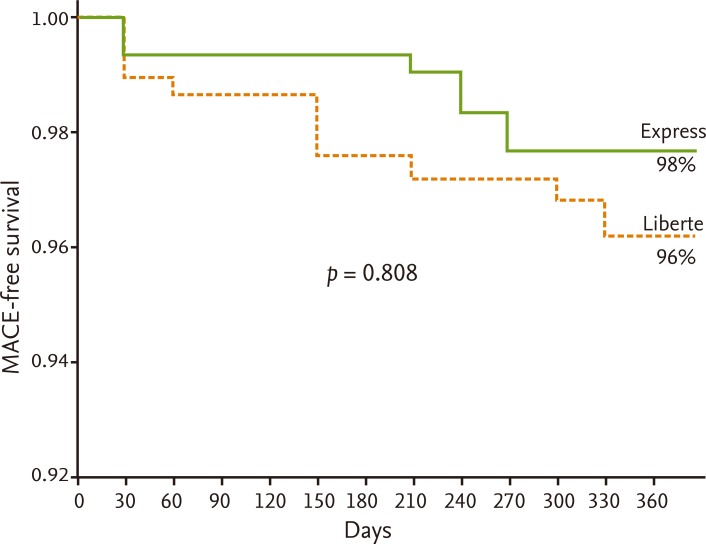
Table 1
Baseline characteristics of the study patients
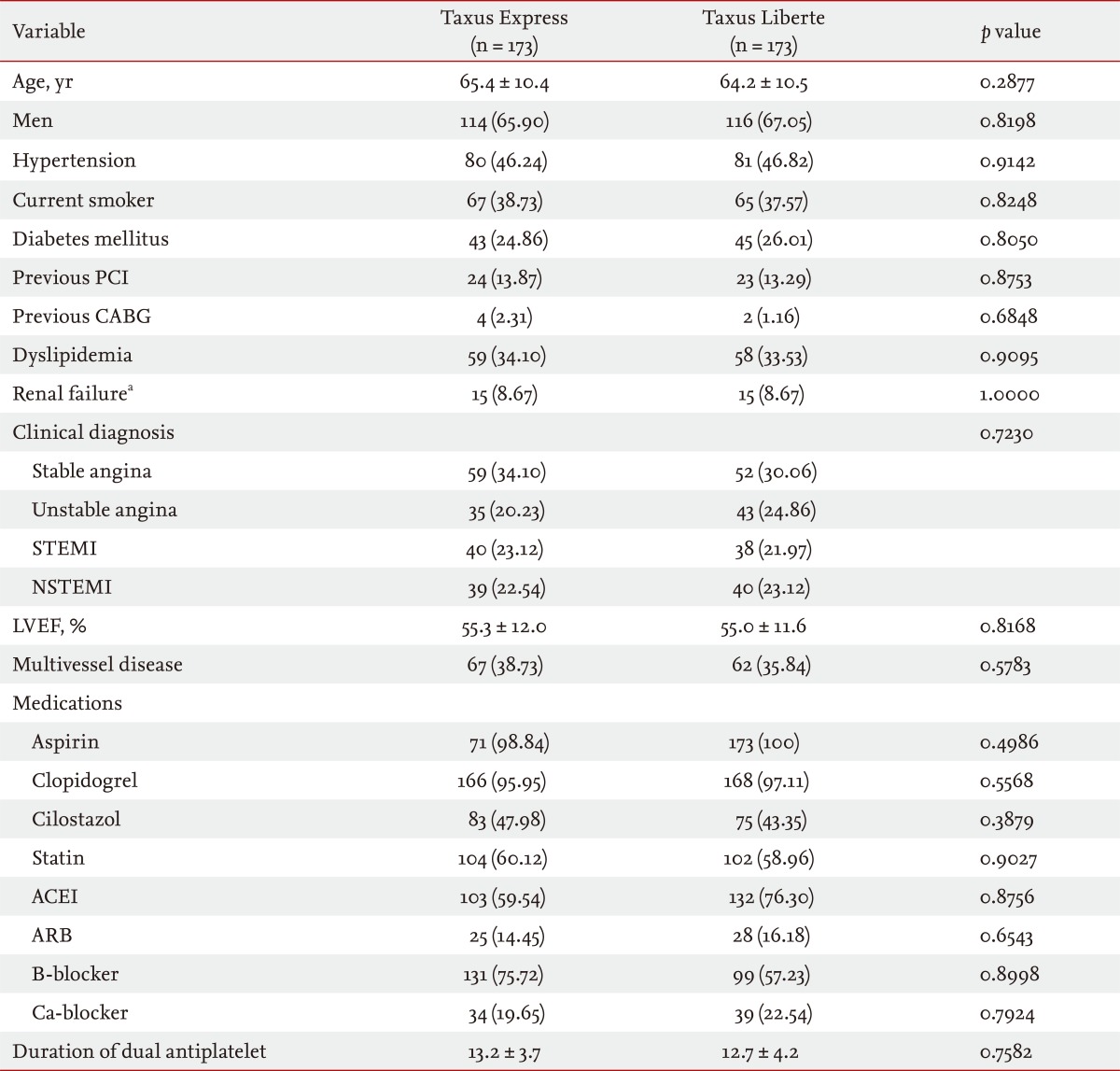
Values are presented as mean ± SD or number (%).
PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; STEMI, ST-segment elevation myocardial infarction; NSTEMI, non-ST-segment elevation myocardial infarction; LVEF, left ventricular ejection fraction; ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker.
aCreatinine > 1.5 mg/dL.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



