 |
 |
| Korean J Intern Med > Volume 27(3); 2012 > Article |
|
Abstract
Background/Aims
The clinical implications of hypocholesterolemia have not been well studied, although some studies have revealed an association between hypocholesterolemia and intracerebral hemorrhage (ICH). We evaluated the clinical characteristics of subjects with very low-density lipoprotein cholesterol (LDL-C) and compared the risk for ICH using various clinical parameters.
Methods
Using hospital records, we evaluated the clinical characteristics of subjects with LDL-C levels Ōēż 40 mg/dL (very low LDL-C group). We also evaluated the risk for ICH in this very low LDL-C group and in subjects with low LDL-C Ōēż 70 mg/dL (low LDL-C group).
Results
Among 34,415 subjects who presented at the laboratory for serum LDL-C measurements, 250 subjects had a very low serum LDL-C level (Ōēż 40 mg/dL). About half of the subjects were statin users; the very low LDL levels in the other subjects were likely attributable to alcohol consumption or a various chronic illness such as liver disease or end-stage renal disease (ESRD). ICH occurred in three subjects with very low LDL-C, all of whom had no history of statin use. ESRD tended to be associated with ICH in subjects with serum LDL-C Ōēż 70 mg/dL.
Remarkable progress has been made in recent years regarding lipid-lowering therapy, particularly with the development of potent statins. Although intensive lipid-lowering therapy has contributed significantly to the primary and secondary prevention of cardiovascular disease (CVD), the large residual risk for CVD is a major limitation [1]. To address this issue, various combination regimens and new therapeutic strategies are being studied. Another concern is that the lower limit of safe and effective cholesterol reduction has not been established. Some experts have suggested that a plasma low-density lipoprotein cholesterol (LDL-C) level of 25 mg/dL is sufficient to supply peripheral cholesterol needs [2]. By comparison, the plasma LDL-C level in human newborns has been reported to range from 40 to 50 mg/dL [3]. To determine the lowest LDL-C level that may be reached therapeutically without causing harm, further studies regarding the clinical state of patients with low serum LDL-C are warranted.
Both low and high serum total cholesterol (TC) levels have been reported to be associated with increased mortality, thus demonstrating a U- or J-shaped relationship [4,5]. CVD represents the major cause of death related to high TC concentrations, whereas non-atherosclerotic conditions, including cancer, hemorrhagic stroke, and liver disease, are mortality causes associated with low TC levels [6,7]. In a 23-year prospective cohort study, when deaths during the first several years of follow-up were excluded, only the association between low TC and hemorrhagic stroke remained [5]. Among various clinical factors, high blood pressure (BP) and alcohol consumption increased the risk for hemorrhagic stroke in subjects with low TC [5,8]. Most guidelines for the treatment of hypercholesterolemia recommend a target LDL-C level of < 70 mg/dL in patients with very high risk for CVD [2,9]. An intensive therapeutic strategy has led to very low LDL-C (Ōēż 40 mg/dL) in statin-treated subjects in many clinical trials of intensive statin treatment; however, an increased incidence of intracerebral hemorrhage (ICH) has not been confirmed [10-14]. The Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial showed an increased incidence of ICH in subjects treated with 80 mg of atorvastatin after a recent (within 1 to 6 months) stroke or transient ischemic attack [15]. Nevertheless, no association was found between the risk for ICH and the baseline or recent LDL-C level in statin-treated patients [16].
Hypocholesterolemia may be defined by a serum TC level lower than the 5th percentile for age and gender, or by the cut-off value that predicts an adverse prognosis by epidemiological studies [17]. The 5th percentile for TC for males in the United States is 131 to 154 mg/dL, and the 5th percentile for LDL-C in Western countries is about 90 mg/dL [18]. Although hypocholesterolemia is not uncommon, it is frequently overlooked. Its prevalence varies according to populations and reaches 2% to 3% in the general population and 6.2% in patients hospitalized in the department of internal medicine [19]. Additionally, data are lacking regarding the clinical meaning of a very low serum LDL-C level. Further research concerning very low LDL-C of primary and secondary origins may lead to important clinical implications for hypolipidemic therapy.
In the present study, we conducted a hospital-based retrospective database analysis to evaluate the clinical characteristics of subjects with very low serum LDL-C, and we compared the risk for ICH using various clinical parameters in subjects with LDL-C levels Ōēż 70 mg/dL.
We reviewed the electronic hospital records of subjects who visited Jeju National University Hospital between January 2005 and March 2011 and who received routine biochemical tests, lipid profiles (including LDL-C), body anthropometry, and vital sign assessments. We selected subjects whose LDL-C level was Ōēż 40 mg/dL. We excluded patients with systemic infectious or inflammatory disease, trauma, primary or secondary brain tumor, type 1 diabetes, or acute systemic illness; pregnant subjects; and patients who had received thrombolytic therapy during hospitalization when a lipid profile was measured.
To compare the risk for ICH according to the serum LDL-C level, we collected clinical data from subjects with an LDL-C level between 41 and 70 mg/dL. Because the present study was cross-sectional and the causal relationship between a low LDL-C level and ICH could not be easily explained, we considered both the sequence of LDL-C measurement and ICH, and the time frame and interval between the date of available low or very low LDC-C levels and the event. Thus, when we evaluated the incidence of ICH in the study subjects, we did not include ICH events occurring before or longer than 6 months after an LDL-C level was measured.
Hypertension was defined as anti-hypertensive drug use, systolic BP Ōēź 140 mmHg, or diastolic BP Ōēź 90 mmHg. End-stage renal disease (ESRD) was defined as the use of renal replacement therapy and an estimated glomerular filtration rate of < 30 mL/min/1.73 m2, based on the Modification of Diet in Renal Disease equation. Obesity was defined as a body mass index Ōēź 30 kg/m2. Diabetes mellitus was defined as the use of insulin or oral anti-diabetic agent, or a diagnosis according to the American Diabetes Association guidelines [20]. Chronic liver disease was considered to be chronic viral hepatitis or alcoholic liver disease. Alcohol use was defined as two or more drinks in a week. Stroke was a neurological deficit lasting longer than 24 hours. Computed tomographic or magnetic resonance imaging of the brain was reviewed in all stroke cases. Lipids (TC, triglycerides [TGs], high-density lipoprotein cholesterol [HDL-C], and LDL-C) were measured by direct methods with an automated biochemical analyzer (TBA-200FR NEO, Toshiba Medical Systems, Tokyo, Japan) using commercially available diagnostic kits (Determiner-L TC-II, TG-II, HDL-C, and LDL-C; Kyowa Medex Co., Ltd., Tokyo, Japan).
The study protocol was reviewed and approved by the Jeju National University Hospital Institutional Review Board (IRB No. #2011-46).
Statistical analyses were performed using SPSS version 14.0 (SPSS Inc., Chicago, IL, USA). Student's t test with equal variance was used to determine significant differences of the means of normally distributed continuous variables (i.e., age) between groups. For nonparametric data, the Mann-Whitney U test was used. Pearson's correlation was calculated between the LDL-C level and other variables. The incidence of ICH was compared between subgroups by using Fisher's exact test. Stepwise regression tests were performed to calculate the relationships between ICH and various risk factors. Statistical significance was taken at ╬▒ = 0.05 (p < 0.05) with an associated 95% confidence interval (CI). Data are presented as means ┬▒ standard error.
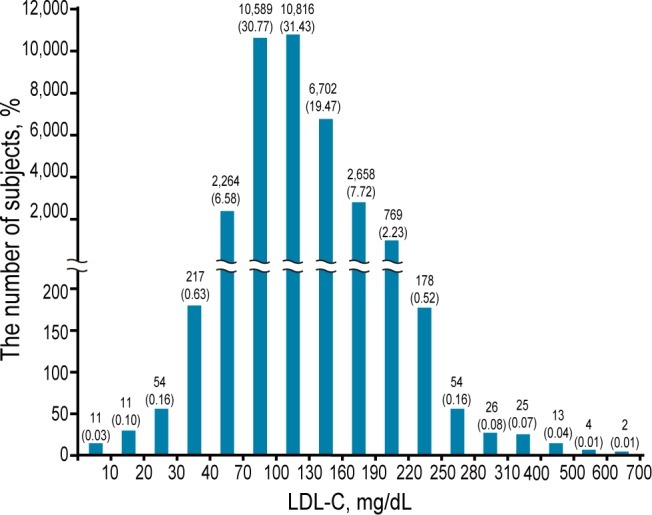
Serum LDL-C levels were measured in 34,415 subjects from January 2005 to March 2011. The LDL-C concentrations had a wide range (Fig. 1), with a median (25th, 75th percentile) concentration of 109 mg/dL (85, 135 mg/dL). In 2,579 subjects, serum LDL-C was Ōēż 70 mg/dL, and 315 subjects had serum LDL-C levels Ōēż 40 mg/dL. We excluded 1,620 subjects according to the exclusion criteria. A total of 959 subjects with a serum LDL-C level Ōēż 70 mg/dL were included in the present study. They were divided into two groups based on serum LDL-C levels: a very low group (Ōēż 40 mg/dL; n = 250) and a low group (41 to 70 mg/dL; n = 709). We focused our analysis on subjects with very low LDL-C to determine the clinical characteristics in these subjects.
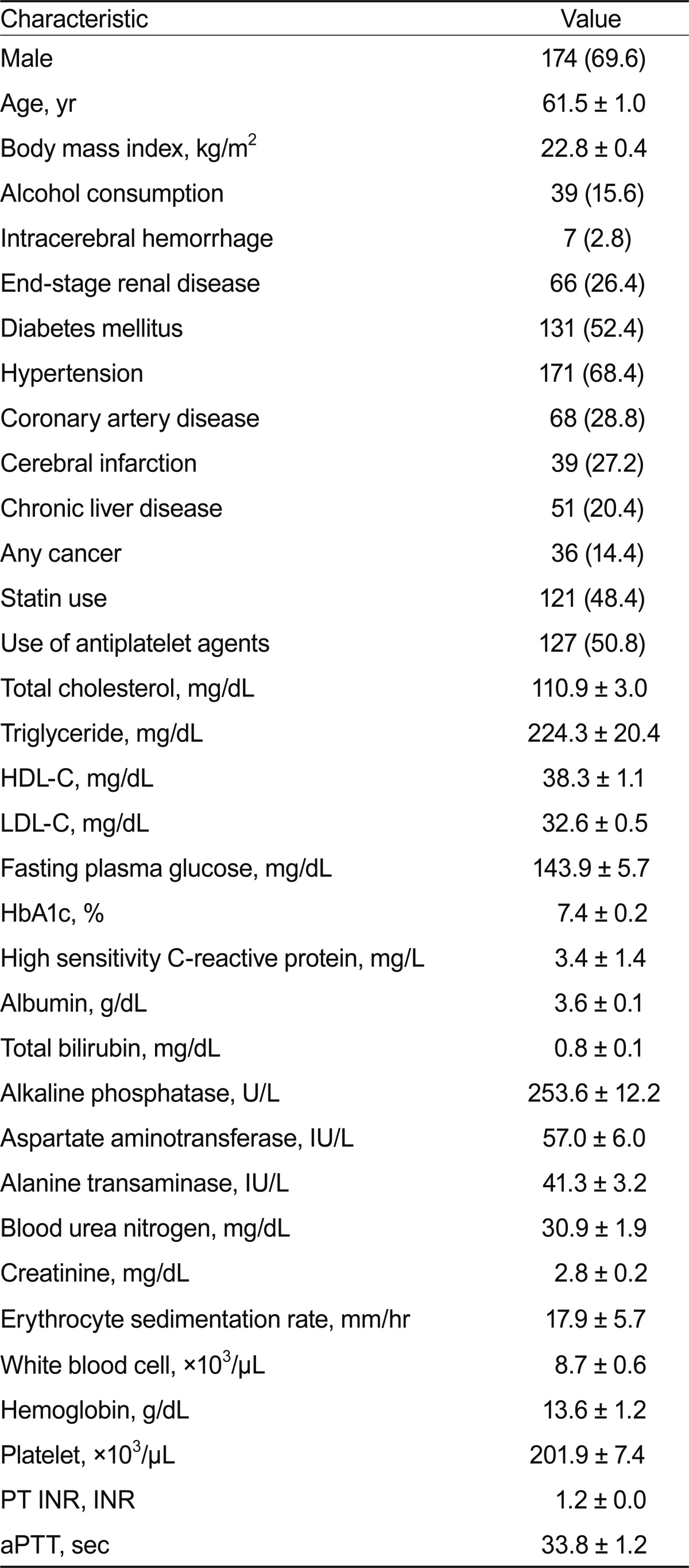
Table 1 shows the demographic and clinical characteristics of the subjects with very low serum LDL-C levels (Ōēż 40 mg/dL). Their mean age was 61.5 years, and about 70% were male. More than half had diabetes, and two-thirds of the subjects had hypertension. These findings indicate that more patients with cardiometabolic disease and/or CVD were subjected to serum LDL-C measurement in the hospital. In addition, 66 subjects (26.4%) had CKD, 39 subjects (27.2%) had cerebral infarction, 51 (20.4%) had liver disease, and 36 (14.4%) had a malignancy. Despite the markedly low LDL-C level in these subjects, the mean serum TG level was relatively high (224.3 ┬▒ 20.4 mg/dL). About half of the subjects with very low LDL-C were current statin users, whereas very low LDL-C was attributable to other causes in the remaining subjects in this group.
The subjects with very low LDL-C levels were divided into two subgroups according to their use of cholesterollowering agents (Table 2). The statin user group (n = 129) comprised subjects who were using a cholesterol-lowering agent, which in all cases was a statin, and the spontaneous group (n = 121) comprised subjects who were not using a cholesterol-lowering agent. Compared with the statin users, the spontaneous group had lower mean values for age, body mass index, and serum levels of HDL-C, LDL-C, and albumin; a higher serum TG level; a longer prothrombin time (PT); and higher prevalence rates of frequent alcohol consumption (24.0% vs. 6.6%, respectively; p < 0.001) and liver disease (27.1% vs. 13.2%, respectively; p < 0.01). The stain user group had a higher incidence of cardiometabolic disease and/or cardiovascular complications, which may be an important reason for their use of statins and anti-platelet agents.
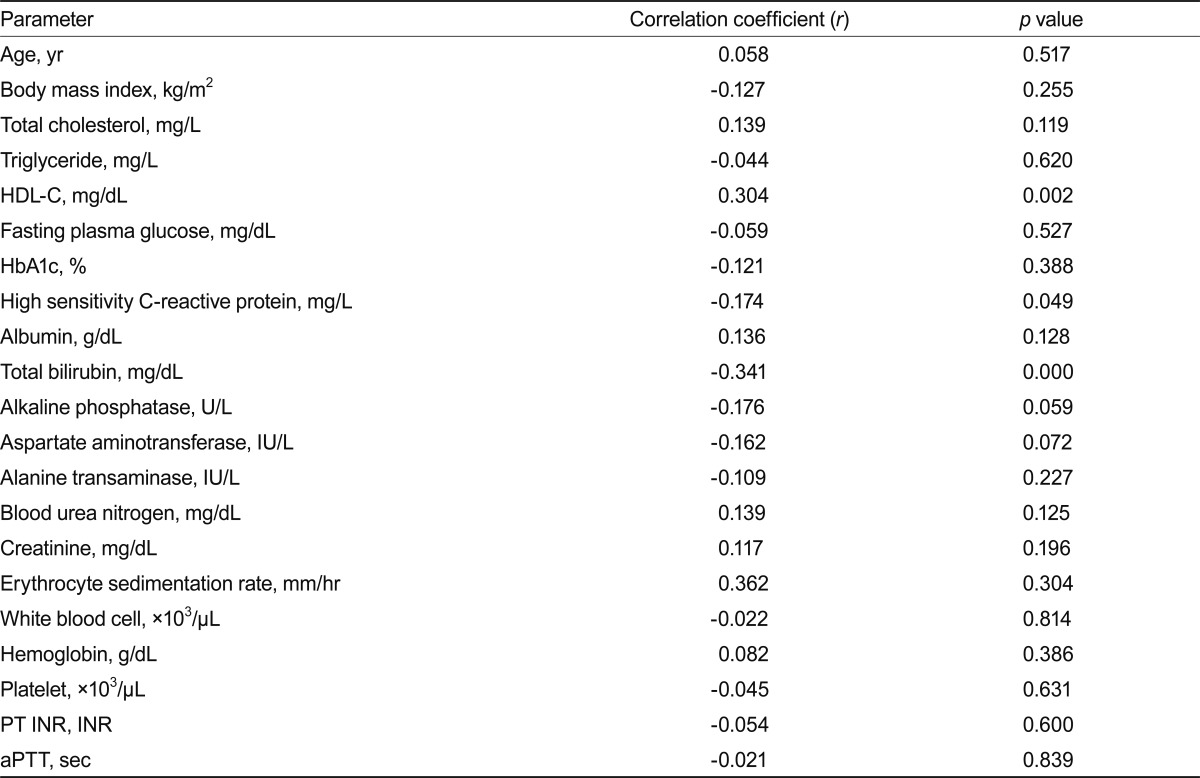
We evaluated correlations between the serum LDL-C level and other factors in subjects with very low LDL-C. High-sensitivity C-reactive protein (hsCRP), total bilirubin, alkaline phosphatase, aspartate aminotransferase, and alanine aminotransferase levels showed negative correlations, whereas HDL-C and albumin levels showed positive correlations with the LDL-C level in subjects with serum LDL-C concentrations Ōēż 40 mg/dL (Table 3). After adjusting for all of the above factors in addition to age and gender, linear regression analysis showed that the serum LDL-C level was associated with the total bilirubin level (╬▓ = -0.265; p = 0.001).
When we evaluated the same correlations in subjects with very low LDL-C and without a history of hypolipidemic therapy, the correlations of the LDL-C level with the hsCRP, total bilirubin, and HDL-C levels remained significant (Table 4).
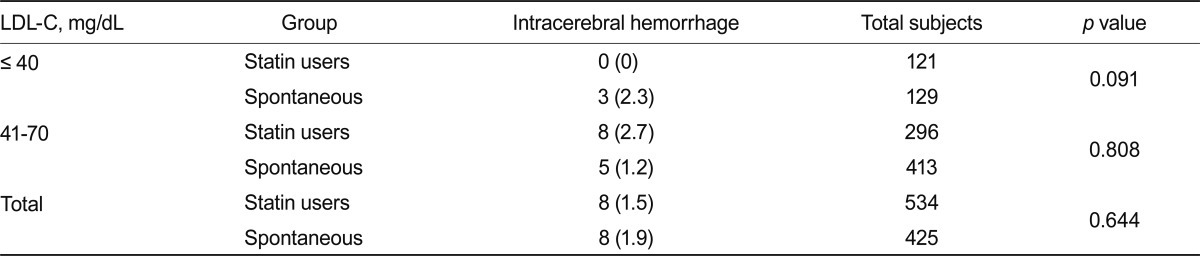
Patients with hypocholesterolemia have been reported to have an increased risk for hemorrhagic stroke [8,21]. Among the 250 subjects with very low LDL-C, three ICH cases were identified, none of whom were statin users (Table 5). However, because of the low number of ICH cases, the difference between the statin and spontaneous groups did not reach a level of significance (p = 0.091). Nevertheless, these results suggest that compared with a spontaneously very low LDL-C level, a statin-induced very low LDL-C level does not lead to an increased risk for ICH. When the analysis was extended to include subjects with a low serum LDL-C level, 13 additional ICH cases were identified among 709 subjects with serum LDL-C levels of 41 to 70 mg/dL (Table 5). Eight of these cases were statin users (n = 296), and five cases had no history of hypolipidemic drug treatment (n = 413, p = 0.808). Taken together, the 16 ICH cases were represented by eight subjects from the statin user group and eight from the spontaneous group, with no difference in the incidence of ICH between these groups (Table 5). The incidence of ICH was comparable between subjects with LCL-C levels Ōēż 40 mg/dL and subjects with LDL-C levels of 41 to 70 mg/dL (data not shown).
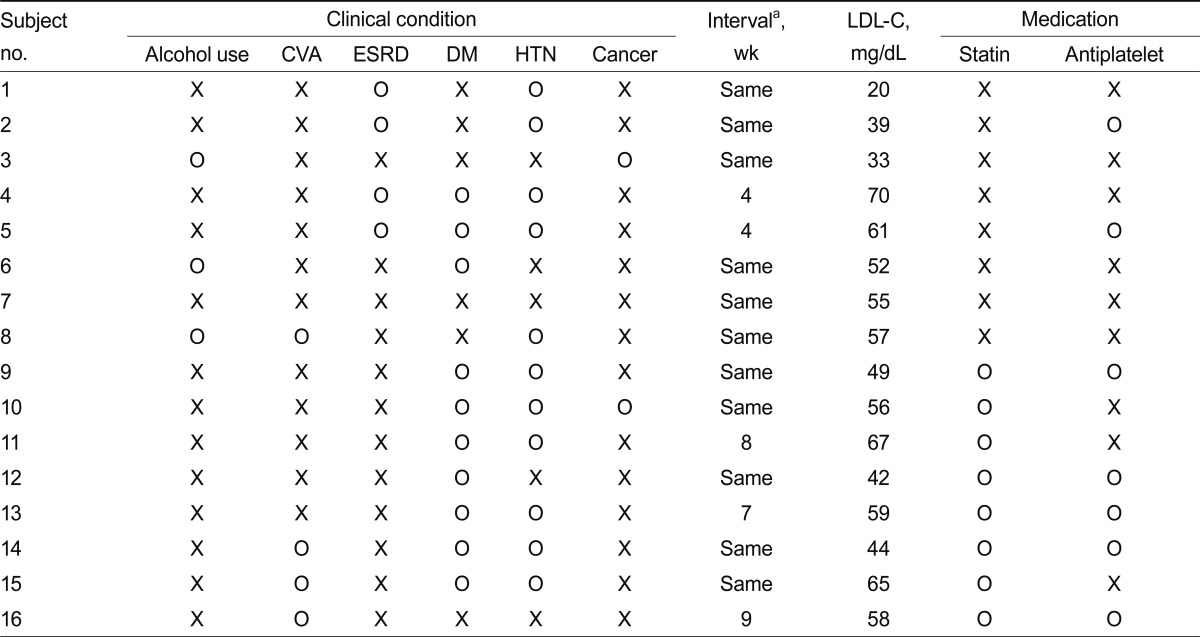
The clinical characteristics of the 16 ICH cases are summarized in Table 6.
In a multiple stepwise regression analysis after adjustments for age, gender, alcohol use, hypertension, ESRD, chronic liver disease, statin use, and LDL-C, only ESRD showed a tendency to be associated with ICH in subjects with serum LDL-C levels Ōēż 70 mg/dL (odds ratio, 2.59; 95% CI, 0.74 to 9.02; p = 0.135) (data not shown).
The causes of primary hypocholesterolemia include certain disorders resulting from genetic mutations in the pathways of cholesterol absorption, biosynthesis, or metabolism; for example, abetalipoproteinemia, hypobetalipoproteinemia, Tangier disease, chylomicron retention disease, and inherited disorders of cholesterol biosynthesis [18]. The causes of secondary hypocholesterolemia comprise anemia, hyperthyroidism, malignancy, chronic hepatitis B or C infection, other liver diseases, critical illness, serious stress, malabsorption or malnutrition, acute or chronic infection, chronic inflammation, and use of some drugs [18]. Although direct causal relationships have not been clarified, hypocholesterolemia can result in adverse events such as increased mortality, ICH, cancer, infection, adrenal failure, and mental disorders [18,22-25]. Therefore, with the practice of intensive lipid-lowering therapy and the tendency toward increased indications for statins, optimal cholesterol levels and the clinical importance of hypocholesterolemia should be considered [17,26,27].
In the present study, we evaluated the clinical characteristics of subjects with very low LDL-C levels of Ōēż 40 mg/dL. We chose this LDL-C concentration as a cutoff value because levels close to this can be achieved with intensive statin therapy in some patients with very high cardiovascular risk [13,28]. Among the study subjects with very low LDL-C, about half were statin users. A significant proportion of the remaining subjects, who had very low LDL-C without statin or other hypolipidemic therapy, had one or more chronic illnesses, consistent with previous reports [18]. Compared with the statin users, the spontaneous group with very low LDL-C had more cases of chronic liver disease and frequent alcohol consumption.
Some concerns exist regarding an association between statin use and ICH [16]. In one study, a history of hypercholesterolemia and statin use was associated with decreased risk for ICH, and no definitive evidence was found to support an increased risk for ICH despite the widespread use of statins [29]. Furthermore, an increase in hemorrhagic stroke was not observed in long-term statin secondary prevention trials that examined hemorrhagic stroke as a secondary endpoint [30-32]. The incidence of hemorrhagic stroke was Ōēż 0.5% in both the placebo and statin-assigned groups.
A post-hoc analysis of the SPARCL trial data showed that the risk for hemorrhagic stroke increased with high-dose atorvastatin treatment after a recent stroke [16]. In particular, having a hemorrhagic stroke as the entry event increased the ICH risk, whereas the risk was not associated with the post-treatment LDL-C level, even for very low levels (< 40 mg/dL) [16]. In the German Diabetes and Dialysis Study involving patients with type 2 diabetes mellitus undergoing hemodialysis, more patients died of stroke in the atorvastatin group than in the placebo group [33]. However, in that study, the fatal stroke was mainly ischemic type; the incidence of fatal hemorrhagic stroke tended to be lower in the atorvastatin group compared with the placebo group [33]. In the present study, the incidence of hemorrhagic stroke was not associated with the LDL-C level or statin use in patients with LDL-C levels Ōēż 70 mg/dL.
Taken together, the present data suggest that post-statin hypocholesterolemia, regardless of the level, is not associated with the development of hemorrhagic stroke. Instead, an underlying condition may cause hemorrhagic stroke in subjects with a low LDL-C level. Nevertheless, in subjects with hemorrhagic stroke at initial presentation, less aggressive therapy may be considered when a statin is indicated [14].
The present study has limitations. First, the data were collected retrospectively from hospital records only; later cases of ICH might have been missed. Second, our results were usually based on a single LDL-C measurement, which can be different during acute versus subacute stages of illness [18]. A single-point study cannot reflect the stage of illness. Although we did not describe in detail the change in the LDL-C level in study subjects, the spontaneous group showed relatively consistent LDL-C levels, whereas statin users showed some variation according to drug compliance and other factors. Third, we used direct LDL-C measurement data, not LDL-C calculated using the Friedewald formula. This led to a significant selection bias because direct LDL-C measurements are not generally included in lipid profile reports. Most of our data were from internal medicine, neurology, neurosurgery, and rehabilitation departments. Fourth, the demographic and clinical data of subjects without ICH whose LDL-C level was between 41 and 70 mg/dL are not emphasized in the present study because our primary goal was to evaluate the causes and clinical characteristics of a very low LDL-C status.
In conclusion, in a hospital record review, we found that about 1% of subjects whose LDL-C levels were measured had an LDL-C level Ōēż 40 mg/dL. Additionally, about half of these subjects had a very low LDL-C level without a history of hypolipidemic therapy and had a high incidence of chronic systemic illnesses such as viral hepatitis and alcoholic liver disease. The incidence of ICH was not associated with statin therapy or the LDL-C level.
References
1. Cziraky MJ, Watson KE, Talbert RL. Targeting low HDL-cholesterol to decrease residual cardiovascular risk in the managed care setting. J Manag Care Pharm 2008;14(8 Suppl):S3ŌĆōS28PMID : 19891279.

2. Brunzell JD, Davidson M, Furberg CD, et al. Lipoprotein management in patients with cardiometabolic risk: consensus statement from the American Diabetes Association and the American College of Cardiology Foundation. Diabetes Care 2008;31:811ŌĆō822PMID : 18375431.


3. O'Keefe JH Jr, Cordain L, Harris WH, Moe RM, Vogel R. Optimal low-density lipoprotein is 50 to 70 mg/dL: lower is better and physiologically normal. J Am Coll Cardiol 2004;43:2142ŌĆō2146PMID : 15172426.


4. Stemmermann GN, Chyou PH, Kagan A, Nomura AM, Yano K. Serum cholesterol and mortality among Japanese-American men: the Honolulu (Hawaii) Heart Program. Arch Intern Med 1991;151:969ŌĆō972PMID : 2025146.


5. Iribarren C, Reed DM, Burchfiel CM, Dwyer JH. Serum total cholesterol and mortality: confounding factors and risk modification in Japanese-American men. JAMA 1995;273:1926ŌĆō1932PMID : 7783302.


6. Frank JW, Reed DM, Grove JS, Benfante R. Will lowering population levels of serum cholesterol affect total mortality? Expectations from the Honolulu Heart Program. J Clin Epidemiol 1992;45:333ŌĆō346PMID : 1569429.


7. Neaton JD, Blackburn H, Jacobs D, et al. Serum cholesterol level and mortality findings for men screened in the multiple risk factor intervention trial: multiple risk factor intervention trial research group. Arch Intern Med 1992;152:1490ŌĆō1500PMID : 1627030.


8. Noda H, Iso H, Irie F, et al. Low-density lipoprotein cholesterol concentrations and death due to intraparenchymal hemorrhage: the Ibaraki Prefectural Health Study. Circulation 2009;119:2136ŌĆō2145PMID : 19364982.


9. Stone NJ, Bilek S, Rosenbaum S. Recent National Cholesterol Education Program Adult Treatment Panel III update: adjustments and options. Am J Cardiol 2005;96(4A):53EŌĆō59E.

10. LaRosa JC, Grundy SM, Waters DD, et al. Intensive lipid lowering with atorvastatin in patients with stable coronary disease. N Engl J Med 2005;352:1425ŌĆō1435PMID : 15755765.


11. Johnson C, Waters DD, DeMicco DA, et al. Comparison of effectiveness of atorvastatin 10 mg versus 80 mg in reducing major cardiovascular events and repeat revascularization in patients with previous percutaneous coronary intervention (post hoc analysis of the Treating to New Targets [TNT] Study). Am J Cardiol 2008;102:1312ŌĆō1317PMID : 18993147.


12. Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 2005;366:1267ŌĆō1278PMID : 16214597.


13. Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004;350:1495ŌĆō1504PMID : 15007110.


14. Amarenco P, Labreuche J. Lipid management in the prevention of stroke: review and updated meta-analysis of statins for stroke prevention. Lancet Neurol 2009;8:453ŌĆō463PMID : 19375663.


15. Amarenco P, Bogousslavsky J, Callahan A 3rd, et al. High-dose atorvastatin after stroke or transient ischemic attack. N Engl J Med 2006;355:549ŌĆō559PMID : 16899775.


16. Goldstein LB, Amarenco P, Szarek M, et al. Hemorrhagic stroke in the stroke prevention by aggressive reduction in cholesterol levels study. Neurology 2008;70(24 Pt 2):2364ŌĆō2370PMID : 18077795.


17. Song JX, Ren JY, Chen H. Primary and secondary hypocholesterolemia. Beijing Da Xue Xue Bao 2010;42:612ŌĆō615PMID : 20957025.

18. Moutzouri E, Elisaf M, Liberopoulos EN. Hypocholesterolemia. Curr Vasc Pharmacol 2011;9:200ŌĆō212PMID : 20626336.


19. Levesque H, Gancel A, Pertuet S, Czernichow P, Courtois H. Hypocholesterolemia: prevalence, diagnostic and prognostic value: study in a department of internal medicine. Presse Med 1991;20:1935ŌĆō1938PMID : 1837087.

20. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2012;35(Suppl 1):S64ŌĆōS71PMID : 22187472.



21. Iribarren C, Jacobs DR, Sadler M, Claxton AJ, Sidney S. Low total serum cholesterol and intracerebral hemorrhagic stroke: is the association confined to elderly men? The Kaiser Permanente Medical Care Program. Stroke 1996;27:1993ŌĆō1998PMID : 8898804.


22. Yang X, So WY, Ma RC, et al. Low LDL cholesterol, albuminuria, and statins for the risk of cancer in type 2 diabetes: the Hong Kong diabetes registry. Diabetes Care 2009;32:1826ŌĆō1832PMID : 19592629.



23. Suh I, Jee SH, Kim HC, Nam CM, Kim IS, Appel LJ. Low serum cholesterol and haemorrhagic stroke in men: Korea Medical Insurance Corporation Study. Lancet 2001;357:922ŌĆō925PMID : 11289349.


24. Segal AZ, Chiu RI, Eggleston-Sexton PM, Beiser A, Greenberg SM. Low cholesterol as a risk factor for primary intracerebral hemorrhage: a case-control study. Neuroepidemiology 1999;18:185ŌĆō193PMID : 10364719.


25. Benn M, Tybjaerg-Hansen A, Stender S, Frikke-Schmidt R, Nordestgaard BG. Low-density lipoprotein cholesterol and the risk of cancer: a mendelian randomization study. J Natl Cancer Inst 2011;103:508ŌĆō519PMID : 21285406.


27. Vyroubal P, Chiarla C, Giovannini I, et al. Hypocholesterolemia in clinically serious conditions: review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2008;152:181ŌĆō189PMID : 19219206.


28. Barter P, Gotto AM, LaRosa JC, et al. HDL cholesterol, very low levels of LDL cholesterol, and cardiovascular events. N Engl J Med 2007;357:1301ŌĆō1310PMID : 17898099.


29. Woo D, Kissela BM, Khoury JC, et al. Hypercholesterolemia, HMG-CoA reductase inhibitors, and risk of intracerebral hemorrhage: a case-control study. Stroke 2004;35:1360ŌĆō1364PMID : 15087556.


30. Heart Protection Study Collaborative Group. MRC/BHF heart protection study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet 2002;360:7ŌĆō22PMID : 12114036.


31. White HD, Simes RJ, Anderson NE, et al. Pravastatin therapy and the risk of stroke. N Engl J Med 2000;343:317ŌĆō326PMID : 10922421.


Figure┬Ā1
Distribution of the low-density lipoprotein cholesterol (LDL-C) level in 34,415 subjects who were subjected to LDL-C measurement from January 2005 to March 2011. The numbers in parentheses indicate the percentage of the total subjects. Note that the LDL-C intervals on the x-axis are different for less frequent and extreme values.
Table┬Ā2
Demographic and clinical characteristics of very low LDL-C (Ōēż 40 mg/dL) patients on statin therapy and those without a history of hypolipidemic therapy (spontaneous group)

Table┬Ā3
Pearson's correlations between the LDL-C level and other parameters in all subjects with LDL-C Ōēż 40 mg/dL (n = 250)

Table┬Ā4
Pearson's correlations between LDL-C level and other parameters in subjects with LDL-C Ōēż 40 mg/dL and without a history of hypolipidemic therapy (n = 121)
Table┬Ā5
Incidence of intracerebral hemorrhage in subjects on statin therapy and those not treated with a cholesterol-lowering agent (spontaneous group)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



