INTRODUCTION
The gastrointestinal tract is the most common site of extranodal lymphomas, but the anorectum is not a common location for gastrointestinal lymphoma. Colorectal lymphoma comprised between 10% to 20% of the primary gut lymphomas in a large, published series1). Anorectal lymphoma is so rare that its frequency among the cases of gastrointestinal lymphoma has been reported as only 4~6% in North America and 2% in Japan2). For patients suffering with acquired immune deficiency syndrome (AIDS), there is a marked increase in gastrointestinal non-Hodgkin's lymphomas (NHL); these are of a higher histologic grade and they originate from B-cells, they are more aggressive and are primary of an extra nodal origin3, 4).
The relative frequency of peripheral T-cell lymphoma (PTCL) of NHL in the gastrointestinal tract is about 2% in Korea5). Most lymphomas with a primary or major location in the anorectum have been reported in AIDS patients; however, peripheral T-cell lymphoma, unspecified (PTCLu) of the rectum is quite rare3, 6). Therefore, rectal PTCLu in non-AIDS patient seems to be an extremely uncommon finding. Moreover, concurrent extranodal involvement of both the stomach and anorectum is also very rare. In some limited studies, PTCLu in the colon has manifested as either a diffuse or focal segmental lesion with extensive mucosal ulceration6, 7). We report here on an unusual case of concurrent rectal and gastric PTCLu without extensive mucosal ulceration in a non-AIDS patient.
CASE REPORT
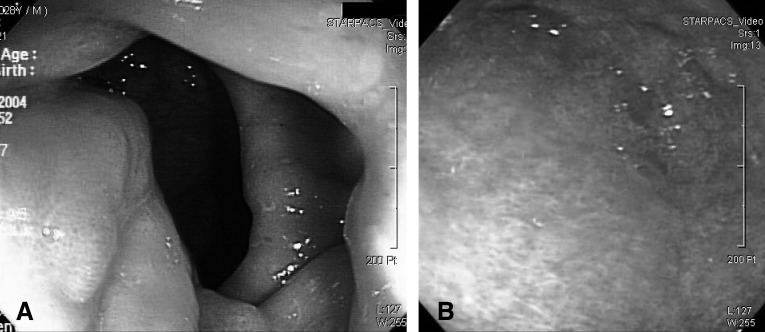
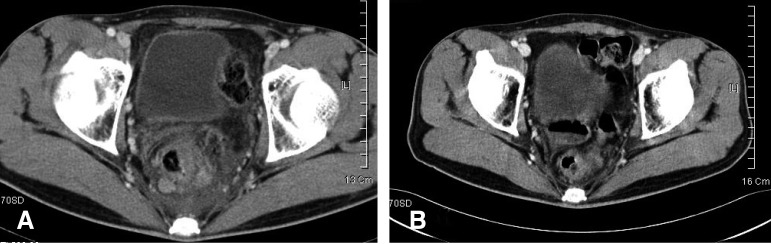
A 29-year-old man was referred to our hospital with complaints of hematochezia and tenesmus for 2 months. He underwent colonofiberscopic examination without biopsy by a primary clinician. The colonofiberscopic examination showed a huge multiple fungating submucosal mass at the anorectal region (Figure 1A). On admission, the patient complained hematochezia, tenesmus and abdominal discomfort, but there was no specific findings noted on the abdominal physical examination. The vital signs revealed a blood pressure of 100/70 mmHg, a pulse rate of 68/min, a respiratory rate of 22/min and his temperature was 36.5℃. The laboratory test showed a white blood cell count of 9,110/µL (84.1% polymorphonuclear cells and 9.2% lymphocytes), a hemoglobin level of 12.3 g/dL, a hematocrit of 36.8%, a platelet count of 412,000/µL, an erythrocyte sedimentation rate of 10 mm/hour and a C-reactive protein level of 0.03 mg/dL (normal range: 0-0.5). The partial thromboplastin time and prothrombin time were within normal ranges. The results of liver function tests were as follows protein 8.7 g/dL, albumin 4.5 g/dL, bilirubin 1.6 mg/dL, aspartate transaminase 26 IU/L, alanine transaminase 53 IU/L, alkaline phosphatase 51 IU/L, cholesterol 138 mg/dL and lactic dehydrogenase (LDH) 514 IU/L. The renal function tests were normal. The serum electrolytes showed sodium 138 mM/dL, potassium 4.9 Mm/L, chloride 100 mM/L, calcium 9.2 mg/dL and phosphate 4.8 mg/dL. Urinalysis was negative for occult blood and there was a trace of protein. No antibodies against syphilis, hepatitis B, hepatitis C or human immunodeficiency virus were detected. Under the clinical impression of anal and rectal submucosal tumor, the patient underwent excisional biopsy. The histologic findings of the specimen showed dense infiltration of angulated lymphoid cells into the lamina propria together with perivascular infiltrations (Figure 2A). On immunohistochemistry, these lymphoid cells were positive for CD3 (Figure 2B), lymphocyte common antigen, Ki-67 and CD45RO, but they were negative for CD20 and CD56. These morphologic features and the immunophenotype profiles were compatible with PTCLu. For further evaluation, he underwent gastrofiberscopy and bone marrow biopsy. Gastrofiberscopic examination showed mottled or patched hyperemic mucosa and round-shaped, flat elevated lesions on the gastric antrum (Figure 1B). Biopsy of the gastric lesion gave the same diagnosis, but there was no lymphoma that involved the bone marrow. The chest CT scan was normal; the abdominal-pelvic CT scan showed wall thickening from the lower rectum to the rectosigmid junction, and there were multiple lymphadenopathies in the perirectal area, sigmoid mesocolon and paraaortic area, and enlarged nodes were noted along the hepatoduodenal ligament.
Finally, the patient was diagnosed as suffering with rectal and gastric PTCLu. The Ann Arbor stage was IVA and the age-adjusted international prognostic index was 3. He was in the high risk group. He was treated with six cycles of combination chemotherapy including cyclophosphamide, adriamycin, vincristine, etoposide and prednisone (CHOEP). After two cycles of chemotherapy, the initially observed rectal wall thickening disappeared on the follow-up abdominal-pelvic CT scan; we observed markedly decreased size of the lymph nodes of the paraaortic and portocarval space. On follow-up gastrofiberscopic biopsy, a few lymphoid cells had infiltrated to the lamina propria, and these were probably reactive lymphocytes. He underwent autologous stem cell transplantation after six cycles of systemic CHOEP chemotherapy. Regular follow-up was done with CT, gastroscopic biopsy and colonofiberscopic biopsy every 3 months. He has been in complete remission for over 300 days after autologous stem cell transplantation.
DISCUSSION
Lymphomas arising primarily in the rectum are rare occurrences that represented only 0.1~0.3% of all malignant rectal lesions according to the study data from a large population8). This is probably because usually there is no lymphoid tissue in the anorectum. Anorectal and perianal lymphoma occurs in an even smaller subset of patients3, 9, 10). There are now increasing numbers of reported cases of anorectal lymphomas that were caused by acquired immunodeficiency syndrome3, 4, 9, 11, 12). Most cases have been homosexual men, suggesting anorectal trauma as a port of entry for a possible infectious, oncogenic virus11). Primary anorectal lymphoma in nonimmunocompromised and heterosexual patients is a rare event with only a few isolated cases being reported10, 13). Most primary gastrointestinal lymphomas have been of a B-cell origin. Only a few cases are reported to be positive for T-cell markers6, 14). The most common site of involvement for gastrointestinal T-cell lymphoma is the small bowel, and other sites are exceptionally rare.
PTCLu is rather uncommon; it comprises approximately 9.4% of all NHLs, but it is more common in East Asia5). PTCL has been subclassified in various ways and usually to include anaplastic large T-cell lymphoma, angioimmunoblastic T-cell lymphoma (AILD), PTCLu and intestinal T-cell lymphoma (ITCL). The PTCLu group showed the second highest response rate, but also the highest relapse rate, and this suggests that while responses are good, they are somewhat brief in duration15). Lopez Guillermo et al.16) concluded that the outcome for PTCLu patients was basically unfavourable with less than half reaching complete remission and mostpatients eventually relapsed. It is generally assumed that PTCLu probably constitutes a heterogeneous group encompassing several different subtypes that are currently not recognized, and this may partly account for the variation in the findings. According to the recent literature, PTCL involving the colon corresponds to a diffuse ulcerative form of colorectal lymphoma, although this is very rare17). The radiologic differentiation of PTCL involving the colon from inflammatory bowel disease is very difficult. Moreover, pathologic differentiation of PTCL from inflammatory bowel disease is also difficult18, 19). In this case, colonofiberscopic examination showed a huge multiple fungating submucosal mass at the anorectal region, and the gastrofiberscopic examination showed mottled or patched hyperemic mucosa and round-shaped, flat elevated lesions on the gastric antrum. These are unusual findings for PTCL involving the colon.
A case of intestinal T-cell lymphoma involving the rectum was previously reported in Korea6). However, although the T-cell markers were expressed, the exact subtype was not defined and whether the patient was non-AIDS or not was not clearly mentioned in that case. Another difference of that previous case was the involvement of the entire gastrointestinal tract from the esophagus to the rectum rather than skipping anorectal and gastric involvement, which was true in seen in our case. Our case had some characteristic findings. The patient showed concurrent anorectal and gastric involvement, he was not an AIDS patient, and there were no extensive ulceration in spite of PTCLu.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





