INTRODUCTION
The pathogenesis of GravesŌĆÖ disease is largely unknown but the characteristic perturbation of the immune network points to an organ specific autoimmune disorder (1ŌĆō4). Various methods (3, 5ŌĆō8) have been developed to detect autoantibodies to the thyrotropin receptor (TRAb) and these autoantibodies are thought to be one of main mechanisms to explain the hyperthroidism in GravesŌĆÖ disease.
Thyrotropin binding inhibiting antibody (TBIAb) is now generally accepted as an autoantibody to the thyrotropin receptor (9, 10) and that at least a portion of this antibody can stimulate thyroid function, even though there have been several examples of its nonspecific thyroid membrane binding (11) along with its occurrence in HashimotoŌĆÖs disease, primary myxedema and other thyroid disorders (12ŌĆō14).
Clinical correlation of TBIAb to various indices of GravesŌĆÖ disease and its significance in diagnosis and treatment is controversial (15ŌĆō18). We tried to clarify the nature of TBIAb in GraveŌĆÖs disease especially its correlations to clinical, thyroid functional indices and thyroid specific autoantibodies since TBIAb by itself is a thyroid related autoantibody.
MATERIALS AND METHODS
1. Subjects
The present study involved 192 patients with GravesŌĆÖ disease (30 males, 87 females, ranging in age from 10ŌĆō75 years), of whom 117 were untreated, 49 were on antithyroid medication and 26 were in remission for at least 6 months. Thirty two normal controls and 77 patients with HashimotoŌĆÖs disease were also included.
The diagnosis of GravesŌĆÖ disease was based on the clinical and laboratory features of hyperthyroidism with or without exophthalmos and dermopathy and increased thyroid uptake of 99m-Tc04. Patients who presented a rubbery and/or nodular thyroid, hypo or euthyroid with positive (above 1:100) thyroid microsomal and/or thyroglobulin antibodies were classified as HashimotoŌĆÖs disease.
2. Methods
Only the data obtained on the same date and under the same conditions on each patient were include in this study.
Thyroid mass on initial examination which was calculated from the surface area and the long axis of both lobes, was measured from the computerized image of the 99m-Tc-thyroid scan, and was graded into 5 groups (from 15 grams to more than 55 grams: 10 gram scale).
TBIAb was determined using solubilized porcine thyroid membrane (3). 50 ul of IgG fraction was added to 100 ul of solubilized thyroid membrane (4mg/ml) for 15 min at room temperature. 125I-TSH in 100 ul of tris-NaCl-BSA (1mg/ml) was added and incubated for another 60 min at 37┬░C. The volume of the reaction mixture was made up to 0.8ml with tris-NaCl-BSA, then 1000 ul of PEG solution was added. After mixing well, the tubes were centrifuged and the pellet containing receptor bound labelled TSH was counted for 125I. Determination of nonspecific binding was carried out by replacing soluble receptors with 1% Lubrol solution in the reaction mixture. Results were expressed as percent inhibition of labelled TSH binding calculated as follows,
Thyroid function tests were done by radioimmunoassay (TSH; Abbott, USA, T3RU, T4, T3; Corning, USA) and antimicrosomal antibody, a passive hemagglutination method (Fuji Zoki, Japan).
RESTULS
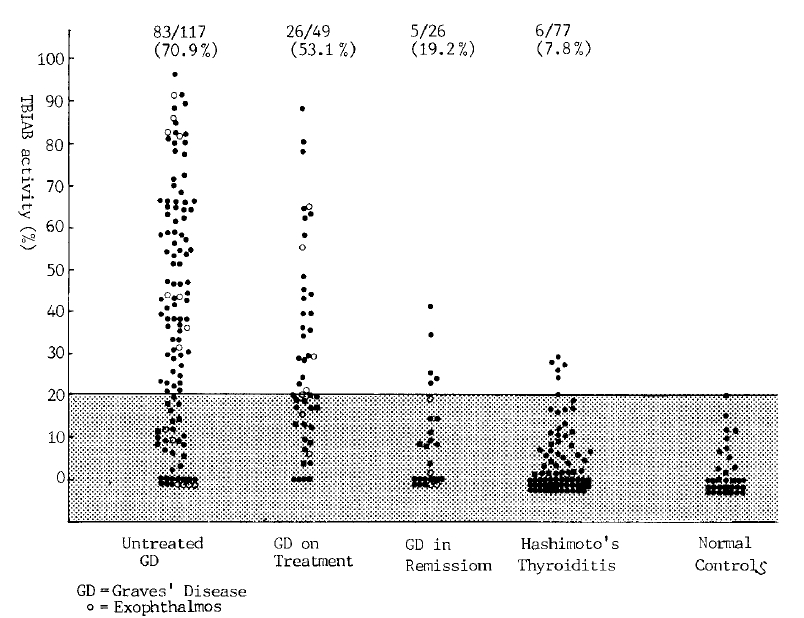
1. Incidence of TBIAb in GravesŌĆÖ and HashimotoŌĆÖs diseases (Fig. 1)
The range of TBIAb activity in 32 normal controls was 0% to 19.8% (data below 0% were regarded as 0% inhibition) and we interpreted the data above 20% inhibition as positive.
Among the untreated GraveŌĆÖs patients, 83 of 117 cases had detectable TBIAb activity (70.9%). The incidence of positive TBIAb in GravesŌĆÖ patients on antithyroid medication regardless of their thyroid function decreased to 53.1% (26 of 49 cases) and of the 26 patients in remission for at least 6 months, 19.1% (5 cases) tested positive.
In HashimotoŌĆÖs disease, 6 of 77 patients (7.8%) had weakly positive results.
2. Correlation of TBIAb to Various Clinical Indices
There were no correlations between TBIAb and age, sex, onset age or disease duration. Also patients presenting exophthalmos and/or dermopathy, or periodic paralysis were no different from patients without them.
But TBIAb and goiter size showed a significant correlation (r = 0.95, p<0.005), as the larger the goiter, the greater the TBIAb activities (Fig. 2).
3. Correlation of TBIAb to Various Thyroid Functional Indices
Only the patients who were not on antithyroid medication for at least 6 months were included in these correlations.
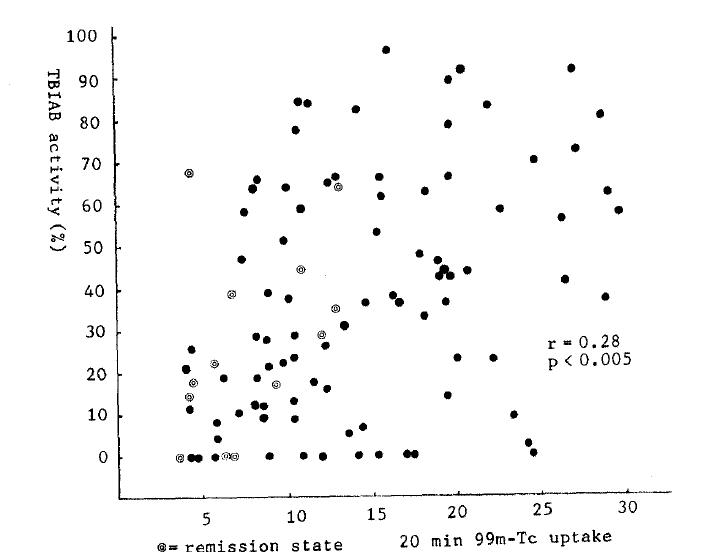
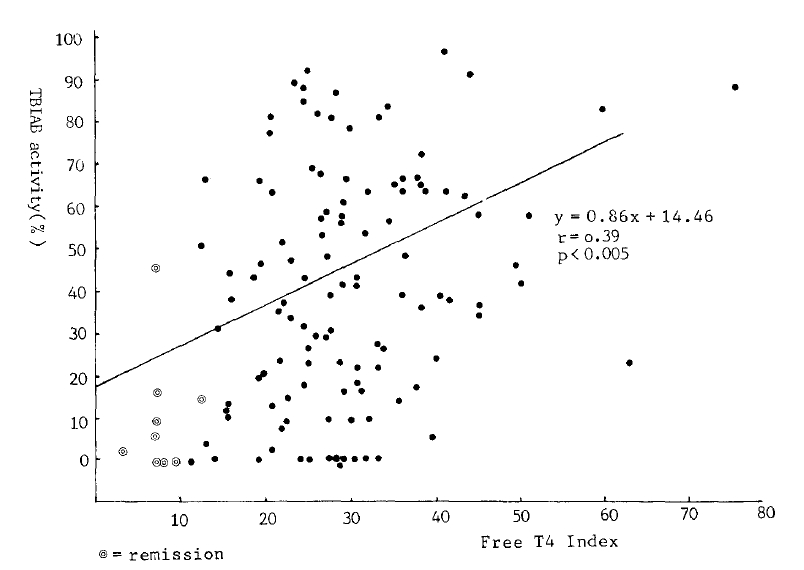
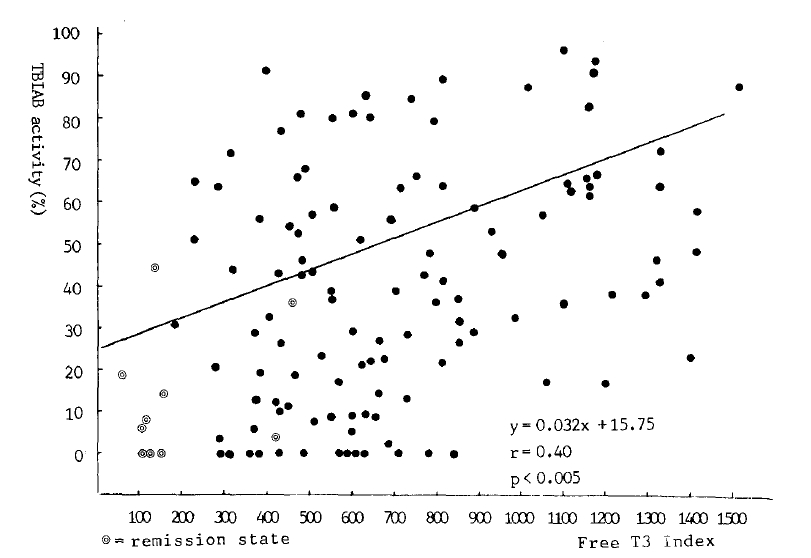
In 87 untreated patients and 12 patients in remission, TBIAb activities were significantly correlated with the values of 20 min 99m-Tc-thyroid uptakes (r = 0.28, p<0.05, Fig. 3). More significant correlations were noticed between TBIAb and the free T4 index (r = 0.39, p<0.005, Fig. 4) in 114 new and 9 remission state GravesŌĆÖ patients, and the free T3 index (r = 0.40, p<0.005, Fig. 5) in 115 fresh and 10 in remission. But there was no correlation to thyroglobulin titers.
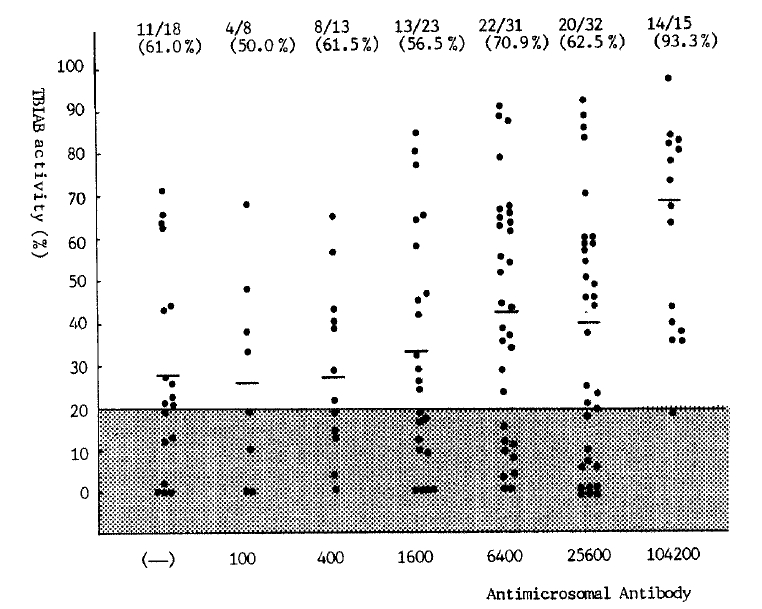
4. Correlation of TBIAb of Antimicrosomal Antibody Activity (Fig. 6)
There were no correlations between TBIAb and the groups who showed rather low antimicrosomal antibody activity. But patients with strongly positive antimicrosomal activity (above 1:102400) had a high TBIAb positiveness and its activity.
DISCUSSION
A number of the humoral and cellular mechanisms of GravesŌĆÖ disease have been discovered.1ŌĆō4) Among them, TRAb has received considerable attention because of its presumed action to lead clinical expression as hyperthyroidism. Still, the role of TBIAb in GravesŌĆÖ disease is not clear, except there is general agreement that these antibodies are directed to TSH receptors on follicular cells9ŌĆō10) and might be heterogenous12ŌĆō13).
In our study we tried to clarify the nature of TBIAb, especially its correlation to clinical, thyroid functional indices and other thyroid specific autoantibodies.
In untreated GravesŌĆÖ patients, the incidence of TBIAb was 70.9% which decreased to 53.1% in the treated patient group and 19.2% in the remission group. Also we noticed a 7.8% positivity in HashimotoŌĆÖs disease which has been discussed elsewhere.16)
Among the clinical indices, remarkable correlation (r = 0.95) was noticed between TBIAb and goiter size. The correlation might be reasonable since it has been suggested that lympthocytes within the thyroid are the major producer of thyroid autoantibodies,19) and TBIAb by itself may act as a growth promoting factor of thyrocytes.13ŌĆō14) Also TBIAb activity is known to be reduced especially after ablation therapy.15)
There have been various studies on the correlation of TBIAb and thyroid functional indices.15ŌĆō18) Mukhar et al.15) and Endo et al.16) found that TBIAb activities correlated only with thyroid uptake of 131I or 99m Tc04ŌłÆ in a rather small number of samples. However, while our data showed low correlation between TBIAb and 99m-Tc-thyrid uptake (r = 0.28), it revealed a more significant correlation to free T4 and T3 indices (r = 0.39, 0.40 each) which suggests that at least some portion of these antibodies can stimulate thyroid function in GravesŌĆÖ disease.
On the correlation of TBIAb to other thyroid specific autoantibodies, it was found that the patients with strongly positive antimicrosomal activity (above 1:102400) exhibited a high degree of TBIAb positiveness and also activity, but the patients with low titers of antimicrosomal antibody didnŌĆÖt show any correlation. The first plausible explanation for the relationship is that the patient group with strongly positive antimicrosomal activity included many cases of Hashitoxicosis, a combination disorder of GravesŌĆÖ and HashimotoŌĆÖs disease, since high titers of autoantibodies and TBIAb positiveness are the prerequisites for diagnosis of Hashitoxicosis. The other explanation, if we assume that the present scale of antimicrosomal antibodies is very narrow in the biological sense, is that the inherent immunological network would be quite different among GravesŌĆÖ patients. This is consistent with the data which showed islet cell surface antibodies, as organ nonspecific antibodies in GravesŌĆÖ disease, positive and TBIAb positive group showed a higher percentage of positiveness in antimicrosomal antibodies than those of the GravesŌĆÖ patient groups without ICSA positiveness, which also implies possibly different clinical courses.20)
In conclusion, TBIAb activities were correlated to goiter size and various thyroid functional indices (Tab. 1), suggesting its role in the manifestation, but other clinical features including exophthalmos didnŌĆÖt have any correlation to regard TBIAb as just one of the immune effector arms in GravesŌĆÖ disease.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





