INTRODUCTION
Somatostatin is a potent inhibitor of secretion of the GH in patients with acromegaly1ŌĆō8). However, the short half-life of somatostatin as well as the rebound hypersecretion of growth hormone that occurs after its infusion, have rendered the native tetradecapeptide impractical for therapeutic use2).
Efforts have been directed toward developing somatostin analogues with a longer and more specific effect9ŌĆō10). Bauer et al11) finally found an octapeptide which they called selective mini-somatostatin (SMS 201-995; Sandoz, Bases, Switzerland). The subcutaneous administration of a single 50 ╬╝g dose of SMS 201-995 to patients with acromegaly has been shown to have a long-acting inhibitor effect on plasma growth hormone without producting rebound hypersecretion12ŌĆō13). Thereafter SMS 201-995 has been reported to be an useful adjunctive therapeutic drug in acromegalic patients14), but the adequate dosage and frequency of administration were variable.
A possible adverse effect of SMS 201-995 is the suppression of insulin and glucagon secretion. Several reports demonstrated no diabetogenic effect of SMS 201-99515ŌĆō17).
Most of acromegalic patients shows a paradoxical response to TRH or LRH. Somatostatin suppressed the response in some of them18) although the mechanism has not been known. However, the effect of SMS 201-995 on the paradoxical response has not been demonstrated so far. Therefore, we have tested the effectiveness and the duration of action of SMS 201-995 as a suppressor of serum GH. Also, we have tested the effects on blood glucose, secretion of insulin and glucagon, and the paradoxical response.
METHODS
1. Subjects
Five patients with acromegaly, one male and four females, were studied. All the patients had clinical manifestations, such as acral enlargement of extremities, excessive perspiration, and radiologic evidence of a pituitary tumor. The duration of symptoms was in the range of 4ŌĆō13 years. One patient (patient 1) had undergone a transsphenoidal adenomectomy followed by bromocriptine. Two patients (patient 2, 4) had had radiation followed by bromocriptine, and one patient (patient 3) had received only bromocriptine. One patient was a new case who had not received any treatment prior to the study (Table 1).
2. Study Design
All patients discontinued medication at least two weeks before the present study. On the first morning, 200 ╬╝g of thyrotropin releasing hormone (TRH) was administered intravenously to observe the paradoxical response of GH. Venous blood was obtained every thirty minutes via inserted antecubital cannula.
During the second day, all the patients were injected subcutaneously with 0.5 ml of normal saline as a placebo thirty minutes before breakfast, then their diurnal variation of GH secretion was measured every two hours for up to twenty-four hours in order to use those data as a reference.
On the third day, all the patients were injected the 50 ╬╝g of SMS 201-995 with the same method and measured the GH concentration at the same time. In addition, the levels of glucose, insulin, and glucagon were measured every hour for up to four hours after the injection of SMS 201-995 which were compared to those of the control group consisting of four healthy young men.
On the last day, four patients were injected the 50 ╬╝g of SMS 201-995 one hour before TRH injection in order to determine the influence of SMS 201-995 on the paradoxical response to TRH.
3. Materials and Laboratory Method
Plasma levels of growth hormone, insulin and glucagon were determined by radioimmunoassay kit purchased from Radioassay System Laboratories, INC., Dinabbot Co. and Daiichi Co. respectively.
SMS 201-995 was kindly suppled by the Sandoz pharmaceutical, Inc. in Seoul, Korea. TRH was purchased from Hoechst pharmaceuticals.
RESULTS
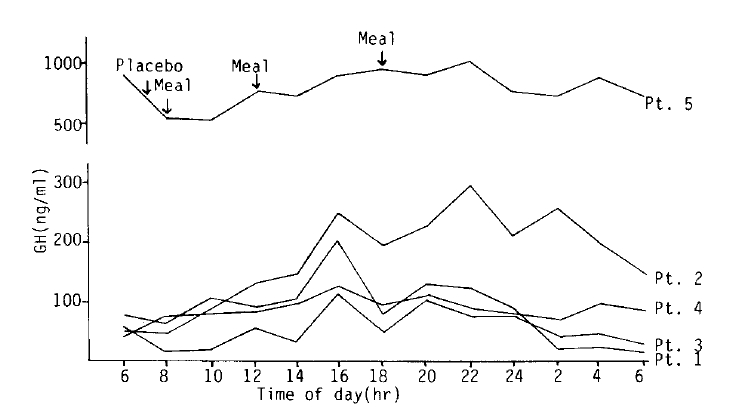
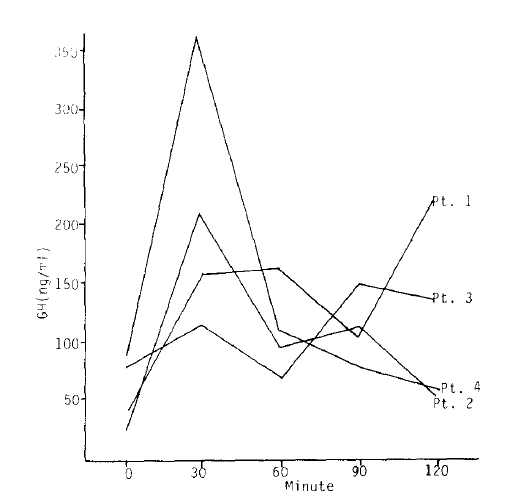
The absence of diurnal variation after injection of normal saline as a placebo was shown in all patients (Fig. 1).
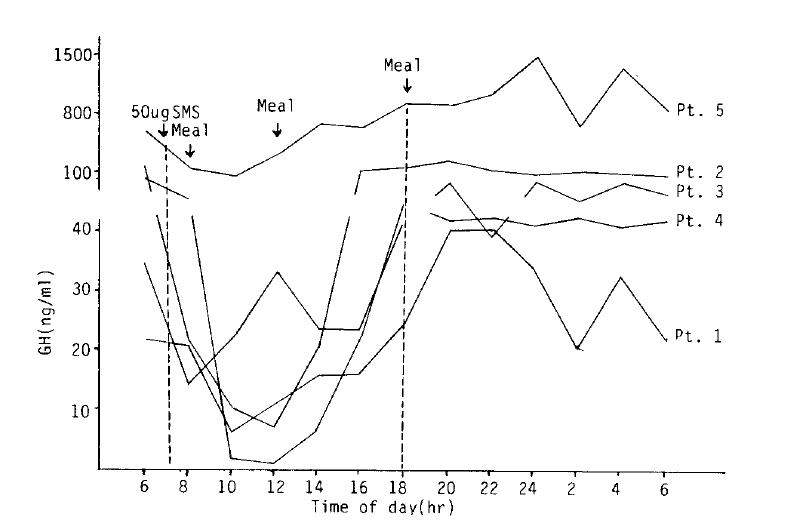
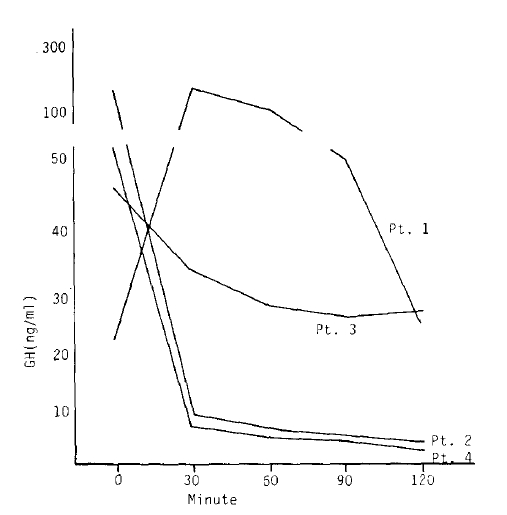
A single dose of 50 ╬╝g SMS 201-995 could suppress the GH level maximally by 85% on average which was noticed at about 4 hours after administration. The suppressive effect gradually decreased thereafter and the GH level returned to that of preinjection period at 8 to 10 hours after injection without rebound phenomenon (Fig. 2).
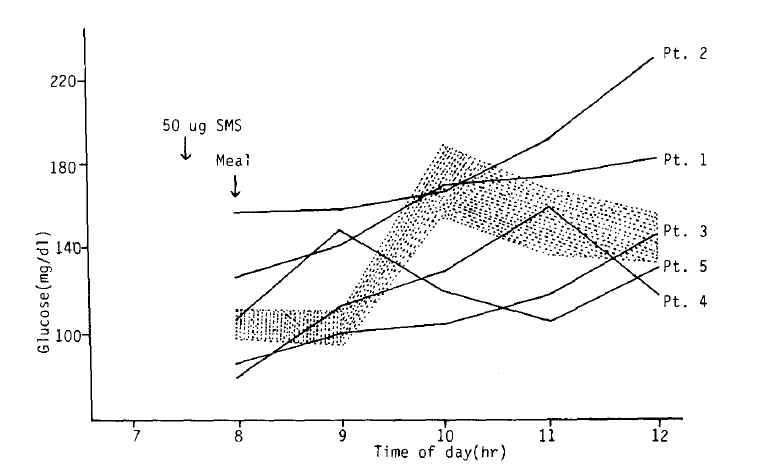
The figure 3 shows the changes in glucose levels during the 4 hours period after the injection of SMS 201-995. The glucose levels of four healthy males reached a maximum 2 hours after breakfast and decreased thereafter shown in the shaded area; however, the glucose levels of four patients (patient 1, 2, 3, 4) continued to increase even after three hours after breakfast.
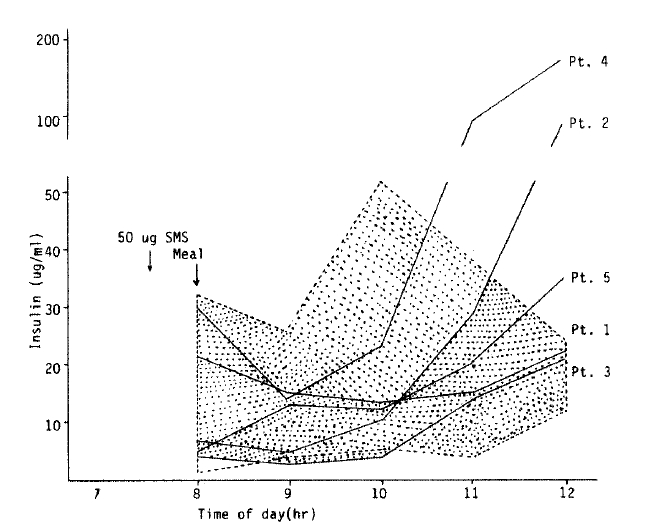
The changes of serum insulin concentration after administration of SMS 201-995 are shown in Fig. 4. The insulin levels of healthy males reached their peak at 2 hours after breakfast and declined thereafter according to the changes of blood glucose level. However, the insulin levels of the patients continued to increase for up to 4 hours after breakfast showing a delayed response similar to the changes of blood glucose levels.
Only one patient (patient 5) among four patients who had their glucagon levels measured showed an adequate decrease in accord with the change of blood glucose after breakfast, and the other three patients showed no definite declining pattern. In general, the changes of glucagon level did not show any significant difference compared with those of healthy males (Fig. 5).
The paradoxical responses of the GH to TRH were apparent in patients 1, 2, and 4 (Fig. 6). However, the paradoxical responses disappeared in two (patients 2 and 4) of them with SMS 201-995 injected one hour before TRH stimulation.
The two patients (2, 4) whose paradoxical response had been more prominent showed more marked suppression of the response with SMS 201-995 (Fig. 7).
DISCUSSION
Somatostatin is produced by processing a larger precursor molecule and exists in both the 28- and 14-amino acid forms. 28-amino acid somatostatin has a longer half-life and is a more potent inhibitor of GH and insulin secretion. It is found in man and seems particulary prevalent in the gastrointestinal tract.
Somatostatin-14 has a greater affinity for hypothalamic and cortical receptors and is more potent in the inhibition of glucagon release. When pharmacological doses of somatostatin 14 are infused, the effects include inhibition of endocrine secretions (growth hormone, thyroid-stimulating hormone, insulin, glucagon, and all the gut hormones), exocrine secretions (gastric acid, pancreatic bicarbonate, and enzymes), small bowel absorption, and depression of splanchnic blood flow.
Somatostatin-14 has two main drawbacks as a potential drug. First, its actions are non-specific and its long-term use is likely to result in adverse effects such as intestinal malabsorption and glucose intolerance. Trials have therefore been made to synthesize analogues that are more specific for individual effects. The second drawback of somatostatin-14 is its short plasma half-life; this means that it must be given continuously by intravenous infusion. Rebound effects, such as severe flushing in patients with carcinoid syndrom, are sometimes seen a few minutes after the infusion has stopped.
Some somatostatin analogues have a selective effect on GH secretion15ŌĆō17) and others have a prolonged action19ŌĆō20), but the effect of SMS 201-995 on serum GH is selective and lasts for up to 9 hours.
The intravenously administered natural somatostatin has a half-life of 2 to 3 minutes whereas the SMS 201-995 has an elimination half-life of 41 to 58 minutes. In addition, SMS 201-995 has been shown to be 45 times more potent than natural somatostatin in suppressing growth hormone release21).
Preliminary clinical trials have confirmed the pharmacological profile of this analogue. Plewe et al12) reported decrease in plasma growth hormone concentrations to normal or nearly normal levels that lasted for 9 hours in a group of subjects with acromegaly who received a single subcutaneous injection of 50 ╬╝g of SMS 201-995.
In this study, we observed the effect of a single subcutaneous injection of 50 ╬╝g SMS 201-995 on five resistant acromegalic patients. Plasma GH level decreased maximally by 85% but the GH levels were not reduced to the normal range. It suggested that more than 50 ╬╝g of SMS 201-995 as one dose should be necessary for sufficient suppression of GH.
The duration of the suppressive effect was about 8 hours, which was comparable to those of others. It indicated that SMS 201-995 should be administered every 8 hours subcutaneously in order to maintain the continuous suppression of growth hormone secretion.
Unlike natural somatostatin, SMS 201-995 had only a slight and short-lasting inhibition effect on insulin secretion. In normal subjects, the administration of 10 to 20 ╬╝g of SMS 201-995 per hour intravenously and 50 to 100 ╬╝g subcutaneously was found to inhibit growth hormone, glucagon, and insulin elevations induced by an arginine infusion22). However, when an arginine infusion was done three hours after the injection of SMS 201-995; growth hormone did not increase markedly above basal levels, although the normal rise in insulin levels did occur. Steven WJ et al14) found that the diabetogenic effect of SMS 201-995 decreased toward the end of the long-term treatment period. This would have been caused by a diminished inhibition effect on insulin secretion during long-term treatment with the drug, but it also might have been due to the beneficial effect on carbon-hydrate tolerance by the reduction of plasma growth hormone level.
We also tried to determine whether SMS 201-995 had diabetogenic effect in this study.
Serum levels of glucose increased to reach their peak at about two hours after meal in healthy male controls acompanied by the simultaneous increase of serum insulin levels. In four acromegalc patients, a delayed increase of serum glucose occurred with delayed increased serum insulin. This delayed increase of serum glucose intolerance is believed to be caused by growth hormone excess in acromegaly rather than the suppressive effect of insulin by SMS 201-99523,24). In fact, many reports proposed postreceptor defect caused by growth hormone excess as a possible pathogenetic mechanism for the glucose intolerance in acromegaly. If SMS 201-995 suppressed the insulin secretion, the serum glucose would have been increased markedly above normal range at about two hours after meal. Furthermore, the fact that serum insulin level increased in accord with the increase of glucose level under the condition which GH was suppressed maximally suggests that SMS 201-995 has no significant suppressive effect on insulin secreation. Because our results cannot exclude the diabetogenic of SMS 201-995 when it is administered in more high dosage or for a longer duration, long-term follow-up study should be needed in the future.
SMS 201-995 also seems to have no significant suppressive effect on the glucagon secretion in small dosage and short term administration. The paradoxical responses of GH to TRH or LHRH are frequently observed in acromegalic patients. However, the mechnism is unknown. To our knowledge, there have been no reports which observed the paradoxical response of GH to TRH or LHRH after SMS 201-995 injection. In this study, two of three patients who had paradoxical response showed the disappearance of the response after administration of SMS 201-995, but the fact that the paradoxical response to TRH and the suppression of GH secretion by somatostatin occur via the adenylate cyclase system25) might explain the mechanism for the suppression of paradoxical response. The paradoxical responses had been more prominent in the two patients (patient 2, 4) whose responses were more markedly suppressed by SMS 201-995 compared to patient 1. This suggests the possibility that the patients might have a deficiency of somatostatinergic activity which caused the paradoxical response. Nevertheless, more studies are mandatory concerning the mechanism of paradoxical response and the effect of SMS 201-995 on the response.
In conclusion, the somatostatin analogue SMS 201-995 can be a candidate for the adjunctive therapy to the medical management of acromegaly. Its long duration of action at relatively low doses without rebound phenomenon are suitable for clinical use, but our data suggests that more than 50 ╬╝g SMS 201-995 should be required at least 3 times a day for the adequate suppression of GH. SMS 201-995 does not suppress the insulin and glucagon secretion in low dosage and short term treatment. Further evaluation should be needed for the suppressive effect on the paradoxical response.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





