 |
 |
| Korean J Intern Med > Volume 12(2); 1997 > Article |
|
Abstract
Candidiasis has emerged as the most common opportunistic fungal disease over recent decades owing to the progressively increasing use of antibiotics, immunosuppressive and cytotoxic drugs, indwelling foreign bodies and organ transplantation. Hematogenous candida endophthalmitis, which has a characteristic finding of single or multiple fluffy white cotton ball-like chorioretinal lesions often extending into vitreous, is the most fulminant manifestation of systemic candidiasis and may result in blindness. These ocular lesions can be easily recognized on funduscopic examination and should be serially looked for in all patients with known and suspected systemic candidiasis.
We report hematogenous candida endophthalmitis in a patient of systemic candidiasis and, to our knowledge, it is the first reported case in our country.
Candida albicans is the most common cause of disseminated fungal disease in severely ill patients and in those who are immunocompromised, and who are hospitalized1ŌĆō3). Hematogenous fungal endophthalmitis usually has an insidious onset with a slow loss of vision due to choroiditis and/or retinitis that can spread to vitreous. Candida species are the most common cause of endogenous fungal endophthalmitis, and endogenous Candida endophthalmitis is an early clinical indicator of disseminated candidiasis4ŌĆō5). The increasing incidence of systemic candidiasis, high incidence of endogenous Candida endophthalmitis and devastating visual consequences emphasize the importance of ophthalmoscopic examinations in patients with suspected systemic candidiasis6ŌĆō8).
We report the first case of hematogenous Candida endophthalmitis in a patient of systemic candidiasis, after intravenous hyperalimentation via the femoral line, in our country.
A 34-year-old woman was admitted to our hospital because of dysphagia, ocular pain and blurred vision.
One month earlier, she swallowed acetic acid accidentally and corrosive esophagitis developed. She visited a hospital where a femoral line was inserted and intravenous hyperalimentation was started. Two weeks later, she had fever, chill, ocular pain and blurred vision. She was treated with several broad-spectrum antibiotics without improvement, and so she was referred to our hospital.
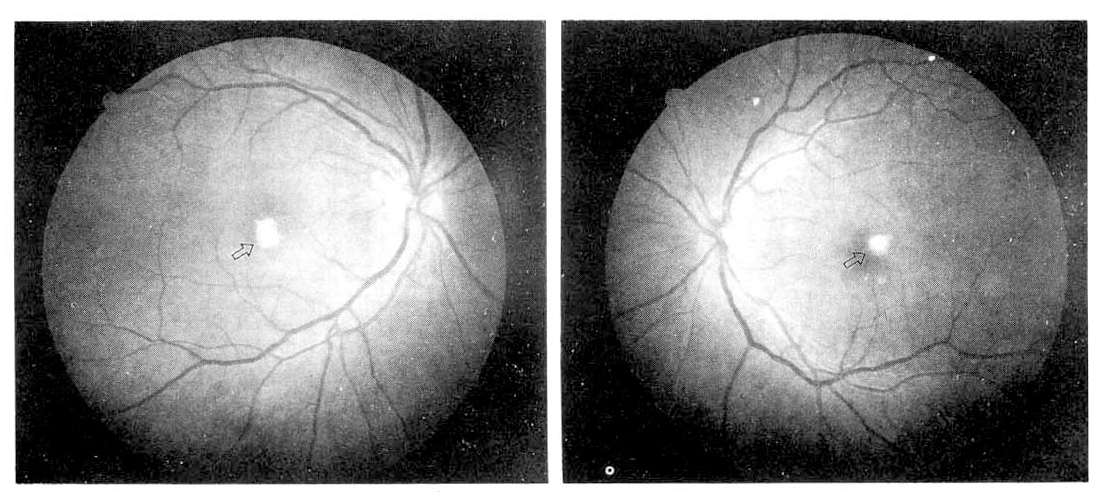
On admission to our hospital, she was acutely ill-looking and body temperature was 38.5┬░C. On laboratory examinations, hemoglobin was 9.3g/dl, hematocrit 26.2%, leukocyte 13,600/mm3 (neutrophil 84.7%, lymphocyte 5.2%, monocyte 6.8%, eosinophil 2.2% and basophil 1.1%) and platelet 216,000/mm3. Bacterial culture of blood, sputum and urine were negative. The fungus cultures of blood and femoral catheter tip were positive for Candida albicans. The ophthalmologic examination revealed visual acuity of 20/200 OD and 20/100 OS. Intraocular pressure OU was normal. The anterior chambers of both eyes were clear. The vitreous of both eyes was slightly hazy with some inflammatory cells. Fundus examination revealed white granulomatous mound on the macula of both eyes (Fig. 1).
Two weeks of intravenous fluconazole 200mg/day could neither control fever nor improve the visual acuity and general condition. So, we replaced the fluconazole with amphotericin B 20mg/day. After four weeks of treatment, fever was controlled and the general condition was improved, but improvement of visual acuity was not prominent. She was discharged with oral fluconazole 200mg/day for 2 weeks. Three months later after discharge, there was no further improvement of visual acuity and no interval change of previously noted fundal lesions.
To our knowledge, this is the first reported case of hematogenous candida endophthalmitis in a patient of candidemia in our country. The incidence of systemic candidiasis has increased in patients on intravenous hyperalimentation, hemodialysis and antibiotic therapy, in intravenous heroin abusers, in immunocompromised patients and after major surgery1ŌĆō3). Candida endophthalmitis is an important metastatic manifestation of disseminated candidiasis. In 1943, Maiale9) reported the first case of hematogenous spread of candida to the eye, and it has been shown that, at least, 78% of patients with candida endophthalmitis have disseminated candidiasis, and that candida endophthalmitis is likely to occur in 28ŌĆō45% of patients with candidemia4).
The most common ocular symptoms of candida endophthalmitis are pain, photophobia and blurred vision. The earliest fundus lesion is a chorioretinitis which is presented as small yellow-white lesions. Extension of the infection and associated inflammatory response into the vitreous body produces the more typical lesions described as ŌĆśpuff ballsŌĆÖ, which can be easily recognized on funduscopic examination6, 10). Although spontaneous resolution of candida endophthalmitis has been reported, in the absence of effective treatment, retinal necrosis and vitreal organization occur with tractional retinal detachment leading to phthisis bulbi with complete loss of visual function6,10). Early detection of such lesions by ophthalmologic examination is important for two main reasons. First, as candidial endophthalmitis has a strong association with disseminated candidiasis, systemic anti-fungal therapy needs to be instituted to prevent the high mortality associated with this condition. Second, prompt treatment of these early ocular lesions may avert the poor visual outcome6ŌĆō8). The presence of candida endophthalmitis is a good indicator of high mortality in seriously ill patients in intensive care units11, 12).
Total parenteral nutrition (TPN) is being used increasingly for surgical patients and medical devastating patients. Catheter-related candidemia is a well known complication in these patients, as like in our case3).
All candidemic patients should receive antifungal therapy because of a high mortality rate and late complications of candidiasis1, 12, 13). However, there is no standardized approach to the management of hematogenous candida endophthalmitis. Although amphotericin B remains the first-line drug for the management of most invasive candidasis, the efficacy may be limited by relatively poor penetration into the vitreous humor and systemic side reactions7, 8, 15). Fluconazole has been reported to be effective, having high intraocular concentration and less toxicity, but further clinical experience is needed16). Intraocular injection of amphotericin B, with or without vitrectomy, has been recommended in case of severe vitreitis17).
In conclusion, it is reasonable that all patients with suspected systemic candidiasis should have fundus examination for early detection of Candida endophthalmitis and confirmative diagnosis of systemic candidiasis.
REFERENCES
1. Fraser VJ, Jones M, Dunkel J, Storfer S, Medoff G, Dunagan WC. Candidemia in a tertiary care hopital: epidemiology, risk factors, and predictors of mortality. Clin Infect Dis 1992;15:414ŌĆō421.


2. Bross J, Talbot GH, Maislin G, Hurwitz S, Strom BL. Risk factors for nosocomial candidemia : a case control study in adults without leukemia. Am J Med 1989;87:614ŌĆō620.


3. Curry CR, Quie PG. Fungal septicemia in patients receiving parenteral hyperalimentation. N Eng J Med 1971;285:1221ŌĆō1225.

4. Parke DW, Jones DB, Gentry LO. Endogenous endophthalmitis among patients with candidemia. Ophthalmology 1982;89:789ŌĆō796.


5. Brooks RG. Prospective study of Candida endophthalmitis in hospitalized patients with candidmia. Arch Intern Med 1989;149:2226ŌĆō2228.


6. Edwards JE, Foos RY, Montgomerie JZ, Guze LB. Ocular manifestations of Candida septicemia: review of seventy-six cases of hematogenous Candida endophthalmitis. Medicine 1974;53:47ŌĆō75.


7. Brod RD, Flynn HW Jr. Advances in the diagnosis and treatment of infectious endophthalmitis. Curr Opin Ophthalmol 1991;2:306.
8. Donahue SP, Greven CM, Zuravleff JJ, Eller AW, Nguyen MH, Peacock JE Jr, Wagener MM, Yu VL. Intraocular candidiasis in patients with candidemia : clinical implications derived from a prospective multicenter study. Ophthalmology 1994;101:1302ŌĆō1309.


9. Maiale JB. Candida albicans infection confused with tuberculosis. Arch Pathol Lab Med 1943;35:427.
10. Griffin JR, Pettit TH, Fishman LS, Foos RY. Blood-borne Candida endophthalmitis. A clinical and pathologic study of 21 cases. Arch Ophthalmol 1973;89:450ŌĆō456.


11. Klein JJ, Watanakunakorn C. Hospital-acquired fungemia : Its natural course and clinical significance. Am J Med 1979;67:51ŌĆō57.


12. Elliot AJ. Endophthalmitis in systemic diseases : a review of Candida albicans endophthalmitis. Semin Ophthalmol 1987;2:229ŌĆō237.

13. Edwards JE Jr. Editorial response : Should all patients with candidemia be treated with antifungal agents? Clin Infect Dis 1992;15:422ŌĆō423.


15. Schmid S, Martenet AC, Oelz O. Candida endophthalmitis : clinical presentation, treatment and out-come in 23 patients. Infection 1991;19:21ŌĆō47.


16. Akler ME, Vellend H, McNeely DM, Walmsley SL, Gold WL. Use of fluconazole in the treatment of candidal endophthalmitis. Clin Infect Dis 1995;20:657ŌĆō664.


17. Pflugfelder SC, Flynn HW. Infectious endophalmitis. Infect Dis Clin Nor Am 1992;6:859ŌĆō873.
-
METRICS

- Related articles
-
The significance of ophthalmologic examination in a patient with xanthoma2024 March;39(2)
Homonymous hemianopia in a patient with BehcetŌĆÖs disease2015 May;30(3)
A case of granulomatous lung disease in a patient with Good's syndrome2008 December;23(4)
Acral Metastasis in a Patient with Ampullary Carcinoma2007 March;22(1)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


