INTRODUCTION
The enzyme of aspartate aminotransferase (AST) is present in a wide variety of tissues-including heart, skeletal muscle, kidney, red blood cell (RBC) and brain, in addition to liver1). So the elevation of AST is suspected of being due to injury of the above mentioned organs. Even in the injury of these, they usually are associated with other abnormalities of enzymes or metabolites such as elevation of creatine kinase (CK) and lactic dehydrogenase (LDH) in myocardiac infarction. An isolated and persistent elevation of AST occasionally can be found in advanced hepatocellular carcinoma, alcoholic liver disease and to some drug effects in hepatic disorders2–4), but these instances are very rare without the above mentioned conditions5–10). We experienced one case of the above condition and determined to study it.
CASE REPORT
A 24-year-old woman visited our department for evaluation of hepatic function because of isolated AST elevation. It had begun two years before. She had no association with any other symptoms. She denied any alcohol use, smoking and drug medication. Her family histories were non-specific except that her father had labile hypertension. Findings on physical examination were unremarkable.
The results of all laboratory studies were normal, except for an unexplainable elevation of AST at 196IU (normal 16–40). The results of radiological examinations, such as plain chest film, liver scanning, ultrasonographic and computer tomographic finding of abdomen, were unremarkable. She was considered as an unusual “hyperaspartemia syndrome” and recommended for interval check of hepatic functions.
5 months later, she revisited our department for accurate evaluation of hepatic function. The results of all laboratory studies were normal, except for isolated AST elevation at 223IU (normal 16–40). Her physical status was as follows; height 160cm, 53Kg, 100/60mmHg. Results of other liver function tests, including bilirubin, albumin, prothrombin time, alkaline phosphatase (ALP) and ALT were normal. On repeated testing, laboratory results, AST was 217IU, ALT 13IU, ALP 39IU, LDH 71IU (N:53–137), creatinine 0.9mg/dl, blood urea nitrogen (BUN) 11mg/dl, CK 59IU (N:60–103) and total bilirubin 0.6mg/dl. All serological marker for hepatitis B, C and E virus were negative. Anti-body of IgG to hepatitis A virus was positive, but IgM was negative. The total protein, serum iron, transferin, CBC, electrolyte, glucose and thyroid functions were all normal. Serological examination for rheumatoid arthritis (RA) factor, LE cell, ANA, AMA and anti-smooth muscle antibody were negative. Special studies confirmed that the patient had an immunoglobulin-complexed AST.
MATERIALS AND METHODS
1. Subjects and material
The samples of our study were collected from a patient with acute viral hepatitis (AVH; due to HBV), the above mentioned female and purified cytozolic enzyme from hemolysed RBC. The cytozolic AST was purified from a normal person’s RBC as previously described by Rej et al11).
2. AST Assay
Routine and our one case of elevated AST determinations were performed as the chemistry part of the clinical pathology on a Spectrum EPX (AbbottR, IL).
3. Exclusion Chromatography
Exclusion chromatographys were done with each 1-ml serum samples, including our isolated AST elevation case, AVH patient and the above mentioned purified cAST. The column used was 1.5-by 45-cm column of Sephacryl G-300 (Pharmacia, NJ). The elutioning was performed with 0.01 M Tris buffer and 0.15M NaCl buffer (pH 7.2) at a flow rate of 15ml/h. Column fractions were assayed directly for AST assay as described in the above paragraph.
4. Electrophoresis
Universal agarose films and buffer were purchased from Ciba-Corning Medical (LA). The samples (1.2ml) of AVH patient’s and subject’s serum and purified cAST were applied to each well. These samples were electrophoresised for 35 minutes at 100volts in universal barbiturate buffer (pH 8.6).
5. Immunoglobulin electrophoresis
When the electrophoresis was complete, the plates were layed and filled with anti-human immunoglobulin complex, G, A, M, GMA, kappa and lambda between the gaps. Then these were incubated 24 hours at room temperature and stained.
6. AST electrophoresis
AST stlaining was done as previously described by Sakakibara et al12). To briefly describe it, after the electrophoresis was complete, the plates were layed with a film of 1ml of the AST reagent mixture [10umol alpha-ketoglutarate, 200umol L-cysteine sulfinate, 0.1mg m-PMS, 0.8mg MTT, 2umol EDTA, 20mg dextran and 100umol immidazole buffer (pH 7.5)] for 20minutes of incubation at 37°C. Then the agarose plates were immersed in 10% acetic acid solution for 5minutes, washed with water and then dried in an oven at 65°C. AST activity was visualized by the appearance of purple bands.
7. Polyethylene Glycol (PEG) precipitation and Identification of Antibody class
As the methods of screening examination of enzyme-immunoglobulin complex and identification of its antibody subclass, we used the methods with slight modification previously described by Litin et al5). Briefly described in our method, PEG 6000 was purchased from Sigma chemical company. We added 100ul of a 24% solution of PEG 6000 to 100ul of serum from each subject. A blank was prepared by substituting 100ul of phosphate buffer solution (PBS) for PEG. The mixtures were incubated at 37°C for 10 minutes and centrifuged at 3,000×g at room temperature for 20 minutes. AST activity was assayed from their supernatants, as previously described. The identification of the immunoglobulin associated with AST was done by selected precipitation with rabbit anti-human antiserum. To 20ul of subject serum was added 180ul of the antiserum and then shaked the mixture was shaken gently and incubated at room air for 60 minutes. After incubation, the mixture was centrifuged at 3,000×g for 15minutes. AST activity was assayed from their supernatants as previously described.
RESULTS
1. Exclusion Chromatography
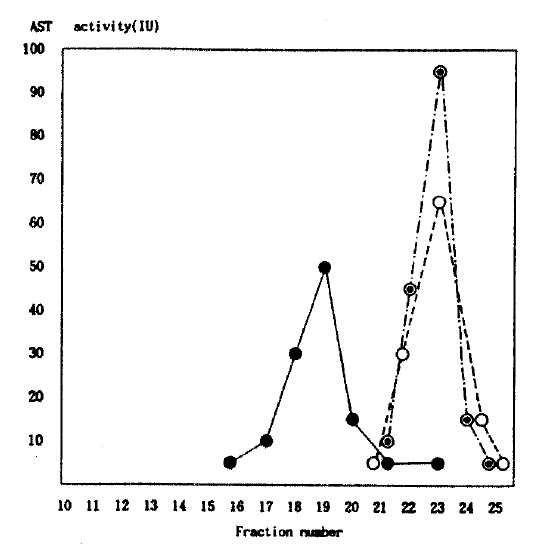
The patterns of AST activity were apparently differenCE from the isolated hyperaspartatemic subject and purified cAST and AVH patient. The patterns of AST activity were similar with purified enzyme from hemolysed RBC and AVH patient, but different with isolated hyperaspartatemic subject. The AST activity was detected in the earlier fraction of the isolated hyperaspartatemic subject and not in the other two groups (Fig. 1). This implies that it is a macromolecule with higher molecular weight.
2. Electrophoresis
1) AST electrophoresis
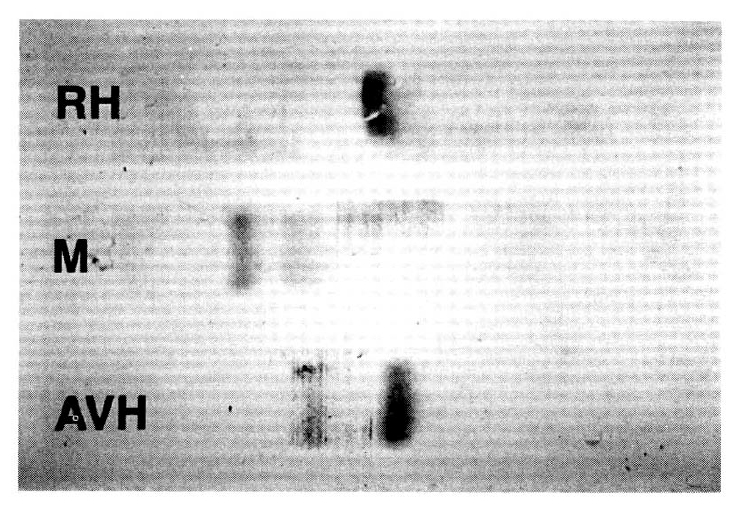
There was a single band from hemolysed RBC which shows in pure cytosomal type of AST (cAST) activity, but there were two bands in the AVH patient and the isolated AST elevation case. There were two bands in the latter group, but the pattern differed with each. In the AVH patient’s serum, there were 2 bands of which the thick band corresponded to cAST activity and the thin one to mitochondrial (mAST) type. This finding means that the major portion of circulating AST activity in AVH patient is a cytosomal type. In the macroaspartatemic case, we can find a thin band corresponding to mAST without cAST activity, but instead of the cAST type we can find abnormal thick AST activity which is near to the slit of the start, which is similar to the characteristic pattern of those previously mentioned macroaspartatemia (Fig. 2). This finding suggests that this case of hyperaspartatemia is due to mitochondrial type and abnormal type of AST activity. The abnormal type of AST activity might be cytosomal type of AST due to other effects.
2) Immunoglobulin electrophoresis
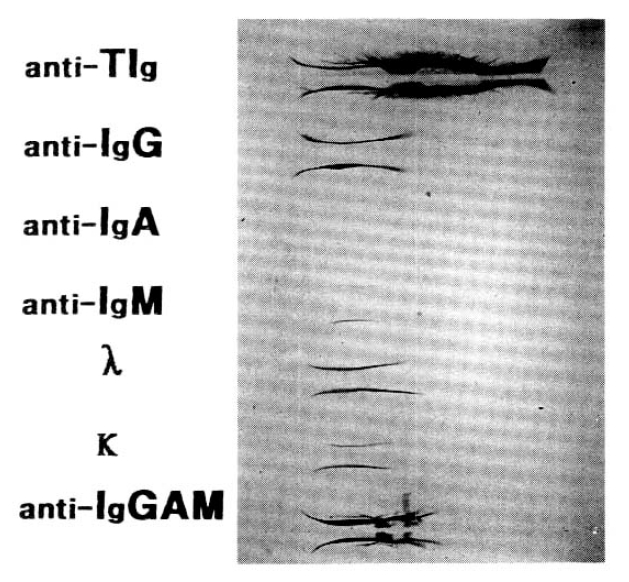
The no abnormal pattern in the AVH patient (not shown), and there was only one abnormality in immunoglobulin G(Fig. 3).
3. Polyethylene Glycol (PEG) precipitation and Identification of Antibody class
PEG is well known to precipitate antibody-antigen complexes and has been suggested as a rapid screening test for macroaspartatemia. Because of this, precipitation experiments with PEG were undertaken in an AVH patient and a macroaspartatemic patient. The rates of AST activity from each supernatant were different. The AST activity was markedly dropped from 223IU to 23IU in the macroaspartatemic patient, but slightly dropped from 430IU to 378IU in the AVH patient. It is also well known as a rapid screening test for the identification of antibody class by addition with the anti-immunoglobulin class in macroaspartatemia. Identification of antibody class was done by selected precipitation with rabbit anti-human anti-serum Ig G, A and M. There were slight changes of the AST activity in the AVH patient, but marked changes in the macroaspartatemic patient, especially to immunoglobulin G from 223IU to 24IU (Table 1). Even if there was any change in its activity, it was not significant as anti-immunoglobulin G. So the change of AST activity might be specific to Ig G in our macroaspartatemic patient.
4. Changes of AST activity according to temperature and time
To evaluate the characteristics of macro-AST due to any variable condition, AST activities were assayed in variable conditions. We could not assay enough because of deficiency of serum. There were no changes of AST activity due to purified enzyme from hemolysed RBC for 10 days, hepatitis serum for 14 days, in macroaspartatemic patient for 6 weeks in room temperature and cooled condition, and 9 weeks in frozen state(Table 2).
DISCUSSION
AST is a well-known enzyme found in a wide variety of tissues-including heart, skeletal muscle, kidney, RBC and brain, in addition to liver. So this elevation is suspected of being due to the injury of the above mentioned organs. The injury is usually associated with other abnormalities of enzymes or metabolites such as elevation of bilirubin, alkaline phosphatase (ALP), alanine transaminase (ALT) and lactic dehydrogenase (LDH) in hepatic injuries due to their etiologies9–10). Even if there is an AST elevation without ALT elevation in advanced hepatocellular carcinoma, alcoholic liver diseases and hepatic disorder with drug effect, they usually are associated with other abnormalities of enzymes or metabolites and their abnormality can be found with other diagnostic stools such as history and radiological methods. However, there rarely exist instances without those pathological conditions.
The usual method for approximating the size of an enzyme while retaining its activity is by exclusion chromatography. In this technique, larger proteins are excluded from the pores of the resin and, therefore, appear in the earlier fractions than do smaller proteins. It is even possible to approximate the molecular size by exclusion chromatography. In this reported case, isolated AST elevation caused clinicians to suspect liver disease, despite unrelated finding in the history and on physical and laboratory examinations. Liver and skeletal muscle biopsy specimens were not obtained to rule out the disease state in our case, microscopically, and were extremely unlikely because of normal CK, LDH, ALT and creatinine. Our chromatographic results easily realized those AST activities of purified enzyme from RBC, and hepatitis patients are an identical pattern, but differ to the isolated AST elevation case. It also appears in the earlier fraction in an isolated AST elevation case. By addition to these findings and rapid screening test by PEG precipitation, our case can be diagnosed as macroaspartatemia composed with enzyme-immunoglobulin complex8).
There are two circulating AST isoenzymes in the serum-one is cytoplasmic and the other is mitochondrial13) in their origin. The isoenzymes can be easily separated by electrophoretic or immunochemical methods. Even the mitochondrial isoenzyme is a major part during some severe tissue injury14), usually circulating serum AST activity which arises from the cytoplasmic isoenzyme. In most of the reports about macro-AST, they mentioned it is a enzyme-immunoglobulin complex especially due to Ig G or rarely Ig G associated with IgA, but did not mention which kinds of AST isoenzyme type were combined with immunobulin, except one5,8,10,15). PEG is well known to precipitate antibody-antigen complexes and has been suggested as a rapid screening test for macroaspartatemia5,9). So, our case might be an AST-immunoglobulin complex. In our case, the macro-AST complex might be an immunoglobulin-cytoplasmic AST isoenzyme, as the generally circulating AST isoenzyme is cAST13–4). Also in our AST electrophoresis, AST activity was not observed as cytoplasmic type but observed only a abnormal type of AST and mitochondrial type. In one report about immune-complexed AST obtained biopsy material from the liver and skeletal muscle9), the electrophotogram was similar to our study. AST isoenzymes from these tissues were normal. As circulating serum AST activity is a major fraction of the cytoplasmic type, it is suggested that normal cytoplasmic AST isoenzyme is bounded to immunoglobulin only after entry to the circulation9, 14–5). This evidence was confirmed later by Stasia et al with monoclonal antibody10).
The type of immunoglobulin associated with macro-AST complex can be easily identified by selected precipitation with PEG and anti-serum Ig G, A and M as a rapid screening test, as mentioned previously by Litin et al5). But with this method, AST activity was mildly reduced due to non-specific binding of immunoglobulin. In our case of the macro-AST, the AST activity was markedly reduced especially to immunoglobulin G. Even though it was slightly changed in the hepatitis patient and more decreased to anti-immunoglobulin A and M, it was not significant as anti-immunoglobulin G. Also there was an abnormal finding of IgG in immunoglobulin electrophoresis. So, the change of AST activity might be specific to Ig G in our macroaspartatemic patient.
A recent review on serum AST pointed out the equivocal and conflicting reports on the effects of its storage16). Generally AST activity in serum or plasma was stable within 2 weeks of storage at 4°C, 4 weeks at −20°C and in vitro frozen state lyphophilized purified AST activity may be maintained for 1 year with minimal loss of its activity17–8). In our case of macroaspartatemia, there was no change of AST activity for 4 weeks at room temperature and cooled condition, and 9 weeks in frozen state. This is the first report about the AST activity on the effect of its storage in various condition. Although we can not confirm that the enzyme activity of macro-AST might be maintained longer than normal AST, this evidence may suggest that the enzyme activity of macro-AST might be more stable than normal AST.
The causes of the increased enzyme activity in serum for a long time are not suggested. The mechanism for this increased activity is best understood for macroamylase. Amylase (half life: about 2hr.), a low molecular weight protein (MW; −55,000Da), is readily filtered and excreted by the kidney, whereas macroamylase has high molecule and is not filtered by kidney19–21). But aspartate aminotransferase (EC 2.6.1.1–100,000Da) is larger than amylase. Also, the clearance of AST is not clearly defined, but its fractional catabolic rate constant from plasma is 0.088±0.016hr−1(half life: about 23hr.)22). Usually the molecular mass of immune-enzyme complexed as determined by gel filtration and ultracentrifugation are consistent with a two-to-one ratio between enzyme and antibody. Also, the antibody (especially IgG; −180,000Da) has a larger molecule and is clear by the reticuloendothelial system (RES)21, 23–5). It has been proposed that antibody binding to serum enzyme interferes with RES clearance mechanism. As mentioned in the above reasons, macro-AST complex might be cleared by RES with intereference, so its activity might be higher in the serum.
Several enzymes complexed with immunoglobulin have been described and this group of immunoglobulin-complexed enzyme disorders has been termed the “ICE disorders”26). These conditions seem to resent a nonspecific dysproteinemic response of the human immune system. Until now, the known immunocomplexed enzymes are amylase, CK, LDH, ALP, ALT, glucose-6-phosphate dehydrogenase, acid phosphatase, lipase and AST20,23,25,27–31). Several corresponding clinical conditions and their enzyme activity, related to their disease activity, have coexisted with these enzyme disorders, although any relationship between disease states and ICE disorders is purely speculative24). In cases of macro-AST complex, it is known that the conditions associated with macro-AST complex are cardiovascular, gastrointestinal, endocrinologic, autoimmune and infectious diseases30–1). In our case, we can not find out any associated disease.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





