INTRODUCTION
Esophagogastric variceal bleeding, which consists approximately in 10–30% of upper gastrointestinal bleeding1,2), often causes shock due to a large amount of bleeding in a short period. As the variceal bleeding caused by liver cirrhosis tends to accompany hepatic coma and liver failure, its death rate can reach as high as 30–60%3); its re-bleeding rate is 67% within a year and 90% within two years.4) Therefore, although the treatment of esophagogastiric variceal bleeding has been attempted through various medical or surgical methods, a complete method has not been established as yet5). As the surgical method of esophagogastric varices, shunt operation or non-shunt operation has been conducted; however, not only is the death rate of the emergency operation in active bleeding cases high, but the incidence of post-operative liver failure is high as well. Therefore, the surgical method, for the present, tends to be an alternative, reserved for cases where the medical treatment is inappropriate or unsuccessful. As the medical methods, transfusion, balloon tamponade, drug therapy for lowing portal pressure, endoscopic variceal sclerotherapy and endoscopic variceal band ligation are used.5).
Nowadays, endoscopic variceal sclerotherapy and endoscopic variceal band ligation of the above non-invasive methods are recognized as the treatment of first choice.6–13) Although the effect of bleeding control in case of esophageal varices is differently reported depending on the researcher, the effect of endoscopic variceal sclerotherapy are reported to be 93% and 100%, according to Johnston et al.11) and Kitano et al.12), respectively, and the effect of endoscopic variceal band ligation, 86% and 90% according to Stiegmann et al.13) and Goff et al.10)-it is clear that these methods are effective for treating esophageal varices.
Since Stray et al.14) tried the treatment using polidocanol for the first time in 1982, treatments for gastric varices which use high-volume of sclerosant15) or tissue adhesive cyanoacrylate16,17) have been reproted. The endoscopic method for gastric varices is, however, difficult to perform compared with esophageal varices. Moreover, although it succeeds in the control of bleeding, it is difficult to completely eradicate varices. Accordingly, this method is not employed extensively.18)
The band ligation developed recently and there are many reports on its use in case of esophageal varices, but there have not been systematic reports on its use in gastric varices, except for a few preliminary trials.19–21). Moreover, Stiegmann, who devised the ligation method, stated ligation alone is not effective for gastric fundal varices.13)
The reason that the endoscopic treatment for the gastric varices is not used generally, as compared with esophageal varices, is the instability of sclerotherapy in gastric varices. We propose the causes of instability as follows. Firstly, because feeding vessels and shunts may be well developed in the gastric varices, it is hard to maintain the effective therapeutic level of sclerosant in the gastric varices. Secondly, blood flow cannot be brought to block by the endoscope itself in gastric varices during the endoscopic therapy, in contrast to the esophageal varices.
Therefore, we propose EVLIS with focus on maintenance of the effective therapeutic level of sclerosant by partially blocking the blood flow of gastric varices using the band ligation method.
The purpose of this study was to evaluate, prospectively, the efficacy and safety in patients given EVLIS in an attempt to control active bleeding and to prevent recurrent bleeding and the recurrence of the gastric varices.
MATERIALS AND METHODS
1. Materials
32 patients with gastric varices at Korea University Hospital, treated with EVLIS were entered into the study. Their ages ranged from 33 to 71 years and the proportion in the age groups was highest in their fifth decade, 15 cases (46.9%). The average age of patients was 47.6years. The ratio of male to female was 2.6:1–23 cases (71.9%), male, 9 cases (28.1%), female. As the causes of the gastric varices, hepatitis B or C associated liver cirrhosis consisted of 30 cases (93.8%) and hepatocellular carcinoma accompanied in liver cirrhosis, 2 cases (6.3%). The average follow-up period was 10.6months (6.2 to 18.8 months) (Table 1).
2. Methods
Foreward endoscopy (GIF Q20, Q200 Olympus Co.) using Stiegmann-Goff endoscopic ligator kit (Bard Co.) for ligation of the varices, 5% ethanolamine oleate (Macarthy Medical Co.) (abbreviated EO) with sclerosant and injection needle (Top Co.) were used. Shim and Hyun’s emergency endoscopy methods22) were used as guidelines.
The actual process of EVLIS was done as follows: after carefully observing the gastric varices to be ligated and their circumferences, the gastric varices were ligated by using an endoscopic ligator kit installed on an endoscope, and then the scloerosant was injected through the endoscope, after taking off the ligator kit.
EVLIS for gastric varices was performed in accordance with the following principles.
Gastric variceal ligation was operated differently on the basis of distribution, form and whether or not a stigma or a bleeding point of gastric varices existed.
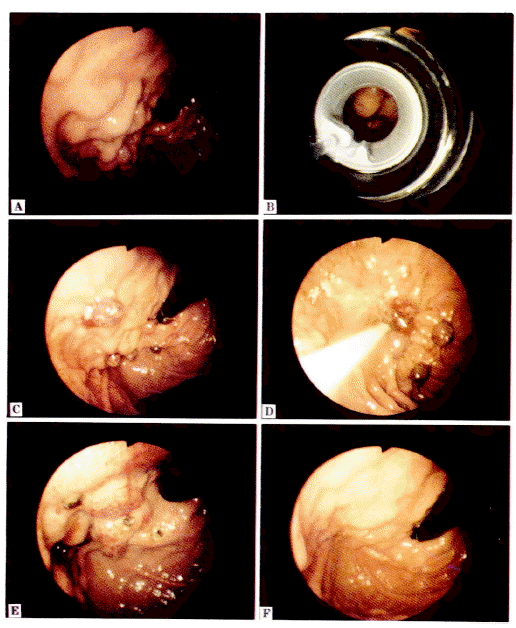
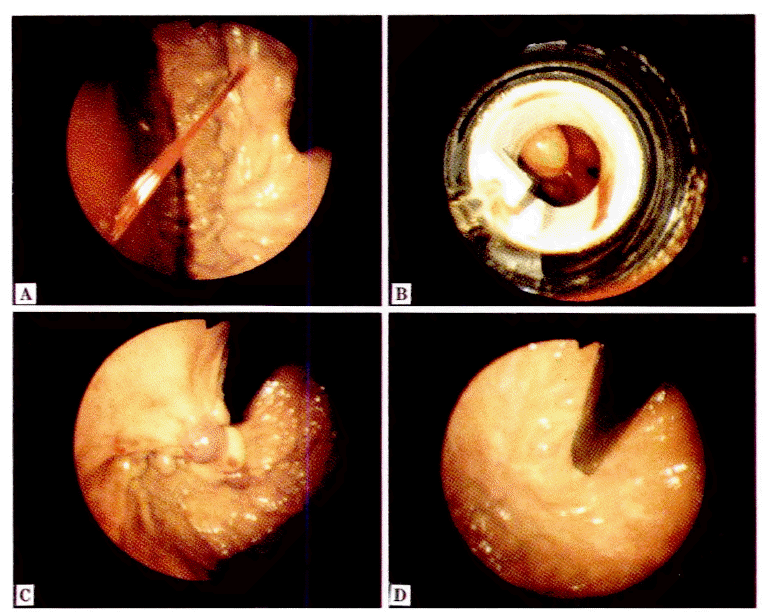
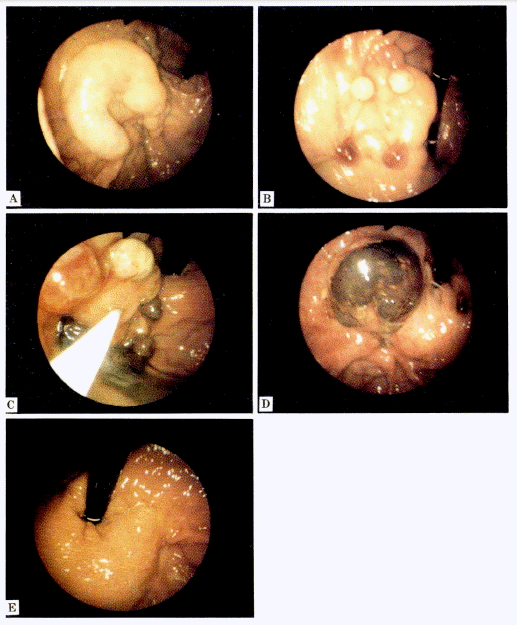
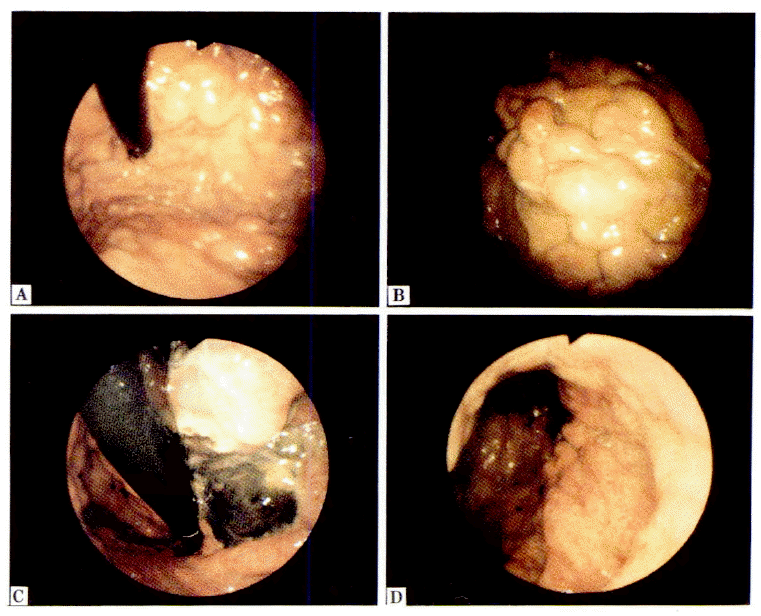
First, when a stigma or a bleeding point existed, it and the communicating varices draining into it were ligated (Fig. 1, 2). Secondly, huge mass like varices were operated according to whether the feeding vessels could be seen or not. In other words, if they could be seen, they were ligated first; if not, the peripheral margins of varices were ligated first (Fig. 3). Thirdly, for isolated tortuous varices the central part was ligated after ligating both ends as a first step. Fourthly, in cases of wide-spread varices, they were ligated after being divided into sections according to the distribution of feeding vessels (Fig. 4). Following the band ligations, the submucosal injections with sclerosant were performed 0.5cc each first on 2–3 places around pseudopolyp formed after the band ligations. Then the intravariceal injections with sclerosant between pseudopolyps were performed until the color was changed to white or white bluish, and the varices was swelled up fully for the diameter.
After EVLIS, patients were made to starve for two days and medicated H2-antagonist for treatment of the postoperative ulcer.
Progress was observed by endoscopic examination after 3 days, 10 days, 24 days and 2 months and, after that, every 3 months. In case of EVLIS for the wide-spread gastric varices, however, follow-up endoscopy had to be done every 10 days.
The efficacy of EVLIS was evaluated in about 2 months after the treatment, when the edema and ulcer would disappear completely.
We classified cases, in which gastric varices disappeared completely, as the complete eradication group, and the cases in which only the number and size of gastric varices decreased after EVLIS as the incomplete eradication group.
We defined the re-bleeding as such cases where there was bleeding from the varices operated by EVLIS.
We also classified the condition of gastric varices and the presence of according to the distribution, form and bleeding as follows.
The distribution and form of gastric varices was classified using Hosking and Johnson’s classification23) and Choi’s classification,24) respectively. On the basis of bleeding conditions, the cases where there was present active bleeding were classified as the bleeding gastric varices group, where as the cases with no bleeding at the time were classified as the non-bleeding gastric varices group, regardless of bleeding stigma or not.
The general conditions of patients were evaluated using Child’s classfication.25)
The results of EVLIS were analyzed on the basis of Child’s classification, the bleeding condition, the distribution and form of gastric varices and the compound factor of bleeding and distribution of the varices.
A statistical analysis was done using the Chi-square test and Fisher’s exact test.
RESULTS
The treatment results of 32 cases of gastric varices operated by EVLIS were analyzed according to the patient’s general condition, the bleeding conditions, the distribution and form of gastric varices and the compound factor of bleeding and distribution of the varices.
1. The Patient’s General Condition
The treatment results of EVLIS according to the patient’s general condition by Child’s classification25) are as follows.
The greatest number of patients were Child C group, in which the patient’s general condition was poor; 5 cases (15.6%) were Child A group, 12 cases (37.5%) Child B group and 15 cases (46.9%) Child C group.
In all cases, the varices were effectively eradicated; there was not a single case of ineffective eradication or re-bleeding. Therefore, no correlation existed between the patient’s general condition and the state of effective eradication (Table 2).
2. The Bleeding Condition
Of the 32 cases, 11 (34.4%) were in the bleeding gastric varices group and 21 (65.6%) in the non-bleeding gastric varices group. In each group, there was 100% effective eradication; no ineffective eradication or re-bleeding was present. Accordingly, there was no correlation between the bleeding condition and the effectiveness of EVLIS (Table 3).
3. The Distribution of Gastric Varices
By Hosking and Johnson’s classification,23) Type 2 was the most common type; 9 cases were Type 1 (28.1%), 22 cases Type 2 (68.8%) and 1 case was Type 3 (3.1%).
The eradication was 100% effective in each group; no ineffective eradication or re-bleeding was present. Consequently, there was no difference in the effect of EVLIS according to the distribution of gastric varices (Table 4).
4. The Form of Gastric Varices
According to Choi’s classification,24) Form 2, moderate variceal form, was the most common type; 12 cases were Form 1 (37.5%) 13 cases Form 2 (40.6%) and 7 cases Form 3 (21.9%).
Gastric varices of all the cases were eradicated completely, regardless of the type of form, and no ineffective eradication or re-bleeding was shown (Table 5).
5. The Bleeding Condition and the Distribution of Gastric Varices
The results according to the compound factor were as follows.
In Type 1, 3 cases were the bleeding gastric varices group; 6 cases, the non-bleeding gastric variceal group. In Type 2, 7 cases were the bleeding group and 15 cases, the non-bleeding group. In Type 3, 1 case was the bleeding group.
Gastric varices of all the cases were eradicated completely, and no ineffective eradication or re-bleeding was present.
The mean number of EVLIS sessions in the bleeding gastric varices group was 2.5 times and in the nonbleeding group, 2.0. There was no significant difference. On the basis of the distribution of gastric varices, however, the mean number of EVLIS sessions in Type 1 was 1.5; in Type 2, 2.3;in Type 3, 8.0. There was a significant difference (p<0.05)
The mean number of band ligations in the bleeding group was higher as compared with the non-bleedig group, 8.3 and 5.4, respectively, but the difference had no statistical significance. However, on the basis of the distribution of gastric varices, there was a significant difference as Type 1 was 2.9;Type 2, 6.8;and type 3, 30.0 (p<0.05) (Table 6).
6. Complications
Out of the 32 cases, epigastric pain occurred in 19 cases (59.4%); fever, in 10 cases (31.3%); oozing from ulcer developed after EVLIS, 4 cases (12.5%). Serious complications such as perforation did not occur (Table 7).
DISCUSSION
Since the gastric varices developed by portal hypertension were first reported by Stadelman26) in 1913, it has been considered as a serious problem clinically because there is no established non-invasive therapeutic method, in spite of its danger of possibly causing death by excessive bleeding.18)
The major blood vessels involved in gastric varices are left gastric vein, short gastric vein and posterior gastric vein.27) The gastric varices acompanied by esophageal varices are generated by a branch of the left gastric vein penetrating the walls of the stomach 2–3cm under from esophagogastric junction that expands in the upper part of the submucosal layer.28) The mass-like gastric varices occur mainly from the short gastric vein and the posterior gastric vein and, in almost all cases, they are accompanied by gastrorenal shunts.29)
Gastric varices can occur in all parts of the stomach, but are formed mainly in fundus and cardia under portal hypertension, because the fundus has a rich venous drainage in the submucosal layer, which has been termed the fundic plexus.30) As it is difficult to make a diagnosis of gastric varices,30) there have not been many reports. However, as Mathur et al.31) and Sarin et al.32) reported the incidence rate of gastric varices as 48% and 20%, respectively, in patients of portal hypertension before esophageal sclerotherapy, they suggested that frequency of occurrence of gastric varices was lower than esophageal varices. The bleeding rate was also lower (10–25%) in gastric varices as compared to those in esophageal varices.31,32)
Gastric varices show a high mortality rate, although both the occurrence rate and the bleeding rate are relatively low as mentioned above. Because gastric varices tend to bring about massive hemorrhage once bleeding occurs,33) and a good view is difficult to obtain in cases where there is a large amount of blood in the stomach, it is difficult to operate an endoscopic therapy and moreover, an established non-invasive treatment method has not been developed yet.18)
Since the non-invasive, aggressive treatment of gastric varices was firstly tried by Stray et al.14) using polidocanol in 1982, some endoscopic treatments using sclerosants such as ethanol,32) sodium tetradecyl sulfate,33) high-volume of sclerosant15) and the tissue adhesive cyanoacrylate16,17) have been reported, but there has not been a single established effective method yet.18)
The mechanism of the action of a sclerosant is unclear but, in intravariceal injection, its use produces a variable combination of intimal injury, initiated by its direct irritant action and denaturation of plasma protein and red blood cells by its contact with the blood. A necroinflammatory response is generated, resulting in variable degrees of thrombosis, necrosis and fibrosis. On the other hand, in paravariceal injection, the sclerosants cause edema and inflammation around the variceal wall, eradicating varices via fibrosis of the tissue.34) A tissue adhesive agent, cyanoacrylate obstructs varices due to its characteristic of hardening within 20 seconds in moist conditions and instantly in blood.17)
The role of sclerotherapy for the treatment of gastric varices remains uncertain. The little information available is derived from a relatively small and uncontrolled series. Sariin et al.35) reported 38% of 33 gastric varices to be completely eradicated and 36% of them to have rebled using ethanol with a combination of para and intravariceal injections. Trudeau and Prindiville33) reported 100% bleeding control rate in the first bleeding episodes and 44% in recurrent bleeding cases (89%) of the 9 patients with bleeding gastric varices, after using intravariceal injection of sodium tetradecyl sulfate. Raws et al.16) reported 69% of 39 bleeding gastric varices cases to have been successfully controlled by use of butylacrylate and the re-bleeding rate to be 41%. Lee et al.15) reported 93% of 28 bleeding gastric varices cases to have stopped and 21% to have rebled by intravariceal injection of high-volume of sclerosant.
As eah study uses different subjects and criteria on the effect of sclerotherapy, it is difficult to compare these results. However, we presume the high re-bleeding rate to have occurred because not all the gastric varices were treated in a short time, or because of incomplete slerotherapy.
Aside from such sclerotherapy methods for gastric varices, Jensen et al.19) recently reported 75% of 4 bleeding gastric varices patients to have been successfully controlled, 50% of them to have rebled after being treated with endoscopic variceal ligation which was first introduced by Stiegmann et al.36) in 1986.
The mechanism of band ligation method is that effect of strangulation of varices form superficial ulcers by ischemic necrosis, and varices are eradicated during the course of the healing of the ulcers.37)
This ligation method is recognized as an effective treatment in cases of esophageal varices6–11,13) stated that band ligation by itself was not effective enough in treating gastric varices.
As the ligation and the sclerotherapy are not established as an effective therapeutic method for gastric varices,18) Hosking et al.23) and Sarin et al.32), indicated that either a medical or a surgical method, as a remedy for gastric varices, should be selected differently according to the presence of connections between gastric varices and esophageal varices. That is, Hosking et al.23) suggested prophylactic sclerotherapy in cases where the gastric varices were connected to the esophageal varices located on the lesser curvature, if they persist for more than 6 months after obliteration of esophageal varices.
However, considering the fact that the risk of operation is high, because most of the bleeding gastric varices patients have decreased liver function or have poor general condition caused by a large amount of bleeding,38) the surgical method cannot be said to be the most effective and appropriate treatment for all patients.
From the reports above, neither the sclerotherapy nor the band ligation can be stated as an effective method of treatment for the gastric varices.
The reasons for the conclusions above are that local effective concentration of sclerosants which cause the formation of thrombus with its usual dose cannot be maintained because most of the gastric varices may have many feeding vessels and accompany huge shunts like gastrorenal shunt. Therefore, significant cases using sclera-osants are not effective. Futhermore, if excessive amount is injected, a deep ulcer can be formed at the injection site, and the risk of systemic complications increase and so, the effectiveness and safety of the sclerotherapy are reduced.
Accordingly, in case of the sclerotherapy, we presume that the temporary hemostatic method is needed in the procedure for effective variceal therapy.
In case of the band ligation, the significant bleeding can occur from some parts of the ulcers secondary to the ligations of the gastric varices.20) Although Saeed et al.39) stated that because the actual depth to which works the suction drum is 12mm, superficial ulcers were formed in cases of the esophageal varices, we suppose, in gastric varices, there is the risk of formation of deep ulcers, as the stomach is not only in the abdominal cavity in contrast to the esophagus located in the mediatinum, but the tip of the suction drum is able to adhere perpendicularly to the gastric mucosa. Also, gastric acidity may play a role in ulcer formation and healing process. Moreover, Jensen et al.20) reported 24% of 17 bleeding esophagogastirc varices patients to have failed after being treated with endoscopic variceal ligation and a significant number of them to have developed severe bleeding from a ligation induced ulcer.
Therefore, the ligation method requires a process to prevent the bleeding of an ulcer secondary to treatment.
Consequently, if only the above defects in the sclerotherapy and the band ligation method for gastric varices could be corrected, it would be possible to treat the gastric varices safely and effectively.
Therefore, we carried out the band ligation and the sclerotherapy at the same time, to cut off the blood flow of the gastric varices, the main problem of the sclerotherapy, and to prevent the bleeding of ligation induced ulcer, the main problem of the band ligation.
The band ligation for gastric varices is performed according to methods mentioned above to prevent re-bleeding from the stigma and to cut off the blood flow of the varices effectively. The sclerotherapy is performed by submucosal injection of sclerosant in the circumference of the ligation site to prevent bleeding of the ligation induced ulcer, and by intravariceal injection for complete variceal eradication.
In this study, gastric varices were eradicated completely in all of the 32 cases; also, all of the 11 cases with active bleeding achieved successfully controlled bleeding and complete eradication.
Especially, the 22 cases (66.8%) of Type 2 and 1 case (3.1%) of Type 3, for which Hosking et al.25) and Sarin et al.34) suggested the surgical treatment to be effective in cases where the gastric varices did not connect with the esophageal varices, were completely eradicated 100% with EVLIS.
Even though these results require long term follow-up, as the period of observation was on the average 10.6 months, all of the 32 cases showed good results where re-bleeding or recurrence of varices did not appear during the period of observation.
As complications, epigastric pain occurred in 19 cases (59.4%), fever in 10 cases (31.3%) and oozing from ulcer in 4 cases (12.5%). Epigastric pain and fever are presumed to be due to the injection of the sclerosant and oozing does not bring about a decrease in blood pressure or palpitation. Serious complications such as perforation did not occur.
In conclusion, this study shows that EVLIS for gastric varices is a safe and effective method. However, long-term controlled studies will be necessary for further evaluation.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





