INTRODUCTION
It has been well known that many stressful conditions, such as trauma,1) burn,2,3) surgical operations,1,4,5) may cause immune suppression which increases the susceptibility to infection. But the mechanism of immune suppression is still not clear.
Occino et al.6) demonstrated a polypeptide that is present in human serum, and its molecular weight is below 10,000 dalton. Menzoian et al.7,8) suggested that the polypeptide suppresses many immunologic reactions which are mediated by T lymphocyte. Also, Viet and Mechael demonstrated an inhibitor of immunologic response increased whenever the immune system was stimulated. ╬▓-endorphin, m.w. 3450, is released with ACTH from the pituitary gland under stressful conditions10,11,12) and its receptor has been demonstrated on the circulating lymphocytes.13) McCain et al.14) demonstrated that PHA stimulated lymphoblastogenesis could be suppressed by the high concentration (105 pM) of ╬▓-endorphin. Because their concentration of ╬▓-endorphin was also much higher than that (15ŌĆō30 pM) of the patient who received stress and it was doubtful that the defect of lymphoblastogenesis caused the suppression of immunologic response, we observed the suppression of PHA stimulated lympho blastogenesis in two conditions, metyrapone test and NelsonŌĆÖs syndrome, in which plasma ╬▓-endorphin concentration increases. Simultaneously, we observed the effect of cortisol on the PHA stimulated lymphoblastogenesis because the hormone increase in the stressful conditions.
SUBJECTS AND METHODS
Between 9 a.m. and 10 a.m. 20 ml of whole blood was withdrawn from the antecubital vein and was mixed with one volume of EagleŌĆÖs minimal essential media. Mononuclear cells were isolated from the heparinized blood over a Ficoll-Hypaque gradient and washed two times with HankŌĆÖs balanced salt solution. The mononuclear cells were suspended in complete media (minimal essential media including 10% fetal bovine serum, streptomycin and amphotericin B) to make the concentration 2 ├Ś 105/ml.
Phytohemagglutinin (PHA) (Lectin, Sigma Co.) was diluted to the concentration 0,1,2,5,7,10 ug/ml with complete media. ╬▓-endorphin (Sigma Co.) was diluted to make final concentration 10.102,104 pM/well and cortisol (Sigma Co.) was also diluted to the final concentration 10,102, 103 ug/dl. 100 ul of cell suspension was incubated with 100 ul of PHA and 20 ul of ╬▓-endorphin or cortisol in the round bottomed microtitration plates for 48 hours under 5 % CO2, 37┬░C. After 6 days of culture, 10 ul of 0.1 uCi of 3H-thymidine (specific activity 2Ci/mmol, New England Nuclear) was added to the wells and incubated for 20 horus. Cells were harvested and dried at 37┬░C for 60 min. before counting with liquid scintillation spectrophotometer. The viability of cells were determined by the trypan blue dye exclusion method. Suppression of lymphoblastogenesis was expressed with % suppression.
Metyrapone (1.5 gm) was administered to five healthy men at 10 p.m. and whole blood was withdrawn at 9 a.m. in the next morning. The PHA stimulated lymphoblastogenesis was done in a patient with NelsonŌĆÖs syndrome.
RESULTS
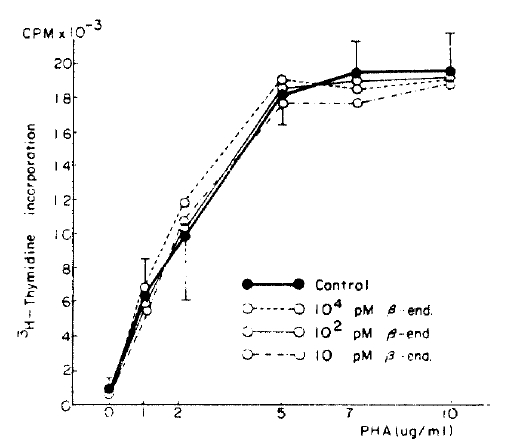
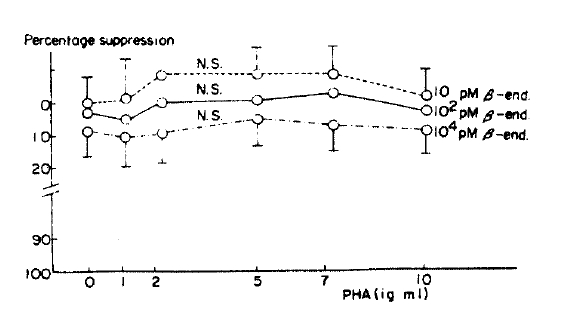
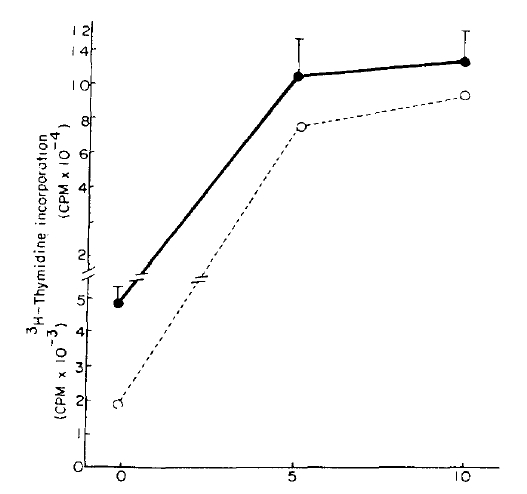
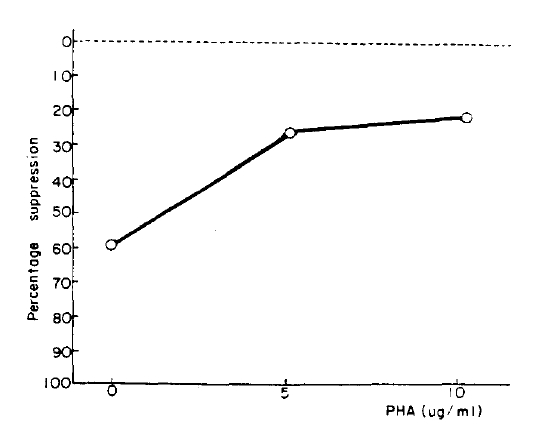
The maximal lymphoblastogenesis was observed at the PHA concentration 5 ug/ml. ╬▓-endorphin did not suppress the lymphoblastogenesis at all concentrations (Fig. 1, 2).
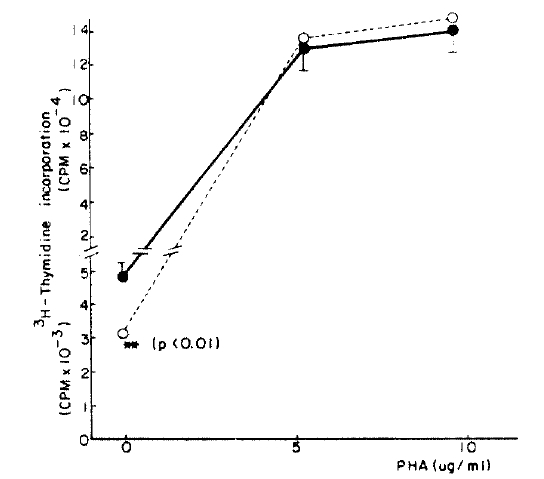
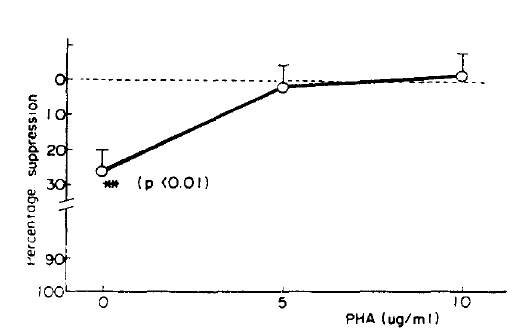
In the cases who received metyrapone the previous night, the lymphoblastogenesis was suppressed 27 percent in the wells containing no PHA (Fig. 3, 4) although there was no suppression in the wells containing PHA.
DISCUSSION
Severe stress may influence the immune system, resulting in the development of a disease.1ŌĆō5),15ŌĆō18) But the mechanism by which the stress can alter the immune function remains unclear.
During the past decades there has been many efforts to find which substance has a capacity to alter the immune function. In 1959, Karmrin19) isolated a protein fraction from human plasma that could suppress the immune function, and Mowbray et al.20) successfully suppressed the rejection and antibody formation by using a ╬▒-globulin fraction isolated from the bovine serum. Occhino et al.6) isolated a peptide subfraction, m.w. 4000 to 9000, from the ╬▒-globulin fraction and also Constantian et al.1) isolated a peptide, m.w. below 10,000, from the patients who received trauma or surgery which suppressed the PHA stimulated lymphoblastogenesis. Although it has not been known what it is, it seems a possible hypothesis that a hormone can modulate the immune system under stressful conditions because the endocrine system responds rapidly to the various stress.
╬▓-endorphin is a peptide hormone, m.w. 3450, released with ACTH from the pituitary gland in response to stress. It was demonstrated that the peptide hormone has a receptor on the surface of the circulating lymphocytes13) and so it was speculated that ╬▓-endorphin may modulate immunologic functions. McCain et al.14) observed that ╬▓-endorphin could suppressed the PHA (10ug/well) stimulated lymphoblastogenesis, 30% suppression at 1019M and 75% suppression at 10ŌłÆ7M, and which suggested that ╬▓-endorphin act through non-opiate receptor because those suppressive effects could not be inhibited by naloxone. Although we could not observe the supressive effect of ╬▓-endorphin at any concentrations of PHA, our result was not in contrast to their result because our PHA concentrations were different from theirs. Their concentrations were ten times as high as ours because their concentration unit ug/well as compared to ours ug/ml. The suppressive effect was not observed at the low concentration as ours.
But it is thought that ╬▓-endorphin has no suppressive effect on PHA stimulated lymphoblastogenesis. There are several reasons as followings: First, even though the PHA concentrations were much lower than theirs, the maximal lymphoblastogenesis was observed at 5 ug/ml (0.5 ug/well) in our experiment. Therefore, the effect should have been observed in our experiment if ╬▓-endorphin had a suppressive effect. Second, Goodwin et al.21) demonstrated that the suppressive effects of various suppressive substances were observed better at the lower PHA concentration than the concentration which stimulates maximal lymphoblastogenesis because the higher concentration induces unphysiological conditions. On that point of view, McCainŌĆÖs results were contradictory: the maximal suppressive effect was observed at a higher PHA concentration and minimal suppression at a lower PHA concentration. It seems likely that their results could not implicate the physiological meaning even if suppression was observed. Third, concentrations of ╬▓-endorphin used in their experiment were 1,000 times as high as those in the plasma of one who is under a stressful condition, and which also suggest that ╬▓-endorphin could not change the lymphoblastogenesis in vivo.
In order to observe the suppressive effect of ╬▓-endorphin indirectly in vivo, we investigated the lymphoblastogenesis in 5 healthy men to whom metyrapone was administered and in a patient with NelsonŌĆÖ syndrome. Although their lymphoblastogeneses were not significantly different from those of normal controls when stimulated with PHA, those were suppressed without PHA stimulation. It was thought that the suppressive effect of ╬▓-endorphin decreases as the PHA concentration increases. Although it can not be mentioned in general, chronic exposure upon the high concentration of ╬▓-endorphin might suppress lymphoblastogenesis in vivo. The difference between in vitro results and in vivo results can not be explained now, not only because an effect of ACTH and an effect caused by cortisol deficiency are not known, but because it is possible that a intermediate step exist in vivo between the elevated ╬▓-endorphin and suppression of lymphoblastogenesis.
Blalock et al.22,23) demonstrated that leukocyte interferon is a peptide which has similar structures as ACTH and ╬▓-endorphin and can be cleaved by the proteolysis to yield ACTH and ╬▓-endorphin. It can be hypothesized that the ACTH and ╬▓-endorphin cleaved from interferon have a negative feedback effect on the hypothalamus and pituitary gland to suppress the release of ACTH and ╬▓-endorphin, and which suggests a close connection between the endocrine system and immune system. Upon the infection, leucocytes synthesize the interferon which suppress the lymphoblastogenesis and simultaneously suppress ACTH and ╬▓-endorphin to reduce cortisol secretion. On the other hand, the stress increases the ╬▓-endorphin secretion which stimulate synthesis of interferon in lymphocytes as a defence mechanism against the infection, but under prolonged stress, interferon has a negative feedback to decrease a release of ACTH and ╬▓-endorphin resulting in a decrease of interferon synthesis. In the cases of a metyrapone test and a patient with NelsonŌĆÖs syndrome, it can be speculated that lymphoblastogenesis is suppressed by the mechanism of the above hypothesis and a patient with nelsonŌĆÖs syndrome is susceptible to infections.
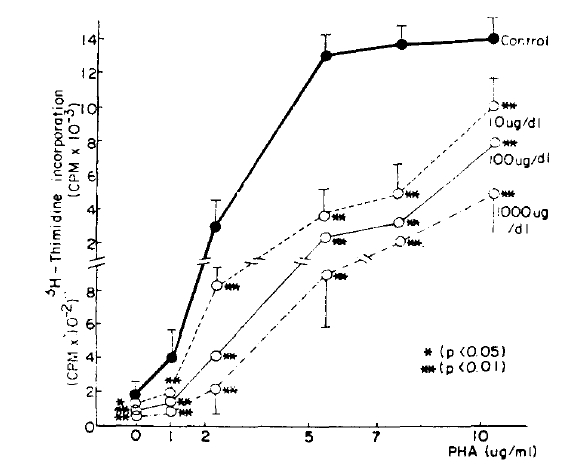
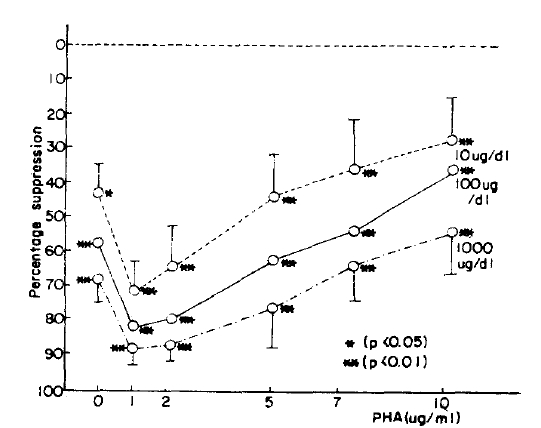
It is well known that cortisol suppresses the lymphoblastogenesis in vitro.24ŌĆō28) In our results, cortisol suppressed the PHA stimulated lymphoblastogenesis in a dose dependent manner. It agrees with other reports and suggests that our experimental method used in the evaluation of suppressive effect of ╬▓-endorphin was not wrong. The suppressive effect of cortisol in vitro has been explained by the direct toxic effect to lymphoblast24) but cortisol has no direct toxic effect even at the pharmacological concentration in vivo. Yu et al.29) and Fauci et al.30) suggested that the circulating lymphocytes decreased, especially T lymphocytes, when cortisol concentration increased resulting in the suppressed PHA stimulated lymphoblastogenesis. Tavida et al.31) demonstrated the negative correlation between the serum cortisol concentration and PHA stimulated the lymphoblastogenesis and suggested that suppression of lymphoblastogenesis is not controlled directly by cortisol but by other factors. The above hypothesis can explain the negative correlation; Decrease of cortisol concentration induces ╬▓-endorphin secretion which stimulates synthesis of lymphocyte interferon via its receptor, resulting in suppression of lymphoblastogenesis.
In conclusion, we suggested that ╬▓-endorphin could not suppress the PHA stimulated lymphoblastogenesis directly in vitro in contrast to cortisol, but we speculated that ╬▓-endorphin can suppress the lymphoblastogenesis by the stimulation of interferon synthesis indirectly in vivo. In the future the studies regarding the relationship between ╬▓-endorphin and interferon activity are needed.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





