INTRODUCTION
Irinotecan is a cytotoxic agent with a low toxicity profile and proven activity against a number of solid tumors [1-3]. As shown in a pharmacokinetic study, nearly 80% of the overall clearance of irinotecan can be attributed to nonrenal processes, including the metabolic degradation of irinotecan [4]. However, it remains unknown whether irinotecan is safe for administration in patients with renal failure and whether dose adjustment is required. We administered a standard dose of irinotecan to a hemodialysis patient with limited disease small-cell lung cancer and end-stage renal disease. The treatment with irinotecan was safe and well tolerated. This is the first study to report the successful treatment of a small-cell lung cancer patient on hemodialysis with irinotecan.
CASE REPORT
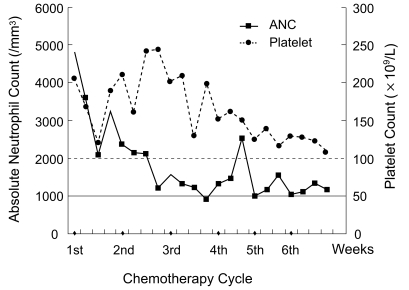
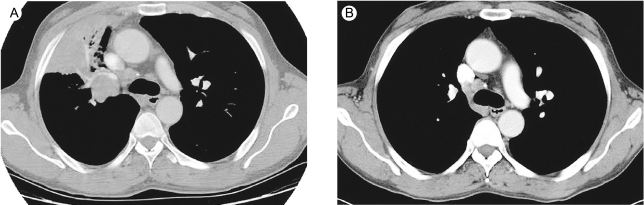
A 49-yr-old man was referred to our hospital because of chronic cough and chest pain. A computed tomography (CT) scan revealed a right hilar lung mass without distant metastases. Percutaneous needle lung biopsy was performed, and histopathologic analysis revealed small-cell lung cancer. Thus, the patient was diagnosed to have limited disease small-cell lung cancer. The patient also suffered from chronic renal failure with end-stage renal disease most probably due to glomerulonephritis, which was being treated with hemodialysis on a 3 times weekly schedule for 5 years. The patient was treated with 6 courses of intravenous irinotecan for 90 min on days 1, 8, and 15 of a 28-day cycle. Hemodialysis was performed 24 h after the administration of irinotecan. The patient was dialyzed for 4 h through an arteriovenous shunt in his left forearm. During the first cycle, he received a 100 mg/m2 dose of irinotecan, and the treatment was well tolerated. Hence, a dose of 100 mg/m2 was used subsequently. Pretreatment and nadir blood cell counts are shown in Figure 1. Grade 2 anorexia and fatigue were observed as non-hematological adverse effects. After the completion of chemotherapy, the tumors showed a complete response as assessed by CT scan (Fig. 2); the patient was then treated with thoracic irradiation. The patient has been free of disease for 12 months.
DISCUSSION
Clinical trials include only selected patients with a good performance status as well as those with normal organ functions. However, patients in practical settings sometimes have comorbidities such as chronic pulmonary diseases, cardiovascular diseases, hepatic dysfunctions, and renal failure. In cancer patients, renal failure is common and is often caused by previous cytotoxic treatment, coexisting diseases (e.g., hypertension, diabetes) or by the tumor itself. The relatively low number of patients with renal failure makes it difficult to conduct pharmacokinetic studies of cytotoxic drugs in these patients. Consequently, for many chemotherapeutic agents, limited information is available regarding the optimal dosing and appropriate treatment schedules for patients suffering from varying degrees of renal failure [5-10]. Most previous studies on chemotherapy of such patients have reported the feasibility of administration at lower doses rather than standard doses in a limited number of patients [8]. Chemotherapy involving such low doses of drug, however, has never been shown to be sufficiently effective for the treatment of solid tumors.
Irinotecan is very effective for various forms of malignant tumors, including small-cell lung cancer and colorectal cancer [1-3]. Diarrhea and neutropenia are the most common side effects of irinotecan treatment [11]. Small-cell lung cancer is a highly chemosensitive cancer; hence, chemotherapy has been established as the primary mode of treatment for this cancer. Stemmler et al. [12] performed chemotherapy with irinotecan for hemodialysis patients with metastatic colorectal cancer; a gradual dose escalation of irinotecan showed that a weekly schedule at a dose of 80 mg/m2 was feasible. A dose escalation to 100 mg/m2 resulted in severe diarrhea; therefore, treatment was continued with a weekly dose of 80 mg/m2. However, in the present case, we administered a dose of 100 mg/m2 irinotecan because diarrhea is an uncommon side effect in Korea [13]. During the treatment, the patient showed no significant adverse effects, including diarrhea.
We conclude that the standard dose of irinotecan can be used safely and successfully for the treatment of hemodialysis patients with small-cell lung cancer and end-stage renal disease, with a close follow-up of potential adverse effects.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





