Atrial fibrillation in critically ill patients who received prolonged mechanical ventilation: a nationwide inpatient report
Article information
Abstract
Background/Aims
To evaluate temporal trends of atrial fibrillation (AF) prevalence in critically ill patients who received prolonged mechanical ventilation (MV) in the United States.
Methods
We used the 2008 to 2014 National Inpatient Sample to compute the weighted prevalence of AF among hospitalized adult patients on prolonged MV. We used multivariable-adjusted models to evaluate the association of AF with clinical factors, in-hospital mortality, hospitalization cost, and length of stay (LOS).
Results
We identified 2,578,165 patients who received prolonged MV (21.27% of AF patients). The prevalence of AF increased from 14.63% in 2008 to 24.43% in 2014 (p for trend < 0.0001). Amongst different phenotypes of critically ill patients, the prevalence of AF increased in patients with severe sepsis, asthma exacerbation, congestive heart failure exacerbation, acute stroke, and cardiac arrest. Older age, male sex, white race, medicare access, higher income, urban teaching hospital setting, and Western region were associated with a higher prevalence of AF. AF in critical illness was a risk factor for in-hospital death (odds ratio, 1.13; 95% confidence interval, 1.11 to 1.15), but in-hospital mortality in critically ill patients with AF decreased from 11.6% to 8.3%. AF was linked to prolonged LOS (2%, p < 0.0001) and high hospitalization cost (4%, p < 0.0001). LOS (−1%, p < 0.0001) and hospitalization cost (−4%, p < 0.0001) decreased yearly.
Conclusions
The prevalence of comorbid AF is increasing, particularly in older patients. AF may lead to poorer prognosis, and high-quality intensive care is imperative for this population.
INTRODUCTION
Atrial fibrillation (AF) is one of the most common cardiovascular health problems in the general population [1], and its prevalence ranges from 0.1% among people younger than 55 years to 9.1% among those older than 85 years [2]. AF is also common in critically ill patients [3], accounting for more than 61% of all kinds of arrhythmias. The incidence of AF is increasing in critically ill patients and a direct relationship between incidence and disease severity has been observed [4].
The outcomes of critically ill patients with AF are somewhat conflicting. Some studies have suggested that AF development in critical illness is associated with increased mortality and longer hospital stay [3,5,6]. However, in a single-center prospective observational study, there was no difference in mortality rates between patients with arrhythmia and those without [7]. Although some organizations published guidelines for AF management, they did not include patients with a critical illness [8,9]; also, the risk of anticoagulation therapy during the critical illness is unclear, so the management of AF in patients with critical illness remains a great challenge [10].
AF in critically ill patients has attracted much attention in recent years. Although AF incidence and risk factors were reported by some studies, the trends and outcomes of critically ill patients with AF remain unclear. The National Inpatient Sample (NIS) offers an opportunity to retrospectively evaluate the association between critical illness and AF. Our primary aim was to describe and quantify temporal trends in the prevalence of AF in critically ill patients in the United States (US) from 2008 to 2014. Secondarily, we evaluated the association between the emergence of AF and different characteristics, such as patient demographics and hospital characteristics. Finally, we examined the impact of AF on subsequent outcomes among patients with critical illness.
METHODS
Data source
We used NIS data from 2008 to 2014. The NIS is the largest all-payer inpatient health care database in the US and was made available by the Healthcare Cost and Utilization Project of the Agency for Healthcare Research and Quality. Before 2012, data of discharges were obtained by sampling 20% of the hospitals in the US. After 2012, the NIS underwent a redesign to estimate a 20% sample of discharges from all participating hospitals, which can represent over 95% of the US population [11]. Details about the NIS are available at www.hcup-us.ahrq.gov/nisoverview.jsp. This study was approved by the Committee on Ethics of Medical Research, Second Military Medical University (Navy Medical University), PLA (approval number: NMUMREC-2019-166). The NIS is a public and open-access database, which does not contain identified information. Written informed consent by the patients was waived due to a retrospective nature of our study.
Study population
We identified critically ill patients by using International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) procedure code for continuous invasive mechanical ventilation for 96 or consecutive hours (96.72).[12] The exclusion criterion was age < 18 years. We identified a total of 2,578,165 patients (weighted) with critical illnesses.
Covariate assessment
The NIS variables were used to identify patients’ demographic characteristics including age, sex, race, income by ZIP code, and insurance type. Hospital characteristics included location/teaching status, bed size, and region [13]. We used the Elixhauser Comorbidity Index (ECI), which includes 29 comorbidities, to account for the burden of comorbidity [14]. The CHA2DS2-VASc score was used to estimate the acute ischemic stroke risk in AF [15]. We also identified the types of organ failure (Supplementary Table 1) and acute medical conditions (Supplementary Table 2) associated with critically ill status using standard ICD-9-CM codes and used them to adjust the multivariable models [16].
Primary and secondary outcomes
The primary objective of this study was to determine the temporal trend of AF among critically ill patients from 2008 to 2014. AF was defined using ICD-9 code (427.31) in discharge diagnosis fields. The code has a specificity of 98% and sensitivity of 80% for AF [17]. The secondary objectives included in-hospital mortality, length of stay (LOS), hospitalization cost, and discharge location. The hospitalization cost was recalculated after adjustment for inflation. Discharge location was divided into home, long-term acute care hospital or skilled nursing facility (LTACH/SNF), and died.
Statistical analysis
We derived the national estimates for the critically ill patients’ measurements using the NIS survey-weighted methods. Using the strata and weights with appropriate survey commands, we can calculate the national estimates from 20% of all admissions in the NIS. According to the occurrence of AF, the chi-square or t test was used to compare patients’ baseline characteristics. The trends of the prevalence in AF were analyzed in subgroups categorized by age, sex, race, insurance type, and discharge status using the Cochran-Armitage trend test.
We constructed three models to assess the association between AF and patient-specific characteristics and hospital and clinical factors. We used a univariate logistic regression analysis (model 1) to identify the putative factors associated with AF. Hospital demographics, clinical factors, ECI score, and CHA2DS2-VASc score are included in model 2.
Several models were performed to elucidate the associations between in-hospital mortality, LOS, and hospitalization cost and AF. In model A, we used AF as the single predictor to identify the risk associated with in-hospital mortality, LOS, and hospitalization cost. To perform a sensitivity analysis, we adjusted for demographic factors, hospital factors, ECI, anticoagulants, and CHA2DS2-VASC score in model B. For binary outcomes, we used unconditional logistic regression. Since LOS and hospitalization cost were violations of normality, the log-transformation was used. In order to analyze whether stroke or bleeding was linked to the relationship between AF and in-hospital mortality, we used stroke and bleeding as mediators respectively to construct two mediation models (adjusted for demographic factors and hospital factors). The models were also used to evaluate the trends in in-hospital mortality, LOS, and hospitalization cost in critically ill patients with and without comorbid AF. Using year as a continuous predictor, we used logistic regression to analyze in-hospital mortality and linear regression to analyze LOS and hospitalization cost.
Two-sided p values ≤ 0.05 were considered significant. All statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
RESULTS
Baseline characteristics
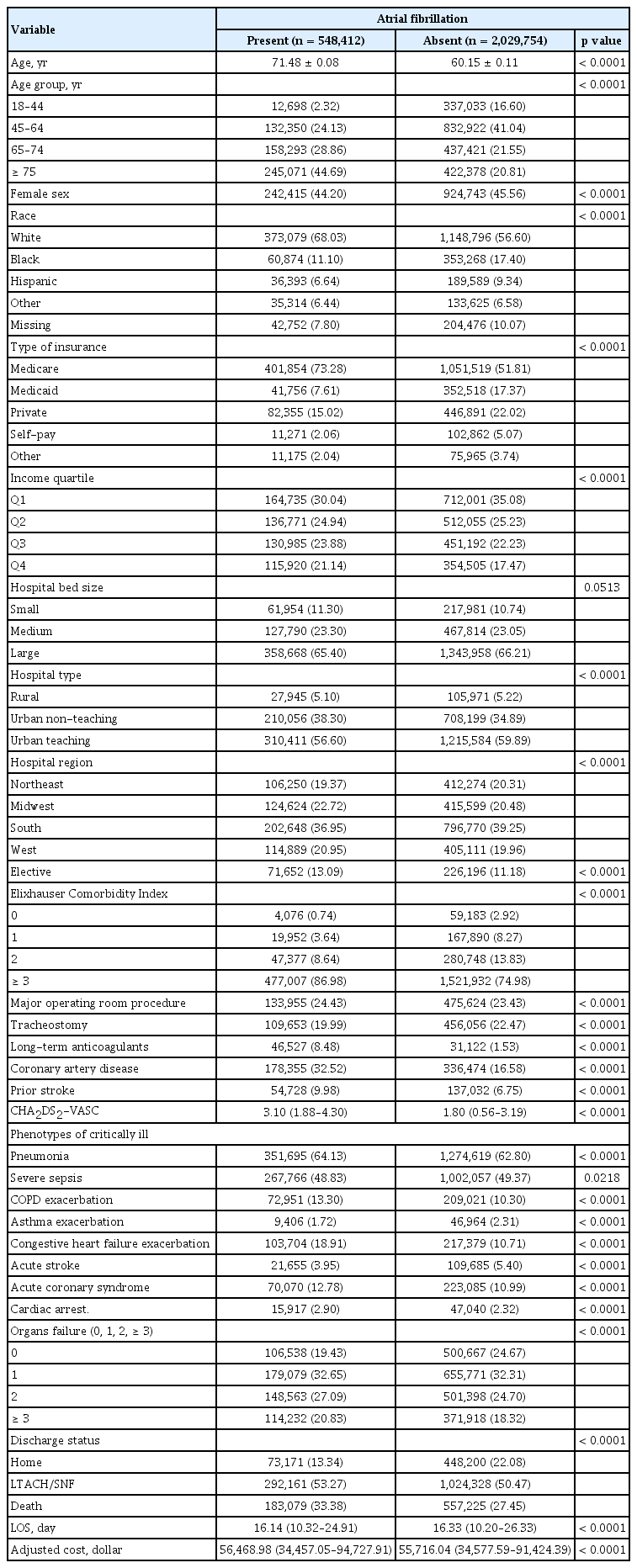
We identified a weighted sample of 2,578,165 critically ill patients from 2008 to 2014, of whom 21.27% had a diagnosis of AF. The characteristics of patients with AF versus those without are displayed in Table 1. Patients with AF were more likely to be older (71.48 years vs. 60.15 years, p < 0.0001), more likely to be of white race (68.03% vs. 56.60%, p < 0.0001), more likely to be covered by Medicare (73.28% vs. 51.81%, p < 0.0001), and more likely to have a higher income (76th to 100th percentile: 21.14% vs. 17.47%, p < 0.0001).
Trends of AF prevalence in subgroups
The AF prevalence in critically ill patients increased from 14.63% in 2008 to 24.43 % in 2014 (p for trend < 0.0001). When we excluded the patients who had undergone cardiovascular operations, the trends of AF increased from 14.26% in 2008 to 23.42 % in 2014 (p for trend < 0.0001). Fig. 1 and Supplementary Table 3 show the temporal trends of AF prevalence in critically ill patients in subgroups categorized by age, sex, race, and insurance. Across all subgroups, AF prevalence steadily increased from 2008 to 2014 (p for trend < 0.0001). The AF prevalence was higher in patients older than 75 years and higher in men than in women (25.14% vs. 23.54%, respectively). AF was more prevalent in people of white race and patients with Medicare. The temporal trends of AF in subgroups categorized by organ failure type also increased consistently over the study period (p for trend < 0.0001). A higher prevalence of renal failure (45.80% to 65.51%), neurologic failure (21.22% to 37.31%), and metabolic failure (14.85% to 28.47%) was noted (Supplementary Table 4). Amongst different phenotypes of critically ill patients, the prevalence of AF increased in the patients with severe sepsis (38.76% to 53.14%), asthma exacerbation (1.59% to 1.83%), congestive heart failure exacerbation (7.98% to 26.42%), acute stroke (3.77% to 4.37%), and cardiac arrest (2.36% to 3.41%) (Supplementary Table 5).
Trends in risk predictors and outcomes with or without AF
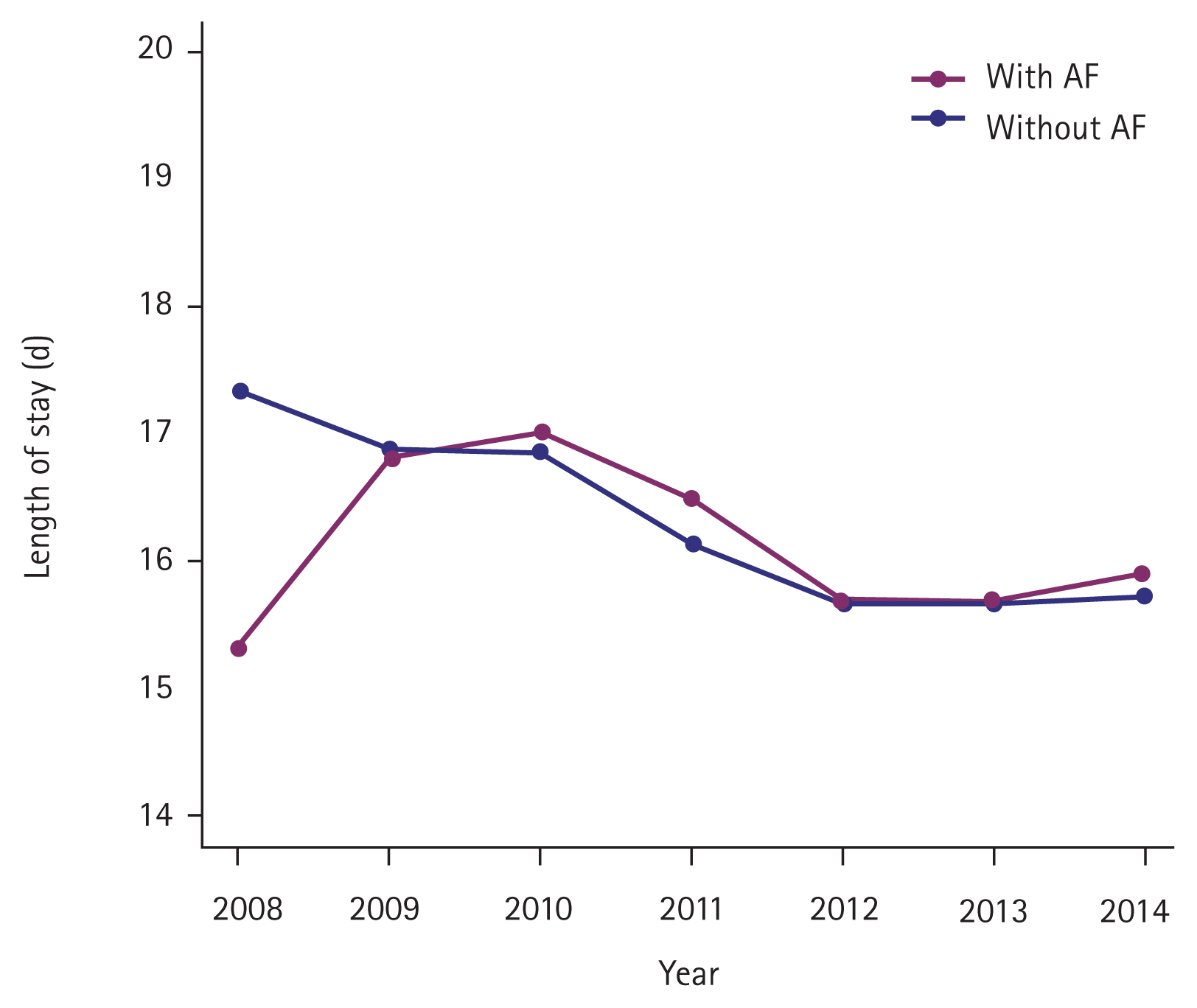
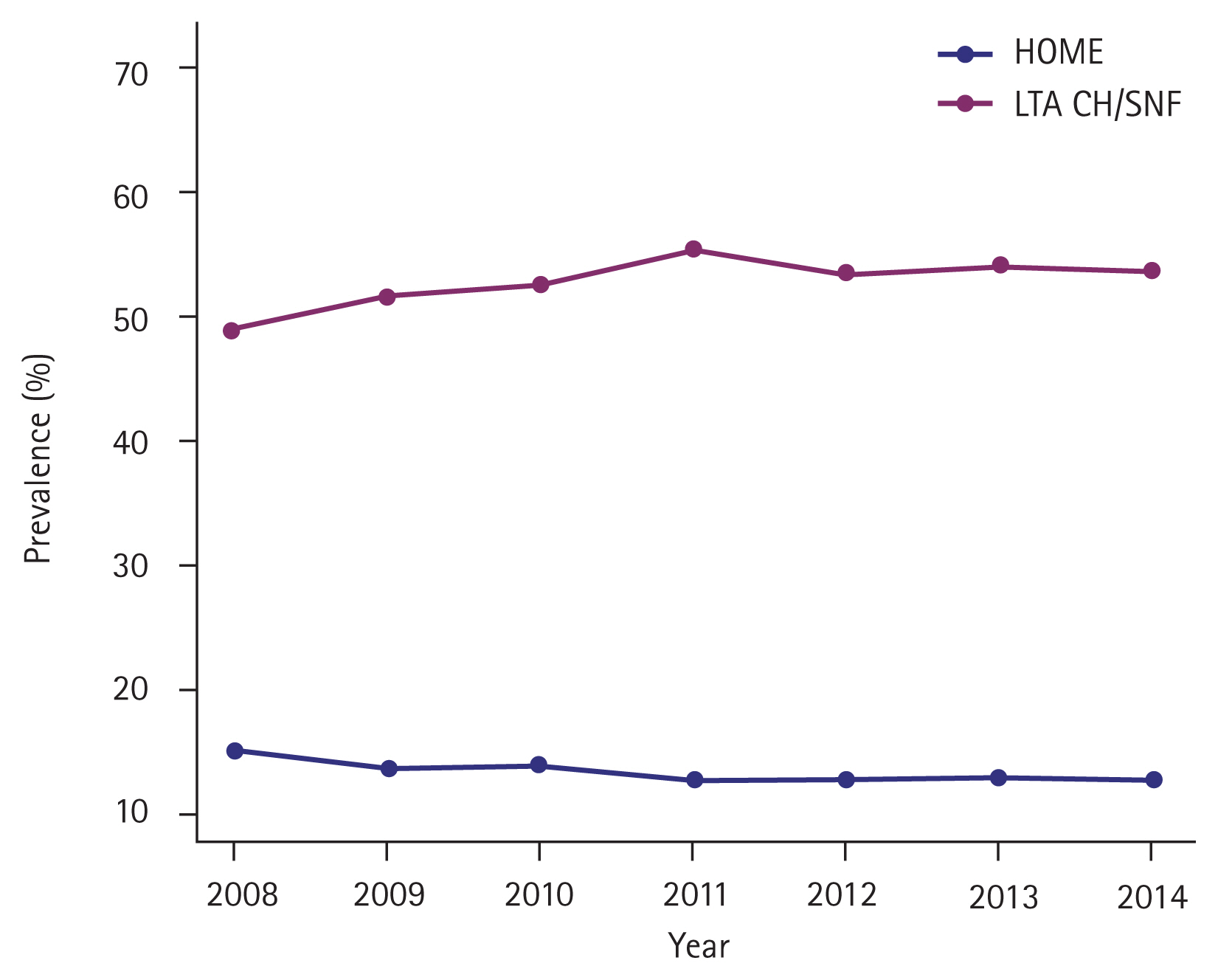
Temporal trends in risk predictor profiles among critically ill patients with AF versus without are shown in Supplementary Fig. 1. The prevalence of all risk predictor profiles increased consistently from 2008 to 2014 (p for trend < 0.0001) and patients with AF had a higher prevalence. On the contrary, in-hospital mortality decreased consistently regardless of AF status (with AF: 35.85% to 33.20%, p for trend < 0.0001; without AF: 30.11% to 26.90%, p for trend < 0.0001) (Fig. 2, Supplementary Table 6). However, the trend of LOS increased in patients with AF (15.33 to 15.88 days, p for trend = 0.6211) and decreased in patients without AF (17.32 to 15.71 days, p for trend = 0.0015) (Fig. 3, Supplementary Table 7). Declining in-hospital mortality was accompanied by changing discharge status for critically ill patients with AF, with a significant increase to LTACH/SNF (49.27% to 53.87%, p for trend < 0.0001) and a decrease in discharge to home (14.91% to 12.94%, p for trend < 0.0001) (Fig. 4, Supplementary Table 8).
Association of AF with patient and hospital characteristics in critical illness
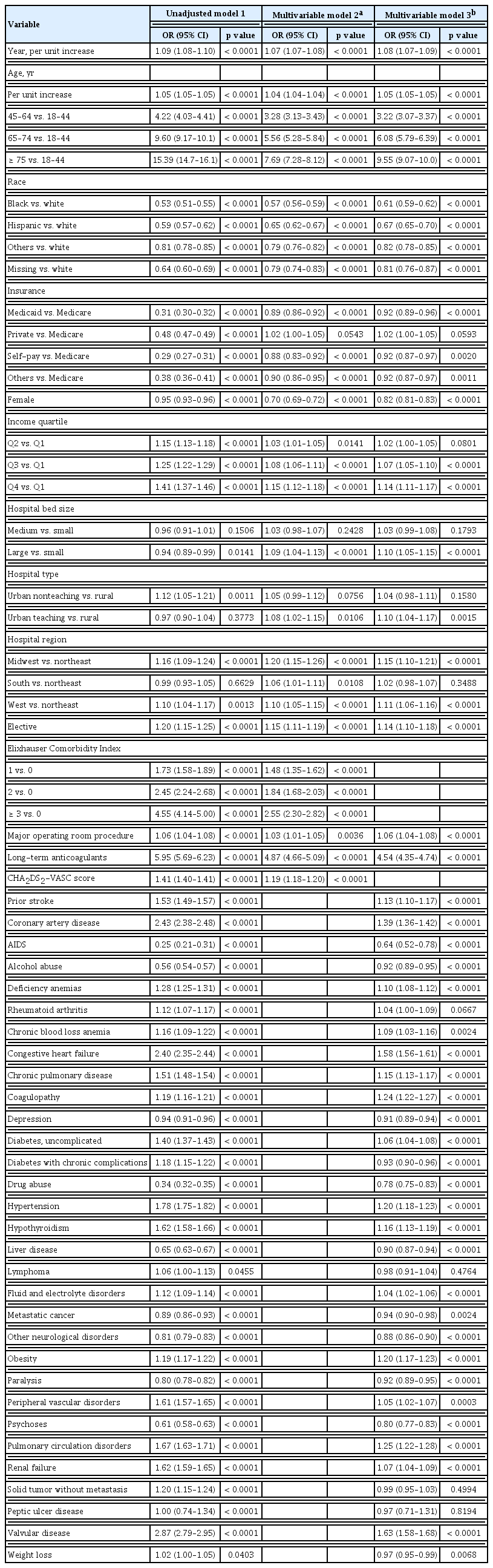
The temporally increasing trend of AF prevalence persisted over the study period after multivariable adjustment. The prevalence of AF increased by 8% each year from 2008 to 2014 (odds ratio [OR], 1.08; 95% confidence interval [CI], 1.07 to 1.09) (Table 2). Age older than 75 years, white race, Medicare insurance, male sex, higher income, urban teaching hospital, larger hospital bed size,, major operating room procedure, higher ECI score, higher CHA2DS2-VASc score, prior stroke, coronary artery disease, and long-term anticoagulant use were significantly associated with the increased prevalence of AF. Among the comorbidities, deficiency anemia, coagulopathy, hypertension, hypothyroidism, fluid and electrolyte disorders, renal replacement therapy use, obesity, chronic renal failure, and valvular disease were more significantly associated with increased prevalence of AF (Table 2, Supplementary Table 9).
Association between AF and clinical outcomes
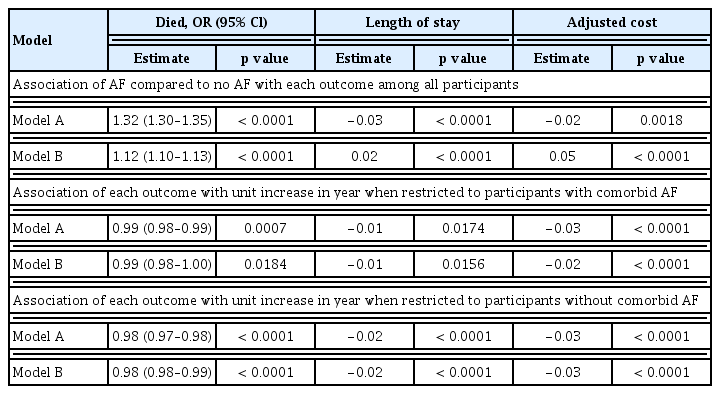
The unadjusted in-hospital mortality rate was significantly higher in patients with AF than in those without AF (OR, 1.32; 95% CI, 1.30 to 1.35) (Table 3). When stroke was used as a mediator, only 4.20% of the impact of AF on in-hospital mortality could be attributed to stroke. When bleeding was used as a mediator, only 0.03% of the impact of AF on in-hospital mortality could be attributed to bleeding. After multivariable adjustment, patients with AF still had a 10% higher risk than those without AF (OR, 1.12; 95% CI, 1.10 to 1.13), but the risk of death decreased each year regardless of AF diagnosis.
In the unadjusted regression model, patents with AF had a shorter LOS (−3%, p < 0.0001) and lower hospitalization cost (2%, p = 0.0018). However, after multivariable adjustment, an AF diagnosis was still linked to a prolonged LOS (2%, p < 0.0001) and higher hospitalization cost (5%, p < 0.0001). The LOS (−1%, p < 0.0001) and hospitalization cost (−2%, p < 0.0001) in critically ill patients with AF also decreased each year.
DISCUSSION
Our analysis is the first to evaluate the temporal trends of AF prevalence in adult critically ill patients in the US. Many prior analyses studied the incidence of AF in critically ill patients [3,5,18] but were limited by sample size and lacked the required granularity to evaluate the AF trend. We found that the proportion of critically ill patients with comorbid AF increased from 14.63% in 2008 to 24.43% in 2014. In this analysis of over 2 million critically ill patients, close to 1 in 5 (21.27%) had a diagnosis of AF, a value that was nearly 10-fold higher than that in the general population [1]. This rate is higher than the prevalence of AF reported by previous studies, which was 4.5% to 15% in intensive care unit (ICU) patients [19–22], but is lower as per the Seguin and Launey [23] report (46%). The prevalence of AF in critically ill patients increased with age; over 70% of affected patients were over 65 years. The prevalence was also greater among patients who were white, male, and covered by Medicare, a finding that is consistent with a previous report of critically ill patients [24].
With population aging and the increasing prevalence of comorbidities, the prevalence of AF is anticipated to increase [25]. Our analysis indicated that the prevalence of AF increased significantly over the study period, which is consistent with previous studies [4]. The increased prevalence of AF in critically ill patients may be associated with several factors. First, with the development of medical treatments, improvements in survival result in more hospitalizations of critically ill patients, although in-hospital mortality rates are declining [26]. Second, the trend of increased AF prevalence may also be the result of advances in detection and surveillance, while the improved awareness of AF among physicians increased the prevalence of AF as well [27]. Third, the aging of the population and the increase in comorbidities in the general US population also increases AF risk factors [28–30]. The same results were seen in patients with end-stage renal disease and end-stage chronic obstructive pulmonary disease [31,32].
AF in critically ill patients may be new-onset or recurrent [33]. Advanced age is considered one of the most potent risk factors for independently predicting AF [34]. In our study, compared to patients younger than 45 years, those older than 75 years were nearly 10-fold more likely to develop AF. In the meantime, each additional year of age was associated with an 8% increase in the risk of developing AF. Admission to a larger urban teaching hospital was significantly associated with AF relative to small and rural hospitals. Larger urban teaching hospitals receive more critically ill patients and have advanced detection and surveillance capabilities [35]. We found that patients with an ECI > 3 were 2.5-fold more likely to develop AF than those with an ECI of 0. Several comorbidities common in critically ill patients are substrates for AF occurrence and potentially impact patient prognosis [36]. Duby et al. [37] reported that the risk of AF in critically ill patients with congestive heart failure, hypertension, or heart disease was 4 to 7 times higher than that of patients without these comorbidities. We found that a higher CHA2DS2-VASc score was significantly associated with the increased prevalence of AF. As a scoring system for predicting cardioembolic risk, CHA2DS2-VASc scores can help clinicians make decisions about antithrombotic therapy, although this has not been validated in critical illness [38].
In our study, the in-hospital mortality decreased from 35.85% to 33.20%. It is possible that treatment advances led to improved survival in critically ill patients. Compared to patients without AF, those with AF had a 10% higher risk of in-hospital mortality, which is lower than that of previous studies [5,39]. Arrigo et al. [5] demonstrated an association between AF and increased risk of in ICU and post-ICU mortality using a prospective observational multi-center cohort study. A prospective single-center study also identified AF as an independent predictor for in-hospital mortality in critical illness [24]. However, their results are limited by small sample sizes. AF can be triggered by accelerated atrial remodeling and arrhythmias, while acute loss of atrial contraction and ventricular tachycardia often result in reduced cardiac output and hemodynamic impairment [40]. Previous studies have showed an increased risk of embolic stroke and heart failure in patients with AF, which can lead to clinical deterioration and mortality [36,41]. Alternatively, AF may simply be a marker of multiple organ failure, leading to increase in-hospital mortality [42]. However, the evidence for AF as an independent risk factor for in-hospital mortality remains limited and further studies are needed. Due to database limitations, our study was limited to in-hospital mortality and was not able to study short-term or long-term post-discharge mortality. We found that patients with AF had a 2% higher risk of prolonged LOS and 4% higher relative hospitalization costs than those without AF; that is, AF exerts a negative impact on hospitalization outcomes.
Managing AF in critically ill patients is challenging, and the strategies used in outpatients may not have the same effect [43]. Meanwhile, the efficacy of antiarrhythmic therapies may be reduced [44], though both rate and rhythm controlling agents are frequently used in the management of critically ill patients. Due to procoagulant activity and ongoing inflammation, the risk of thrombus formation and embolization is higher in critically ill patients [45]. Although guidelines and recommendations are lacking, AF in critically ill patients will trigger anticoagulation [46]. Considering the need for urgent surgery or risk for coagulopathy in the ICU, the choice of anticoagulant is a big problem for physicians, and we cannot exclude the association between mortality and anticoagulant use in critically ill patients [42].
Our study has some strengths. To our knowledge, it is the newest assessment of temporal trends of AF in critically ill patients in the US. Previous studies identified the association between AF and morbidity outcomes and in-hospital mortality, but they were insufficiently sized. To analyze sensitivity, we constructed three models to access the association between AF and patient-specific characteristics and hospital and clinical factors as well as six models to elucidate the associations between in-hospital mortality, LOS, and hospitalization cost and AF. However, our study also has several limitations. We used the ICD-9-CM procedure code to capture the critically ill status. However, the continuous mechanical ventilation does not equal critically ill. There are critically ill patients who are not mechanically ventilated and mechanically ventilated patients who are not critically ill. It’s specific but not sensitive to representative critically ill [12,47]. In addition, we couldn’t evaluate the frequency of new-onset AF, history of recurrent AF or permanent AF in this setting of patients. Compared to true clinical data, the NIS contains administrative data that may be subject to coding errors [48]. Meanwhile, we are not sure that the temporal trend observed in AF prevalence is not related to coding habits. Because mechanical ventilation is a predictive risk factor for AF and the need for mechanical ventilation are significantly greater in patients with AF [49], so the AF prevalence in our study may be higher than estimated. In addition, NIS cannot track personal records or information, including post-discharge survival status. These analyses were based on inpatient participants, so the conclusions can be generalized to inpatient settings. Long-term or post-discharge mortality is beyond the scope of our study, and future studies need to take these aspects into further consideration.
In conclusion, we found that the proportion of critically ill patients with comorbid AF steadily increased from 2008 to 2014 in a large national sample. AF was associated with higher in-hospital mortality, longer LOS, and higher hospitalization costs. More knowledge about the incidence and risk predictors of AF can benefit critical patients; thus, high-level evidence is needed to guide the management of AF in critically ill patients.
KEY MESSAGE
1. The newest assessment of temporal trends of atrial fibrillation (AF) in critically ill patients in the United States.
2. Prevalence of comorbid AF is increasing, particularly in older patients.
3. AF in critical illness was a risk factor for in-hospital death.
Notes
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This work was supported by The Fourth Round of Shanghai Three-year Action Plan on Public Health Discipline and Talent Program: Evidence-based Public Health and Health Economics (No. 15GWZK0901).