Methods for the morphological and functional evaluation of microvascular damage in systemic sclerosis
Article information
Abstract
Systemic sclerosis is a connective tissue disease characterized by alterations in microvascular structure and function. In these patients, numerous studies have demonstrated a relationship between capillary morphology and peripheral blood perfusion. Nailfold videocapillaroscopy reveals the peripheral microvascular morphology and thus allows classification and scoring of capillary abnormalities with respect to different microangiopathy patterns (early, active, and late). Laser Doppler flowmetry and laser speckle contrast analysis can be used to estimate cutaneous blood flow through microvessels and to assess and quantify blood perfusion at peripheral sites. These two methods are also used to identify changes in digital blood perfusion after the infusion of vasodilators.
INTRODUCTION
Systemic sclerosis (SSc) is an autoimmune connective tissue disease characterized by early impairment of the microvascular system, leading to functional and organic manifestations, including digital ulcers [1,2]. Progressive deficiency in the vasodilatory capacity of the vessels, along with intimal fibroproliferation, thrombosis of the digital arteries, and tissue ischemia have been postulated as mechanisms explaining the persistent vascular involvement [3,4]. Microvascular damage and dysfunction are the earliest morphological and functional markers of SSc and are correlated with prognosis and therapy planning [5].
The vascular changes occur during the early stage of SSc, with Raynaud phenomenon (RP) as the clinical expression of abnormal blood flow regulation due to altered microvascular structure and function [5,6]. Therefore, morphological and functional assessment of the peripheral microvasculature in SSc patients is a diagnostic necessity, with prognostic and therapeutic consequences [5]. Nailfold microangiopathy can be evaluated easily by nailfold videocapillaroscopy (NVC). Laser Doppler flowmetry (LDF) allows evaluation of the extent of perfusion in the peripheral microcirculation [7,8], while laser speckle contrast analysis (LASCA) is a relatively new method that permits quantitative assessment of the peripheral perfusion in larger areas of different body sites [9,10,11]. These three methods for evaluating SSc patients are described below.
NAILFOLD VIDEOCAPILLAROSCOPY
Nailfold microangiopathy can be evaluated easily using NVC. This non-invasive, well-accepted, safe method makes use of a magnification system (best at ×200 magnification) [12,13]. The video-capillaroscope has an optical/digital probe that can be moved over the surface of the fingernails from the second to the fifth digits [5,14,15].
NVC is used to evaluate the morphology of nailfold dermal papillary capillaries, thus allowing recognition of the qualitative, morphological patterns of microangiopathy (early, active, and late) (Fig. 1) [16].

Nailfoild videocapillaroscopic (×200) patterns of microangiopathy: (A) normal versus (B) early, (C) active, and (D) late patterns. SSc, systemic sclerosis.
The early SSc pattern is characterized by the presence of few giant capillaries, few capillary microhemorrhages, no evident capillary loss, and a relatively well-preserved capillary distribution. The active NVC SSc pattern is a marker of disease progression. It is characterized by frequent giant capillaries, frequent capillary microhemorrhages, moderate capillary loss, absent or mildly ramified capillaries, and a mild disorganization of the capillary architecture. In the late pattern, giant capillaries and microhemorrhages are nearly absent, but there is severe capillary loss, with evident extensive avascular areas, ramified or bushy capillaries, and severe disorganization of the normal capillary array [16].
In patients with RP, NVC can be used to distinguish the primary from the secondary forms of the disease [5,6]. Under normal conditions or in the presence of primary RP, NVC shows a regular array of capillary loops along the nailfold bed, no abnormal enlargements, and no capillary loss [5,17,18]. In secondary RP, the morphological signs of giant capillaries, microhemorrhages, capillary loss, the presence of avascular areas, and angiogenesis are indicative of microvascular damage. These sequential capillaroscopic changes are typical of the microvascular involvement observed in more than 95% of SSc patients and are described by the term "SSc pattern" [5,19,20].
NVC also provides a semiquantitative assessment of microvascular damage that can be used to follow its progression, based on a semiquantitative rating scale (score 0 to 3) for the assessment of microvascular parameters [21]. The microangiopathy evolution score [14,15] is the sum of the three individual scores: capillary loss, disorganization of the microvascular array, and capillary ramifications.
Qualitative and semiquantitative assessments also have been used to predict the clinical complications of SSc [15,21,22]. For example, patients with a reduced capillary density are at high risk of developing digital skin ulcers and pulmonary arterial hypertension [15,23].
Inclusion of the NVC patterns in the diagnostic procedure could increase the sensitivity of the classification criteria for SSc, such as the new European League Against Rheumatism and American College of Rheumatology criteria [24]. Moreover, NVC-based assessment of microvascular structure may be useful in the evaluation of drug effects during the treatment of SSc patients [25].
PERIPHERAL BLOOD FLOW EVALUATION BY LDF
LDF is a user-friendly, well-accepted, and non-invasive assessment of microvascular flow at a single skin point. It provides an index of skin perfusion by measuring the Doppler shift induced by coherent light scattering caused by moving red blood cells [26]. An example of peripheral blood perfusion (BP) evaluated by LDF is shown in Fig. 2.
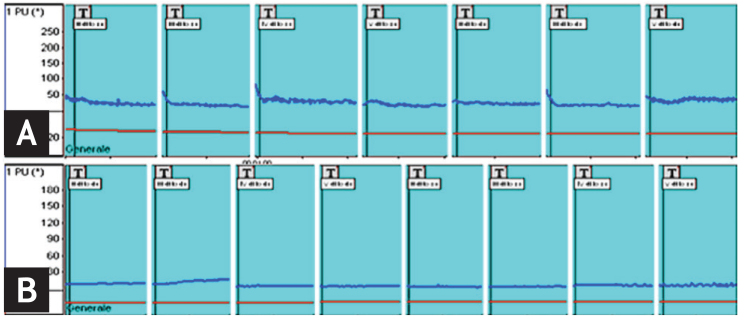
Laser Doppler flowmetry: 1-minute analysis of blood perfusion in a healthy subject (A) and in a patient with systemic sclerosis (nailfold videocapillaroscopy late pattern) (B). Entity of the blood flow reported as perfusion units (PU).
The instrument comprises a heatable probe, an optic fiber connector, an instrument that detects both tissue perfusion and skin temperature, and a computer to evaluate the data. LDF measures local perfusion in a cubic millimeter of tissue (capillaries, arterioles, venules, shunts), which is recorded in perfusion units (PUs), for example at the level of the fingertips. Basal finger temperature (usually at the level of the fingertips, from the second to fifth digits of both hands) is measured first, after which the probe is heated to 36℃ and capillary dilation capacity evaluated [8].
In the study by Binaghi et al. [27], a significant correlation was found between the digital flow parameters and capillaroscopy score in SSc patients. The results suggested the relevance of LDF in evaluating apparently primary RP during a standardized thermic test, because even in the absence of a definite scleroderma-like capillaroscopic pattern, flowmetry can be used to identify patients with secondary RP [27].
Some studies have demonstrated that SSc patients have lower blood flow compared with healthy subjects, and that among the patients evaluated by NVC, those with a late SSc microangiopathy pattern have a lower rate of blood flow on LDF compared with those with the early or active pattern [8,11,28,29]. SSc patients were also shown to have abnormal microvascular regulatory responses to heat stimulation, suggesting that the degree of endothelial dysfunction is related to the degree of peripheral microvascular involvement [8].
In addition, LDF can assess changes in peripheral BP during treatment with vasodilator drugs either within a few days or over a period of years [8,30]. However, one problem with the technique is the large site-to-site variation, which limits its efficacy in comparing blood flow among sites and in monitoring changes over time [26]. A new technique, laser Doppler imaging, which evaluates flow over an anatomic area might overcome this problem [7].
PERIPHERAL BLOOD FLOW EVALUATION BY LASCA
This relatively new technique quantifies BP over an area rather than only at a single point, and thus it may obviate the problems that occur when a single probe is used, especially site-to-site variability. LASCA is based on the principle that when an object is illuminated by laser light, the backscattered light will form a random interference pattern made up of dark and bright areas. This so-called speckle pattern is stationary, but when movement occurs, as in red blood cells within a tissue, the speckle pattern changes over time. These changes can be recorded by a detector camera [9,11]. An advantage of LASCA is that it allows commonly used settings (e.g., working distance, point density, frame rate resolution, and the width and height of the measurement area) to be saved for repeated use [11]. Moreover, LASCA does not require direct contact between the probe and the skin, which might in itself affect blood flow. It also evaluates the fluxes in different regions of interest (ROIs) and time regions of interest may also be created [11].
Using LASCA, fingertip BP, during or after recording (for example, at the fingertips and dorsum of the hand), is evaluated and the results reported in PUs. The different values can be represented graphically, and the BP ROI, whether depicted in color or as intensity images, are superimposed (Fig. 3) [11].

Imaging of cutaneous blood perfusion using laser speckle contrast analysis at the level of the palmar region in a healthy subject (A) and in a patient with systemic sclerosis (nailfold videocapillaroscopy late pattern) (B).
More recently, LASCA has been applied in research on RP and SSc [10,11]. One such study demonstrated that peripheral BP, evaluated by LDF and LASCA, correlates with the extent of the microangiopathy detected by NVC [11]. As noted above, when patients whose disease had been classified using NVC were then evaluated using LDF and LASCA, those with the late SSc microangiopathy pattern had a lower blood flow than that of patients with early or active SSc patterns [11].
A very recent study showed that BP, when monitored by LASCA, is significantly lower in SSc patients than in healthy subjects at the level of the fingertips, periungual areas, and palms of the hands. In addition, in SSc patients, there is a statistically significant negative correlation between the extent of nailfold microangiopathy and BP at the level of those same skin areas [31]. Similar combined investigations might optimize the follow-up of SSc patients [11].
Whether the combination of all three techniques (NVC, LDF, and LASCA) improves the classification of SSc, including distinguishing patients with SSc from those with primary RP and from healthy controls, has yet to be confirmed by the appropriate studies. However, thus far, when only a single microcirculatory tool is used, NVC seems to be the best discriminator [32,33].
CONCLUSIONS
Peripheral microvascular morphology can be assessed using NVC, which also allows the classification and scoring of capillary abnormalities. NVC is the best method to distinguish patients with secondary RP from those with primary RP and from healthy subjects [32,33]. NVC, LDF, and LASCA together constitute an essential set of safe and noninvasive tools for the early diagnosis and reliable quantification of SSc, the response to therapy, and disease progression [22]. Extensive employment of these tools in rheumatology clinics is recommended accordingly [8,25,30,34].
Notes
No potential conflict of interest relevant to this article was reported.