Diagnosis and therapy of multiple myeloma
Article information
Abstract
Many advances in the treatment of multiple myeloma have been made due to the use of transplantation and the introduction of novel agents including thalidomide, lenalidomide, and bortezomib. The first step is recognizing the symptoms and starting prompt treatment. Different strategies should be selected for young and elderly subjects. Young patients are commonly eligible for transplantation, which is now considered the standard approach for this setting, and various inductions therapies containing novel agents are available before transplantation. Elderly patients are usually not eligible for transplantation, and gentler approaches with new drugs combinations are used for their treatment.
EPIDEMIOLOGY
Multiple myeloma is a neoplastic plasma-cell disorder characterized by clonal proliferation of malignant plasma cells in the bone marrow, and the presence of monoclonal protein (M protein) in the blood or urine. The disease is associated with organ dysfunction [1].
Multiple myeloma accounts for approximately 1% of neoplasms and 13% of hematologic cancers. In Western countries, the annual age-adjusted incidence is 5.6 cases per 100,000 persons [2]. The median age at diagnosis for multiple myeloma was 69 years from 2005 to 2009. No cases were diagnosed in patients < 20 years; 0.5% in those 20 to 34 years; 3.2% between 35 to 44 years; 11.8% between 45 to 54 years; 22.3% between 55 to 64 years; 26.9% between 65 to 74 years; 25.6% between 75 to 84 years; and 9.6% > 85 years of age [2,3]. The treatment paradigm for multiple myeloma has changed considerably and extended overall survival (OS) times have been observed due to the introduction of autologous stem-cell transplantation and the availability of novel agents, such as the immunomodulatory agents thalidomide and lenalidomide and the proteasome inhibitor bortezomib [3-5]. Overall, 5-year relative survival increased from 28.8% to 34.7% (p < 0.001), and 10-year relative survival increased from 11.1% to 17.4% (p < 0.001) between 1990 to 1992 and 2002 to 2004. More pronounced increases were observed in the age group < 50 years, leading to a 10-year relative survival rate of 41.3% in 2002 to 2004, and in the age group 50 to 59 years, leading to a 10-year relative survival rate of 28.6% in 2002 to 2004. Only moderate improvement was seen in older patients [4].
DIAGNOSIS
The diagnosis of multiple myeloma is based on the presence of at least 10% clonal bone marrow plasma cells and M protein in serum or urine. Multiple myeloma can be active/symptomatic or smoldering/asymptomatic. Therapy should be started immediately for symptomatic disease, whereas asymptomatic disease requires only close monitoring, as early treatment in asymptomatic patients has so far shown no benefit [5-7]. Symptomatic disease is characterized by the presence of end-organ damage (CRAB features): hypercalcemia [8]; renal dysfunction, occurring in 20% to 40% of patients with newly diagnosed multiple myeloma [8,9]; anemia, reported in approximately 73% of patients at diagnosis [10]; and bone lesions, reported in almost 80% of patients with newly diagnosed multiple myeloma [8]. Notably, patients with symptomatic disease have an increased risk of infection; however, this decreases with response to therapy [11].
A detailed medical history and physical examination are recommended to diagnose multiple myeloma. Laboratory tests are also necessary, including a complete blood count, creatinine and calcium, serum and protein electrophoresis with immunofixation, quantification of M protein, and a 24-hour urine Bence-Jones protein evaluation are fundamental parts of the diagnostic procedure. In addition, the free light-chain (FLC) assay is suggested in patients with plasma cell disorder at diagnosis and particularly in those with nonsecretory myeloma (absence of the monoclonal component), small amounts of monoclonal component (oligosecretory myeloma), and light-chain only myeloma [12]. Bone marrow tests (trephine biopsy plus aspirate for cytogenetic analysis or fluorescence in situ hybridization [FISH]) should always be performed [7,13].
Osteolytic lesions can be detected by a skeletal X-ray survey. Magnetic resonance imaging (MRI) may be necessary in cases of negative radiographs [14]. MRI and positron emission tomography integrated with computed tomography (PET/CT) may be useful in select circumstances, such as to detect soft tissue lesions arising from bone lesions, spinal cord compression, asymptomatic lesions, and to evaluate a painful skeletal area [15]. MRI is indicated for the initial assessment and follow-up of nonsecretory myeloma and to detect occult lesions in patients with smoldering myeloma [16].
PROGNOSIS
Although the prognostic value of Durie and Salmon stages is not high, this staging continues to be used, as it is easy to perform. Three stages are defined, and > 70% of patients with multiple myeloma present with stage III, which is the worst stage [17]. The International Staging System (ISS) is a new, simple, and more widely used classification that only considers β-2 microglobulin level, which is closely related to renal function, and to the tumor mass, and albumin level [18].
The presence of chromosomal abnormalities also has a prognostic role in multiple myeloma. Detection of t(4;14), t(14;16), t(14;20), chromosome 1 abnormalities, and del17p by FISH are associated with a poor prognosis. Conversely, the patient is not considered at high risk if only a 13q deletion is present with no other abnormalities. The combination of FISH data and ISS staging improves the risk assessment [19].
New prognostic markers are now emerging. For example, an abnormal k/λ FLC ratio is also predictive of poor outcome [20]. Gene expression profiling and PET/CT seem to play a role in the prognosis of patients with multiple myeloma as well; however, further investigation is needed [21-23].
THERAPY FOR YOUNG PATIENTS
Patients < 65 years of age with no comorbidities are usually considered eligible for high-dose chemotherapy and autologous stem cell transplantation (ASCT). Induction therapy containing novel agents should be administered before transplantation [24]. Patients eligible for ASCT typically receive a limited number of induction cycles to reduce tumor burden followed by the collection of peripheral blood stem cells and then receive single or double ASCT conditioned with 200 mg/m2 melphalan.
Whether single or double ASCT is better remains controversial [25-29]. Considering that achieving a deeper response is associated with a longer OS [30-32], tandem ASCT should be suggested in patients who fail to achieve at least a very good partial response (VGPR) after the first ASCT [25,26,28,32,33].
Thalidomide-containing therapies
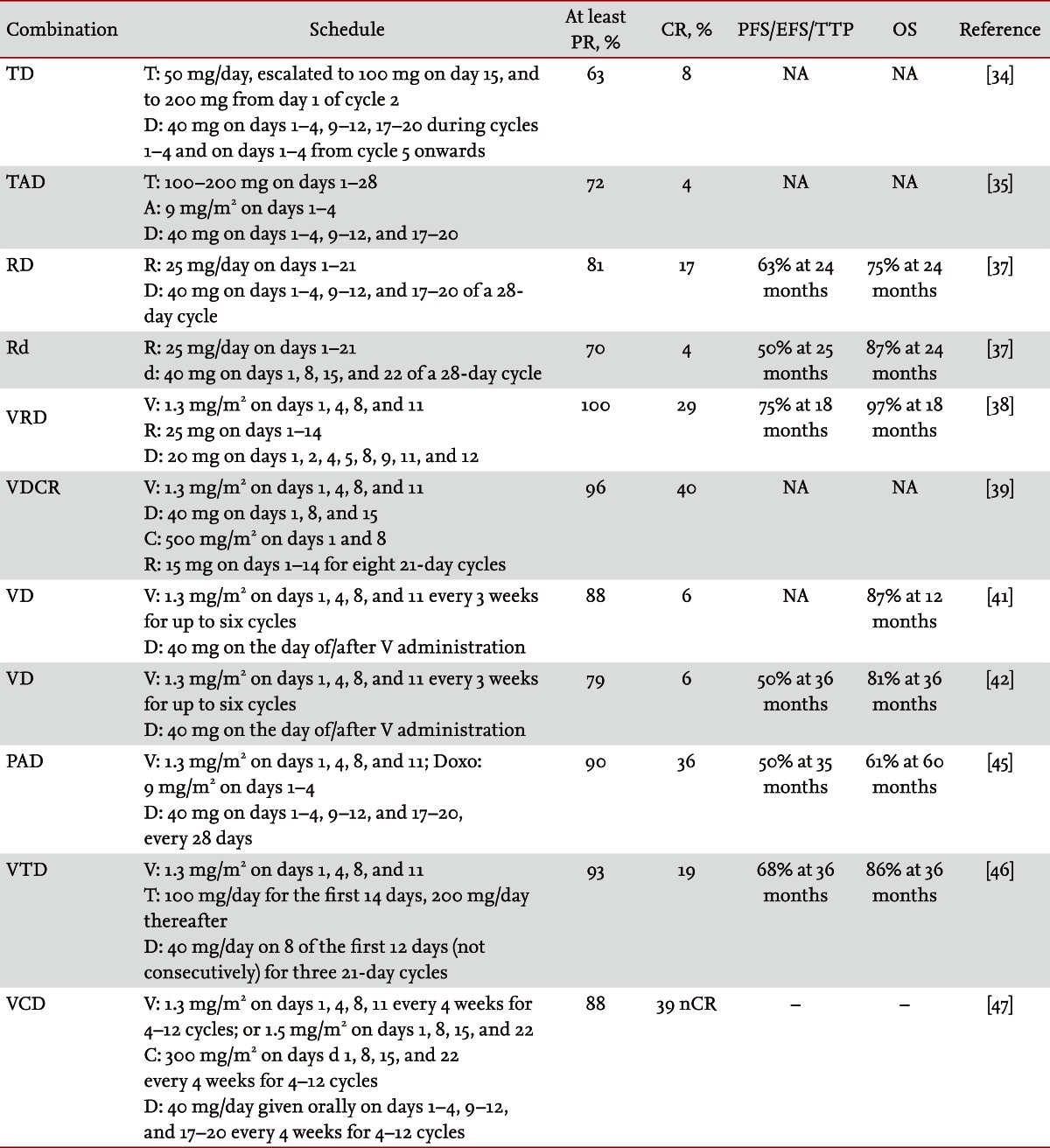
Although vincristine-doxorubicin-dexamethasone (VAD) was long considered the standard induction therapy before ASCT, new and more effective combinations are used today. The combination of thalidomide-dexamethasone (TD) is more effective than dexamethasone alone as induction therapy before ASCT (Table 1) [34]. The overall response rate is significantly higher with TD (63% vs. 46%; p < 0.001). Time to progression (TTP) was significantly longer with TD compared with that of dexamethasone alone (median, 22.6 months vs. 6.5 months; p < 0.001). Grade 4 adverse events were more frequent with TD than with dexamethasone alone (30.3% vs. 22.8%), and grade 3 to 4 deep vein thrombosis (DVT) was more common with TD (11.5% vs. 1.7%). As a result, TD has emerged as one of the most commonly used induction regimens before ASCT in the past decade. The good results obtained with the TD combination supports the use of additional cytotoxic drugs, such as doxorubicin (thalidomide-adriamycin-dexamethasone, TAD) or cyclophosphamide (cyclophosphamide-thalidomide-dexamethasone, CTD), in transplant-eligible patients. Both TAD and CTD followed by double ASCT provided a significantly higher VGPR/complete response (CR) rate and longer progression-free survival (PFS) compared with those reported with the VAD combination [35,36]. In particular, in the MRC Myeloma IX study [36], transplant-eligible patients were randomly allocated to receive CTD or cyclophosphamide plus VAD (CVAD) induction therapy; the overall response rate was higher with CTD than with CVAD (82.5% vs. 71.2%). Median PFS was 27 months with CTD versus 25 months with CVAD, and OS was comparable in both treatment arms. CTD was associated with more constipation and somnolence but less cytopenia compared with CVAD.
Lenalidomide-containing therapies
Lenalidomide is a derivative of thalidomide. The good results achieved with the TD combination provided the basis for combining lenalidomide with dexamethasone. A phase 3 study compared lenalidomide plus high-dose dexamethasone (RD) with lenalidomide plus low-dose dexamethasone (Rd) as an induction regimen for both young and elderly patients (eligible and ineligible for ASCT) [37]. RD showed higher response rates than those of Rd, with at least a VGPR rate of 42% versus 24%, respectively, but it was also more toxic and led to a higher early mortality rate. At the second interim analysis at 1 year, OS was better for those receiving Rd (96%) compared with those receiving RD (87%; p = 0.0002). Therefore, the trial was stopped and patients on high-dose therapy were crossed over to low-dose therapy. The more common toxicities included DVT (26% vs. 12%, with RD and Rd, respectively; p = 0.0003); infections including pneumonia (16% vs. 9%; p = 0.04); and fatigue (15% vs. 9%; p = 0.08).
RD with added bortezomib (VRD) was an effective and safe option in a phase 1 to 2 study [38]. All patients (100%) obtained at least a partial response (PR), including 30% CR, and 42% proceeded to transplantation. The estimated 18-month PFS and OS for the combined treatment with/without transplantation was 75% and 97%, respectively, with a median follow-up of 21 months. Grade 3 to 4 hematologic toxicities included neutropenia (9%) and thrombocytopenia (6%). The most common extrahematologic toxicities included grade 2 to 3 sensory neuropathy (80%) and fatigue (64%). DVT occurred in < 10%, and no treatment-related mortalities were observed. To date, there are no data to confirm the superiority of VRD over RD in terms of efficacy and outcome.
VRD plus cyclophosphamide (VCRD) has been recently evaluated in a phase 1 study in previously untreated patients with multiple myeloma [39]. Responses included at least a PR rate of 96%, and a CR rate of 40%. Follow-up was too short to assess PFS and OS. Interestingly, a phase 2 study found that, although the VCRD combination was associated with higher VGPR rate (58% vs. 51%) and CR (25% vs. 24%), VRD appeared less toxic and led to fewer discontinuations and treatment-related deaths. Thus, VRD may be preferred; however, further evaluation in a phase 3 study is needed [40].
Bortezomib-containing therapies
Bortezomib and dexamethasone (VD) is an effective and safe frontline approach to treating patients with multiple myeloma [41]. The VD combination has been given as induction therapy before ASCT in two clinical trials [42,43]. As a result, the at least VGPR rate increased from 30% before transplantation to 55% to 60% after ASCT. A phase 3 study compared the standard VAD regimen with VD as induction therapy in preparation for ASCT, followed by maintenance with lenalidomide [44]. Response rates were higher with the VD than the VAD treatment (≥ at least VGPR 38% vs. 15% after induction and 68% vs. 47% after double ASCT). Median PFS tended to be longer with VD (36 months vs. 30 months), but the difference was not statistically significant. The respective 3-year OS rates were 81.4% and 77.4%. The incidence of severe adverse events was similar between the two groups, but hematologic toxicity and deaths related to toxicity were more frequent with VAD. Conversely, rates of grade 2 (20.5% vs. 10.5%) and grades 3 to 4 (9.2% vs. 2.5%) peripheral neuropathy during induction through the first transplantation were significantly higher with VD than those with VAD.
A phase 3 study compared VAD followed by ASCT and thalidomide maintenance with bortezomib, doxorubicin, and dexamethasone (PAD) followed by ASCT and maintenance with bortezomib [45]. CR plus near CR rate was superior after PAD induction (15% vs. 31%; p < 0.001) and maintenance with bortezomib (34% vs. 49%; p < 0.001). After a median follow-up of 41 months, PFS was superior in the PAD arm (medians, 28 months vs. 35 months; p = 0.002). In a multivariate analysis, OS was also better with PAD. Nevertheless, adverse events were more frequent with PAD; grade 3 to 4 adverse events were detected in 54% treated with VAD versus 63% treated with PAD (p < 0.01), and the peripheral neuropathy rates were 10% with VAD and 24% with PAD (p < 0.001).
Another phase 3 study assessed the role of bortezomib plus TD (VTD) compared with TD as induction treatment before tandem ASCT followed by consolidation/maintenance with previous regimens [46]. The CR rates were 19% and 11% in the VTD and TD arms, respectively, after induction (p < 0.0001), and CR increased to 42% and 30%, respectively, after the second ASCT (p = 0.0004). Responses were also higher after VTD consolidation compared with that after TD consolidation (CR rate of 49% vs. 34%; p = 0.0012). The 3-year PFS was 68% for the VTD arm and 56% for the TD arm (p = 0.005) but the 3-year OS was similar (86% vs. 84%, respectively). More frequent adverse events, but with an incidence not > 10%, were skin rash, peripheral neuropathy, infection, and DVT.
Promising results were reported with bortezomib in combination with cyclophosphamide and dexamethasone (VCD) with a CR/near CR rate of 43%. Grade 3 peripheral neuropathy appeared in < 10% of patients and milder yet symptomatic peripheral neuropathy was quite common but no grade 4 was reported [47].
THERAPY FOR ELDERLY PATIENTS
Patients > 65 years, or younger patients with significant comorbidities, are usually considered ineligible for ASCT. Gentler approaches should be used for these patients, and dose adjustments should be made when required. Melphalan-prednisone (MP) was considered the standard of care for many years [48]. The introduction of novel agents has challenged this combination and new and more effective combinations are available.
Thalidomide-containing therapies
The TD combination is more effective than MP in elderly patients but is associated with a higher incidence of adverse events, treatment discontinuations, and nondisease-related mortality, mainly due to infections [49].
The role of thalidomide added to MP (MPT) has been assessed in six randomized studies (Table 2) [50-55]. A meta-analysis pooled data from 1,685 patients included in these trials to evaluate MPT efficacy [56]. The 2-year PFS was 42.5% for MPT and 28.4% for MP, and median OS was 39.3 months with MPT versus 32.7 months with MP. Improved responses were detected with MPT compared to those with MP; the at least VGPR rate was 25% for MPT versus 9% for MP. This meta-analysis confirmed that MPT improved OS and PFS in previously untreated patients with multiple myeloma. A safety meta-analysis based on the same trials was conducted on data from 1,680 patients [57]. In all six trials, the incidence of grade 3 to 4 adverse events was higher (at least 75%) during the first 6 months of treatment with both MPT and MP. Grade 3 to 4 hematologic toxicity increased with MPT (28% vs. 22%). Similarly more nonhematologic adverse events occurred with MPT than those with MP (39% vs. 17%). Grade 3 to 4 nonhematologic adverse events increased significantly in patients with poor performance status. Occurrence of grade 3 to 4 nonhematologic toxicities negatively impacted PFS and OS. ISS stage, high creatinine levels, poor performance status, and advanced age had a negative impact on OS. The positive results obtained with MPT in the six studies confirmed the role of this combination as a new standard of care for elderly patients.
A phase 3 trial also assessed the role of an attenuated CTD regimen compared with MP [58]. Median PFS was comparable in the two arms (12 to 13 months), as was OS (31 to 33 months). CTD was associated with better responses, with an overall response rate of 63.8% compared with 32.6% for MP (p < 0.001). CTD was particularly beneficial in subjects with a good cytogenetic profile by FISH. Common toxicities associated with CTD were constipation (41%), infection (32%), sensory neuropathy (24%), and DVT (16%). DVT decreased with thromboprophylaxis administration to patients receiving thalidomide. These data show that CTD is a feasible approach for selected elderly patients, particularly for standard-risk patients as assessed by FISH.
Lenalidomide-containing therapies
The RD versus Rd trial mentioned previously included both young and elderly patients [37]. Because more adverse events occurred when RD was given compared with Rd (DVT or pulmonary embolism: 26% vs. 12%; infections: 16% vs. 9%), particularly in patients > 65 years, Rd seems preferable for the elderly. However, RD remains a good option for patients with renal failure, hypercalcemia, pain, or spinal cord compression.
The role of lenalidomide has been assessed in a recent phase 3 trial. That study compared melphalan-prednisone-lenalidomide followed by lenalidomide maintenance (MPR-R), with MPR and MP [59]. PFS was longer with MPR-R compared with MPR and MP (31 months vs. 14 months vs. 13 months; p < 0.001). Yet, MPR induction did not improve PFS compared with MP in patients > 75 years of age. This was due to the increased incidence of toxicities associated with MPR, which led to more frequent dose adjustments in elderly patients. The 3-year OS rates were similar among the three treatment arms (70% vs. 62% vs. 66%). Neutropenia is a common event with lenalidomide, and grade 4 neutropenia occurred in 35% of MPR-R patients and 32% of MPR patients. There have been some concerns about the lenalidomide-related occurrence of second primary malignancies (SPMs); the 3-year SPM rate was 7% for both MPR-R and MPR, and 3% with MP. However, the benefits associated with MPR-R outweigh the increased risk of SPMs.
A phase 2 study including both young and elderly patients assessed the role of lenalidomide associated with another alkylant agent, cyclophosphamide, plus dexamethasone [60]. The 2-year PFS was 57%, the 2-year OS rate was 87%, and the at least VGPR rate was 30%. Neutropenia was common but easily manageable through cyclophosphamide dose adjustment. Fatigue was the most frequent nonhematologic adverse event. Thromboprophylaxis was recommended only for high-risk patients, and the rate of DVT was 15%. Future phase 3 trials are needed to validate the role of this combination.
Bortezomib-containing regimens
A phase 2 trial reported that the VD combination is a good therapeutic strategy for both young and elderly patients [41]. Median PFS was 21 months and the median OS was not reached in patients ineligible for ASCT. The overall response rate was 90%, including a VGPR rate of at least 42%, and a CR/near CR rate of 19%. VD had a favorable toxicity profile, with few cases of grade 3 to 4 neutropenia and peripheral neuropathy, and no DVT.
The phase 3 VISTA study compared MP versus bortezomib plus MP (VMP) [61,62]. TTP was 24 months for VMP and 17 months with MP. The 3-year OS was 69% with VMP and 54% with MP. Toxicities were higher with the 3-drug combination; the rates of grades 3 to 4 peripheral sensory neuropathy were 14% with VMP and 0% with MP. Gastrointestinal complications were more frequent with VMP (19%) than those with MP (5%). The rate of treatment-related deaths was unchanged in the two groups (2%). Based on these positive results, VMP is considered a standard strategy for treating elderly patients with multiple myeloma.
The VMP combination has also been compared with bortezomib-thalidomide-prednisone (VTP). A weekly schedule of bortezomib was used in both arms [63]. Although the two combinations induced similar OS, VTP was associated with an increased incidence of serious adverse events (15% vs. 31%; p = 0.01); grade 3 to 4 cardiac toxicity rate was (0% vs. 8.5%; p = 0.001), thromboembolism (1% vs. 2%; p = 0.5), and peripheral neuropathy (5% vs. 9%; p = 0.6), with VTP and VMP, respectively. In contrast, VMP was associated with higher incidences of neutropenia (39% vs. 22%; p = 0.008), thrombocytopenia (27% vs. 12%; p = 0.0001), and infections (7% vs. 1%; p = 0.01). The discontinuation rate was higher with VTP (12% vs. 17%; p = 0.03).
The addition of thalidomide to the new standard VMP followed by bortezomib-thalidomide maintenance (VMPT-VT) is a valid alternative [64]. The 3-year PFS was 56% with VMPT-VT and 41% with VMP (p = 0.008). Longer follow-up is needed to detect any OS advantage. The rate of CR was also higher with VMPT-VT (38% vs. 24%; p < 0.001). However, patients treated with VMPT-VT inevitably experienced more adverse events; grade 3 to 4 neutropenia (38% vs. 28%; p = 0.02), cardiac complications (10% vs. 5%; p = 0.04), and thromboembolic events (5% vs. 2%; p = 0.08). That study adopted a once-weekly bortezomib schedule instead of standard twice-weekly administration to reduce neuropathy associated with bortezomib administration. This strategy positively impacted toxicity, particularly peripheral neuropathy, and did not negatively affect efficacy [65]. Therefore, VMP-VT with once-weekly bortezomib seems a valid alternative for elderly patients, particularly those 65 to 75 years of age.
CONCLUSIONS
Patients with multiple myeloma have a wider variety of treatment options due to the availability of new drugs. Patients < 65 years are usually suitable for ASCT. Induction treatment with new drugs is now common, and the 3-drug combinations of VTD and PAD seem more effective than 2-drug combinations.
Patients > 65 years do not usually tolerate high-dose therapy and ASCT; thus, gentler approaches are more appropriate. MPT and VMP are now regarded as the new standards of care for elderly patients with multiple myeloma. Recently, MPR-R has proven to be a good alternative. The 4-drug combination VMPT-VT can also be considered a valid option. Future trials will investigate the role of novel second-generation agents, such as carfilzomib, pomalidomide, elotuzumab, and bendamustine.
Acknowledgments
The authors thank the editorial assistant Giorgio Schirripa.
Notes
Antonio Palumbo has received consultancy fees and honoraria from Amgen, Bristol-Myers Squibb, Celgene, Janssen, Millenium, Onyx.