Cross-neutralizing antibody responses among individuals with or without bivalent vaccine: a six-month prospective cohort study
Article information
Abstract
Background/Aims
Although the COVID-19 pandemic has officially ended, SARS-CoV-2 continues to circulate with periodic surges and may exhibit seasonal patterns. Understanding the importance of prompt immunization, particularly in individuals with prior infection, is crucial for developing future vaccination protocols. This study aimed to assess the durability and breadth of neutralizing antibody (nAb) responses against Omicron subvariants based on infection and bivalent vaccination status.
Methods
In this six-month prospective cohort study, we evaluated nAb responses to the original SARS-CoV-2 strain (D614G), as well as Omicron subvariants BA.4/5 and XBB.1.5 variants, in 79 healthcare workers stratified by prior Omicron infection and bivalent vaccination status. Blood samples were collected at baseline, 3 months, and 6 months, and nAb titers were measured using an optimized pseudovirus neutralization assay.
Results
At 3 months, individuals with prior Omicron infection followed by bivalent vaccination showed significantly higher nAb titers against BA.4/5 and XBB.1.5 compared to those with infection alone or vaccination alone. At 6 months, the highest titers persisted in the group with both prior infection and bivalent vaccination, while titers declined in previously infected but unvaccinated individuals. Notably, individuals with prior infection alone exhibited comparable nAb titers to infection-naïve vaccinated individuals, suggesting limited durability of infection-induced immunity without vaccine-induced boosting.
Conclusions
These findings underscore the importance of timely vaccination, even among previously infected individuals, to ensure sustained humoral immunity and broader cross-nAb responses against emerging SARS-CoV-2 variants.
INTRODUCTION
The emergence of multiple SARS-CoV-2 variants has substantially reshaped the global public health landscape [1,2] . Among them, the Omicron variant, with its high transmissibility and immune evasion capacity, has caused widespread infections worldwide [3,4] . Despite its high infection rate, vaccination remains a critical tool for reducing both infection risk and disease severity [5] . Although monovalent vaccines based on the ancestral strain provided strong protection against severe disease, their effectiveness against the Omicron variant was limited [6,7] . In response, bivalent mRNA vaccines targeting both the ancestral strain and Omicron subvariants were introduced, with rollout in South Korea beginning in November 2022 [8] . Although both epidemiological studies [9–12] and immunological data [13–15] support the enhanced effectiveness of bivalent vaccines, uptake of bivalent booster doses has declined substantially in several regions, including South Korea. This decline can be attributed to several factors. First, a substantial portion of the population has already experienced SARS-CoV-2 infection, leading to a widespread perception that natural immunity provides sufficient protection against reinfection [16] . Second, the occurrence of breakthrough infections among vaccinated individuals, as reported in observational studies, has further eroded public confidence in vaccine efficacy [17] . Moreover, despite the availability of bivalent vaccines, data on the durability and breadth of humoral immune responses, particularly among previously infected individuals with varying bivalent vaccination status, remain limited. As the global emergency of the COVID-19 pandemic has officially ended, SARS-CoV-2 is expected to exhibit seasonal resurgence patterns, similar to influenza [18,19] . In this context, understanding the longevity and breadth of immunity, particularly in individuals who forgo updated vaccination, is more critical than ever. Therefore, we aimed to investigate the longitudinal neutralizing antibody (nAb) responses elicited by bivalent vaccination against various Omicron sublineages across groups with different immunological histories of SARS-CoV-2 infection and subsequent vaccination.
METHODS
Study design and sample collection
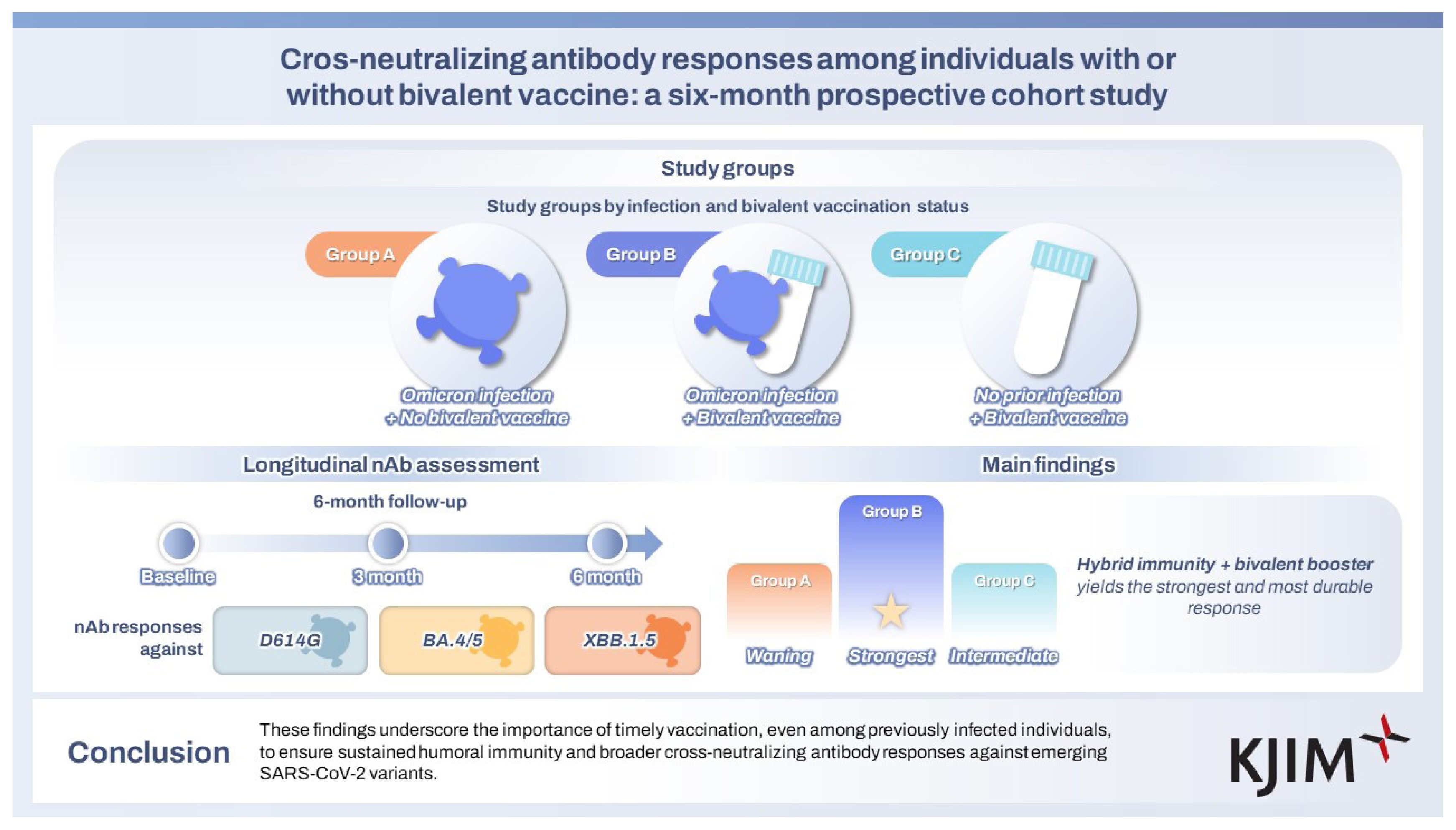
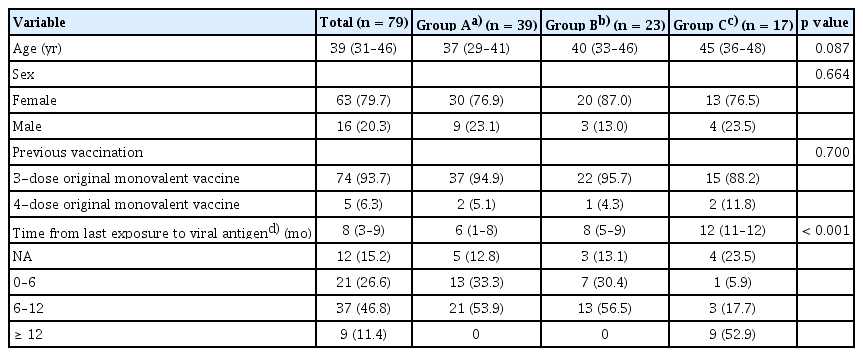
This six-month prospective study involved the collection of samples from healthcare workers (HCWs) at two centers in Seoul, South Korea: Asan Medical Center (December 2022 to July 2023) and Chung-Ang Gwangmyeong University Hospital (December 2022 to August 2023). A total of 79 HCWs who had received three or four doses of monovalent COVID-19 vaccines were enrolled. The participants were categorized into three groups according to their Omicron infection history and bivalent vaccination status: previously Omicron-infected without bivalent vaccination (Omicron-infected, bivalent unvaccinated), previously Omicron-infected with bivalent vaccination (Omicron-infected, bivalent vaccinated), and infection-naïve with bivalent vaccination (infection-naïve, bivalent vaccinated). The characteristics of each group are summarized in Table 1. The blood samples for the bivalent vaccinated groups were collected at baseline (before bivalent vaccination), and at 3 and 6 months after BA.4/5 bivalent mRNA vaccination. In the bivalent unvaccinated group, blood samples were collected at the same time points as the vaccinated groups to allow direct comparison across groups.
Production of pseudoviruses
For pseudovirus production, codon-optimized SARS-CoV-2 spike gene plasmids derived from the original D614G strain (plasmid #177960; Addgene, Watertown, MA, USA), the BA.4/5 variant (plasmid #186031; Addgene), and the XBB.1.5 variant (plasmid #196585; Addgene) were utilized. These spike plasmids were co-transfected into Lenti-X 293T cells (Cat. 632180; Takara Bio, Kusatsu, Shiga, Japan) along with the lentiviral packaging plasmid psPAX2 (plasmid #12260; Addgene) and the transfer plasmid pHAGE-CMV-Luc2-IRES-ZsGreen-W (plasmid #164432; Addgene) to produce pseudoviruses.
Lenti-X 293T cells were seeded in T75 flasks and cultured in high-glucose Dulbecco’s modified Eagle medium (DMEM) supplemented with GlutaMAX™, 10% fetal bovine serum (FBS), and 1% penicillin–streptomycin (culture medium). Cells were incubated overnight at 37°C and in a humidified 5% CO2 atmosphere. Transfection was performed using a modified protocol based on the manufacturer’s instructions for Lipofectamine™ 3000 (Cat. L3000075; Invitrogen, Carlsbad, CA, USA), with optimizations to enhance pseudovirus production. For transfection, two mixtures were prepared: tube A contained Lipofectamine™ 3000 diluted in Opti-MEM™ I Reduced Serum medium, while tube B consisted of P3000™ Enhancer and three plasmids—SARS-CoV-2 Spike, psPAX2 (a packaging plasmid), and pHAGE-CMV-LUC2-IRES-ZsGreen-W (a transfer plasmid)—mixed in a ratio of 3:12:9 and diluted in Opti-MEM™. The two mixtures were vortexed separately for 15 seconds, gently shaken for 3 minutes at room temperature, and then mixed and incubated together for 15 minutes. Before transfection, the culture medium was discarded, and cells were washed with plain DMEM. A transfection medium consisting of Opti-MEM™ supplemented with 5% FBS and 1 mL of sodium pyruvate was added (10 mL per flask). The transfection mixture was then added to each flask containing 8 × 106 cells. After 6 hours of incubation at 37°C, the medium was replaced with fresh transfection medium. At 52 hours after transfection, the supernatants containing pseudoviruses were harvested, centrifuged at 1,500 rpm for 10 minutes at 4°C, and filtered using a 0.45-μM filter.
Pseudovirus neutralization assay
Human ACE2-293T cells (Cat. 631289; Takara Bio) were seeded in 96-well plates at a density of 2 × 104 cells/well in cell culture media and incubated overnight at 37°C in a humidified atmosphere with 5% CO2. SARS-CoV-2 pseudoviruses, each with a titer exceeding 1,000 TCID50/mL, were prepared for the respective variants. Plasma samples were serially diluted in seven 3-fold steps, yielding final dilutions ranging from 1:10 to 1:7,290. The diluted plasma samples were then mixed with the pseudoviruses and incubated at 37°C for 2 hours. After incubation, the culture medium in the ACE2-293T cell plates was removed, and 100 μL of the plasma–virus mixture was added to each well. Positive control wells (pseudovirus only) and negative control wells (media only) were included. The plates were incubated for an additional 2 hours at 37°C, followed by the addition of 50 μL of fresh DMEM to each well. The cells were further incubated for 48 hours at 37°C in a 5% CO2 incubator. After 48 hours, relative luminescence units were measured using a fluorescence microplate reader (Victor X3; PerkinElmer, Shelton, CT, USA) with a luciferase substrate (Bright-Glo™ Luciferase Assay System; Promega, Madison, WI, USA). The 50% neutralization antibody titers (NT50) were calculated using the Reed–Muench formula. This formula was applied directly to the luminescence values at each plasma dilution to determine the dilution corresponding to 50% neutralization. This study employed optimized pseudovirus-based neutralization assays, which correlate well with live-virus assays and are suitable for biosafety level 2 settings [20] .
Statistical analysis
The normality of the NT50 data was evaluated using the D’Agostino–Pearson test and the Shapiro–Wilk test, as appropriate. None of the datasets satisfied both assumptions of normality; therefore, all continuous variables were compared using the Mann–Whitney U test. Categorical variables were compared using Fisher’s exact test. Geometric mean titers (GMTs) and 95% confidence intervals (CIs) were obtained using descriptive statistics. All statistical tests were two-tailed, and p values < 0.05 were considered statistically significant. Statistical analyses were conducted using GraphPad Prism software (version 10.0; GraphPad Software, San Diego, CA, USA).
Ethics statement
All participants provided written informed consent. The study was approved by the Institutional Review Board of Asan Medical Center (IRB No. 2022-1269).
RESULTS
Baseline characteristics of study participants
A total of 79 HCWs were enrolled in this study. Of these, 39 (49.4%) had experienced Omicron infection but had not received the bivalent vaccine, 23 (29.1%) had experienced Omicron infection before receiving the bivalent vaccination, and 17 (21.5%) were infection-naïve but had received the bivalent vaccine. The median age across all participants was 39 (interquartile range [IQR], 31–46) years. The median interval from the last Omicron infection or monovalent vaccination to baseline sample collection was approximately 6–8 months and 12 months, respectively (Table 1).
nAb responses
The NT50 against the SARS-CoV-2 original D614G strain, Omicron BA.4/5, and Omicron XBB.1.5 variants were assessed at three standardized time points: baseline, 3 months, and 6 months. Time points for vaccinated individuals were based on the date of bivalent vaccination, whereas those for unvaccinated individuals followed the same schedule to ensure comparability. The results of the three groups—previously Omicron-infected, bivalent unvaccinated (A); previously Omicron-infected, bivalent vaccinated (B); and infection-naïve, bivalent vaccinated (C)—are summarized in Figure 1.
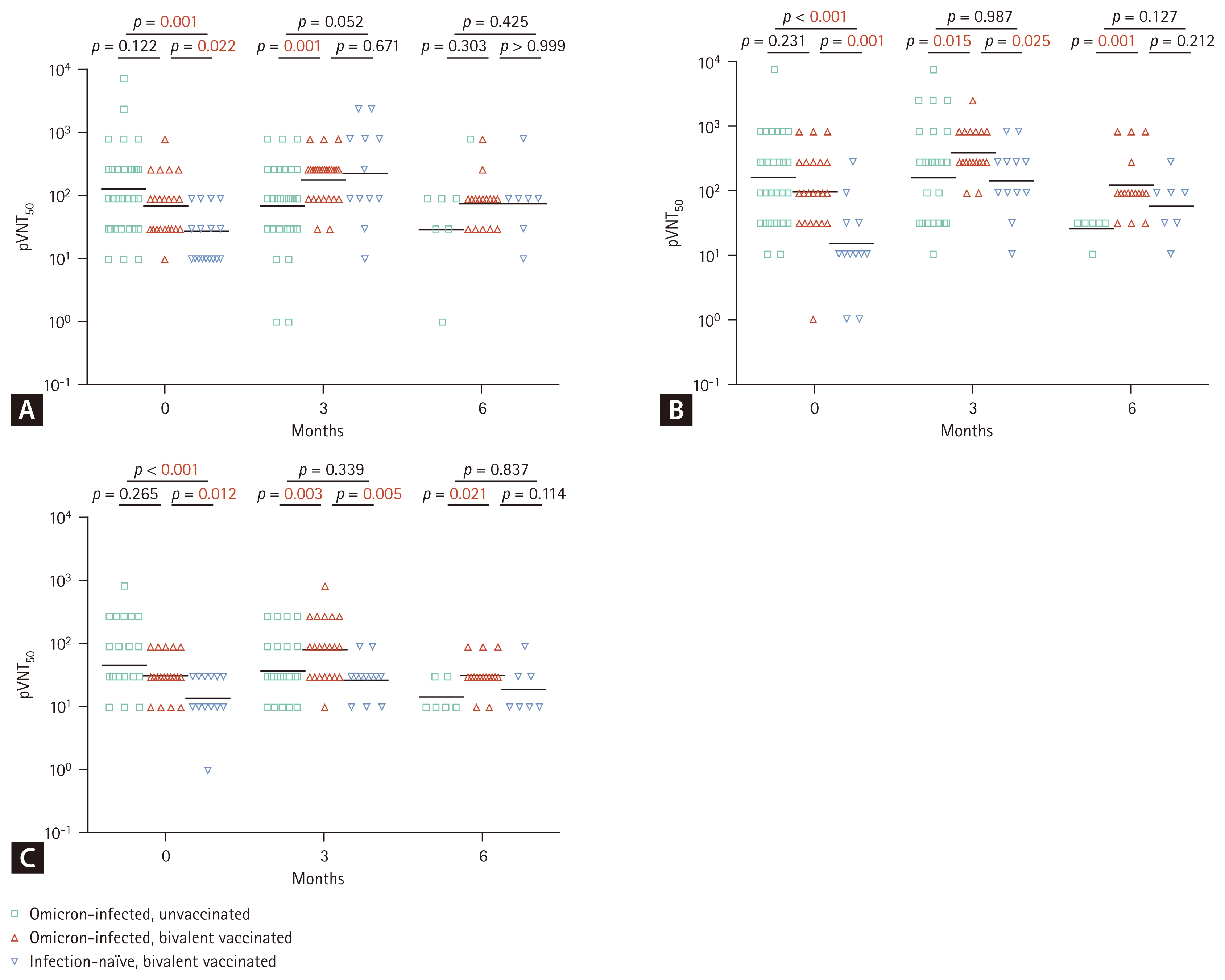
Comparison of pseudovirus neutralizing antibody responses against SARS-CoV-2 original strain D614G (A), Omicron BA.4/5 (B), and XBB.1.5 (C) variants. Pseudovirus 50% neutralization titers (pVNT50) were assessed using a pseudovirus neutralization assay optimized for this study. Neutralizing antibody responses were measured at three standardized time points—baseline, 3 months, and 6 months—across three groups: (a) previously Omicron-infected, bivalent unvaccinated (green squares); (b) previously Omicron-infected, bivalent vaccinated (red triangles); and (c) infection-naïve, bivalent vaccinated (blue inverted triangles). For vaccinated individuals, time points indicate intervals following bivalent vaccination, whereas for unvaccinated individuals, samples were collected following the same schedule to allow comparison with the vaccinated groups. Horizontal lines represent geometric mean titers. Statistical analysis was performed using the Mann–Whitney U test, with statistically significant p values highlighted in red.
At baseline, individuals previously infected with Omicron (Groups A and B) exhibited significantly higher nAb responses across all tested variants compared to Group C. Statistically significant differences were observed between Group A and C for the original D614G strain (p = 0.001), and BA.4/5 (p < 0.001) and XBB.1.5 (p < 0.001) variants. Similarly, Group B showed significantly higher nAb titers than Group C for original D614G (p = 0.022), BA.4/5 (p = 0.001), and XBB.1.5 (p = 0.012). However, no significant differences were observed between Groups A and B for any of the variants (original D614G: p = 0.122; BA.4/5: p = 0.231; XBB.1.5: p = 0.265).
At the three-month time point, nAb levels against the original D614G strain did not differ significantly between Groups B and C (p = 0.671). However, for the Omicron variants BA.4/5 and XBB.1.5, Group C exhibited significantly lower titers compared to Group B (p = 0.025 and p = 0.005, respectively), while showing similar titers to those of the previously infected, bivalent unvaccinated group (Group A) for both BA.4/5 (p = 0.987) and XBB.1.5 (p = 0.339).
At the six-month time point, nAb titers against the original D614G strain did not differ significantly among the three groups (Group A vs. Group B: p = 0.303; Group A vs. Group C: p = 0.425; Group B vs. Group C: p > 0.99). In contrast, among individuals previously infected with Omicron, nAb titers against Omicron BA.4/5 and XBB.1.5 were significantly lower in the unvaccinated group (Group A) compared to the bivalent vaccinated group (Group B) (p = 0.001 and p = 0.021, respectively). Detailed numerical results, including GMTs, 95% CIs, and medians with IQRs, are provided in Supplementary Table 1.
DISCUSSION
In this study, we evaluated nAb responses against the SARS-CoV-2 original D614G strain, as well as Omicron BA.4/5 and XBB.1.5 variants, over 6 months. Our findings demonstrate that individuals with prior Omicron infection who received the BA.4/5 bivalent vaccine (Group B) exhibited significantly higher nAb titers against both BA.4/5 and XBB.1.5 compared to bivalent-unvaccinated individuals (Group A) and infection-naïve, bivalent vaccinated individuals (Group C), with differences sustained up to 6 months after vaccination.
There are two important findings in our study. First, this study provides a longitudinal evaluation of cross-nAb responses up to 6 months following BA.4/5 bivalent mRNA vaccination across three immunologically distinct groups. Early studies conducted shortly after the introduction of the BA.4/5 bivalent vaccine reported that it elicited cross-neutralizing activity against the XBB variant, with individuals previously infected with SARS-CoV-2 showing higher nAb titers compared to infection-naïve individuals; however, these data were based on a short-term follow-up of approximately one month after vaccination [21,22] . Consistent with these observations, Urschel et al. [23] observed robust cross-nAb responses against multiple Omicron subvariants—BA.1, BA.2, and BA.4/BA.5—following bivalent BA.4/5-adapted mRNA vaccination, with stronger responses in individuals with prior infection. However, their assessment was limited to just two weeks after vaccination. Similarly, Nham et al. [24] reported that individuals with prior SARS-CoV-2 infection showed stronger nAb responses against Omicron subvariants—including BA.5, BN.1, and XBB.1.5—following BA.4/5-containing bivalent mRNA vaccination, compared to infection-naïve individuals. These findings are consistent with our results. However, the duration of follow-up in that study was limited to 3 months. In contrast, the extended six-month follow-up in our study provides additional insights into the durability of vaccine-induced antibody responses, particularly in the context of emerging Omicron subvariants. Second, our study comprised individuals with prior Omicron infection who had not received a bivalent vaccine (Group A), and our findings indicate that the nAb titers in Group A were comparably low to those observed in infection-naïve, bivalent vaccinated individuals (Group C). Notably, the inclusion of Group A provides direct evidence that prior infection alone may not confer sufficient or durable protection without additional vaccination. While Nham et al. [24] compared bivalent-vaccinated individuals solely based on prior infection status, our study offers a more comprehensive evaluation by including a non-vaccinated control group. Notably, we evaluated the durability of antibody responses after previous infection without updated booster vaccination and demonstrated that previous infection alone may not elicit adequate or lasting nAb responses against emerging variants. Moreover, a prospective cohort study by Shigayeva et al. [25] provided epidemiological evidence that individuals with a prior SARS-CoV-2 infection were associated with an increased risk of Omicron reinfection if they had not received an updated vaccine. Specifically, those with an early SARS-CoV-2 infection alone had the highest risk, while individuals with both early and BA.1/2 infections, or those who had received four or more vaccine doses, exhibited significantly lower reinfection rates. However, their analysis focused solely on clinical outcomes and did not include immunological assessments such as nAb titers. In contrast, our study directly assessed humoral immunity by quantifying nAb responses in individuals with prior Omicron infection who had not received a bivalent booster, revealing insufficient immune durability in the absence of subsequent vaccination. These findings underscore the importance of timely booster vaccination to maintain protective immunity, even in individuals with prior infection.
This study has several limitations. First, the sex distribution within the study population was skewed, with a higher proportion of female participants, which may limit the generalizability of the findings. Second, other critical components of the immune response, such as T cell-mediated immunity, were not assessed, even though they may play a complementary and potentially more durable role, particularly in protection against severe disease. Third, the relatively small sample size may reduce the statistical power and limit the generalizability of our results. Given that other studies have demonstrated significant differences between hybrid and naïve groups at approximately 6 months after vaccination [26,27], some of the statistically non-significant comparisons, particularly at the 6-month time point, may have been underpowered to detect existing differences, which raises the possibility of a type II error, and therefore should not be interpreted as evidence of equivalence. Fourth, this study focused on immune responses during the BA.4/5 and XBB.1.5-predominant period (December 2022 to August 2023). Meanwhile, more recent Omicron sublineages, such as JN.1 and its descendants, have become globally dominant since early 2024 [28,29] . Updated vaccines targeting JN.1 are now being deployed and have been reported to elicit enhanced immune responses against JN.1 and its related subvariants [30,31], but understanding the cross-nAb responses against a broad range of other Omicron subvariants remains essential for predicting the breadth and durability of protection against future emerging variants. Finally, the interval since the last SARS-CoV-2 exposure differed among groups, and Group A may have experienced more pronounced waning of immunity owing to a longer time since infection. However, this variability may reflect real-world circumstances in which some individuals forgo additional booster doses, highlighting the importance of timely booster uptake as recommended by public health authorities.
In conclusion, individuals with previous Omicron infection followed by BA.4/5 bivalent vaccination exhibited the most robust and durable nAb responses against both the BA.4/5 variant and the subsequently emerging XBB.1.5 subvariant over 6 months. These findings underscore the importance of updated vaccination, even among previously infected but unvaccinated individuals, who showed waning antibody responses comparable to those observed in infection-naïve vaccine recipients. Timely booster vaccination is, therefore, essential to maintain broad and sustained humoral immunity against newly emerging SARS-CoV-2 variants.
KEY MESSAGE
1. Prior Omicron infection followed by bivalent vaccination elicited the most robust and durable cross-nAb responses over 6 months.
2. Individuals with prior infection alone showed limited neutralizing activity, which was comparable to that of infection-naïve individuals who received bivalent vaccination.
3. These findings highlight the importance of timely booster vaccination to maintain broad and sustained protection, even in previously infected individuals.
Notes
CRedit authorship contributions
Hye Hee Cha: methodology, investigation, data curation, formal analysis, validation, software, writing - original draft, writing - review & editing; Ji Yeun Kim: methodology, investigation, validation, writing - review & editing; Seung Beom Kim: investigation, writing - review & editing; Junho Cha: investigation, writing - review & editing; Ji-Soo Kwon: formal analysis, writing - review & editing; Woori Kim: investigation, writing - review & editing, project administration; Ju Yeon Son: investigation, writing - review & editing, project administration; Choi Young Jang: investigation, writing - review & editing, project administration; Min-Chul Kim: investigation, writing - review & editing; So Yun Lim: conceptualization, data curation, formal analysis, validation, writing - original draft, writing - review & editing; Sung-Han Kim: conceptualization, validation, writing - review & editing
Conflicts of interest
The authors disclose no conflicts.
Funding
This research was supported by the National Institute of Infectious Diseases, National Institute of Health (NIH) research project (project No. HD22C2045 and 2026-ER1902-00).