Clinical outcomes of heart failure with improved ejection fraction in patients treated with angiotensin receptor–neprilysin inhibitor
Article information
Abstract
Background/Aims
Improvement of left ventricular ejection fraction (LVEF) in heart failure with reduced ejection fraction (HFrEF) has been associated with favorable outcomes; however, corresponding data in patients treated with angiotensin receptor–neprilysin inhibitors (ARNIs) remain limited. This study aimed to examine the characteristics, predictors, and outcomes of HF with improved ejection fraction (HFimpEF) compared with persistent HFrEF in the RECORD-SV registry.
Methods
Patients with LVEF ≤ 40% in the RECORD-SV registry were classified based on echocardiographic assessments at baseline and 1-year follow-up. Patients with HFimpEF were defined as those with follow-up LVEF of > 40% with an absolute improvement of ≥ 10%, whereas the persistent HFrEF group included those who did not meet these criteria. Baseline characteristics, predictive factors, and clinical outcomes of the patients were analyzed. The primary composite outcome was all-cause mortality and hospitalization for HF management, with secondary outcomes including each component of the primary outcome and frequency of urgent emergency room visits.
Results
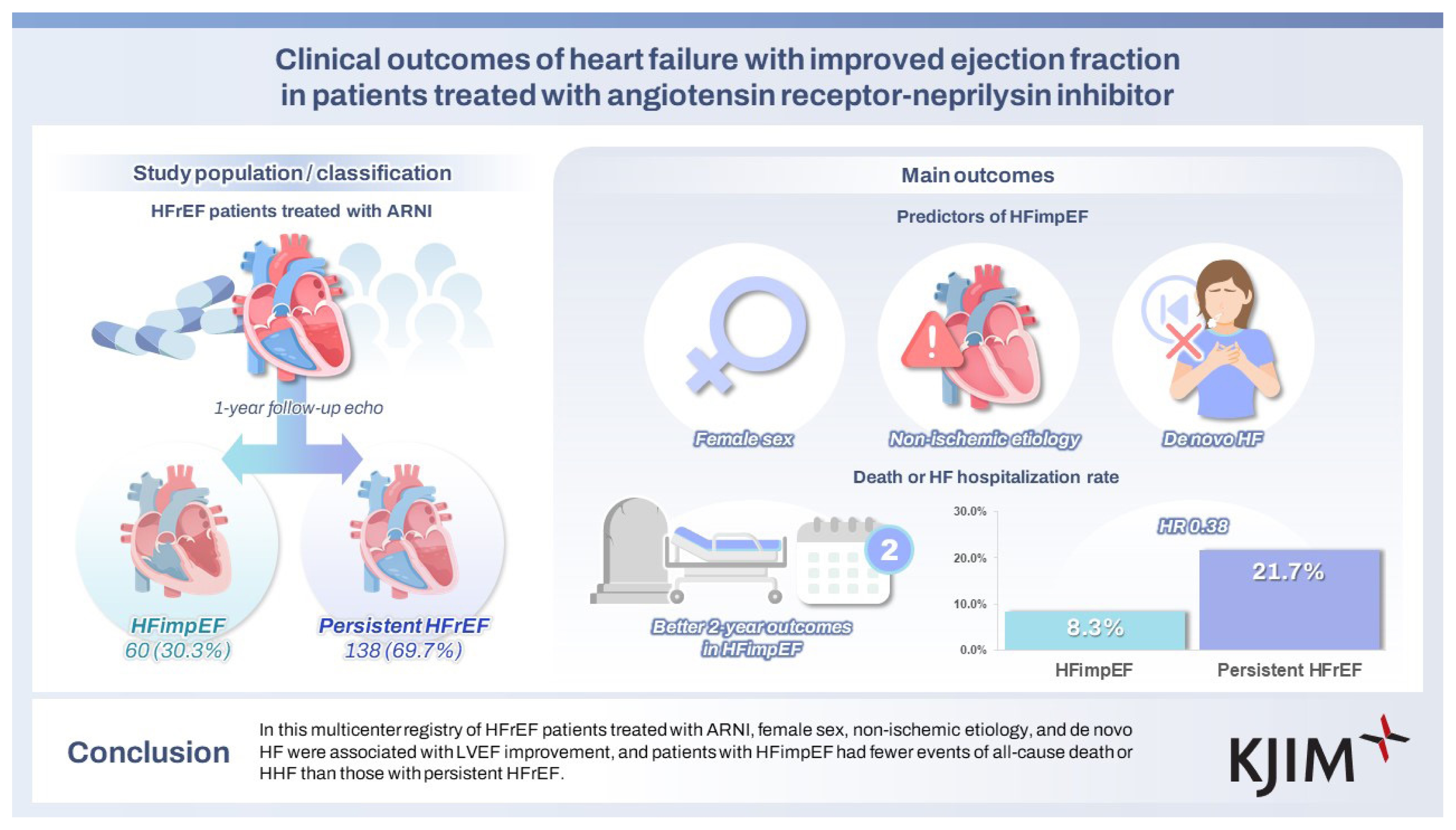
Among 198 patients with HFrEF, 60 patients (30.3%) achieved HFimpEF after one year of ARNI-based therapy. Independent predictors of HFimpEF included female sex, non-ischemic etiology, and de novo HF. HFimpEF patients exhibited significantly lower risks in the primary composite outcome (HR 0.382, 95% CI 0.148–0.985) compared to those with persistent HFrEF, with a similar trend observed for secondary outcomes.
Conclusions
Patients achieving HFimpEF with ARNIs exhibited distinct clinical features and significantly lower risk of cardiovascular events compared to those with persistent HFrEF, highlighting the prognostic benefit of EF recovery with ARNI therapy.
INTRODUCTION
The increasing elderly population and rising prevalence of cardiovascular risk factors, including hypertension, diabetes, and dyslipidemia, had increased the global incidence of heart failure (HF) to 1–2% among the general adult population, and it is anticipated to continue rising [1–3] . In Korea, the prevalence of HF nearly tripled from 0.81% in 2008 to 2.38% in 2020, with an especially high burden observed in patients aged ≥ 80 years, who exhibit a > 15-fold higher HF prevalence than the younger populations [4] .
The primary goals of HF management are to alleviate symptoms, reduce rehospitalization, and improve survival. The achievement of these objectives is closely linked to left ventricular (LV) reverse remodeling, which has been recognized as a key mechanism underlying functional improvement and favorable outcomes. In this context, left ventricular ejection fraction (LVEF) serves as a central marker of cardiac structure and function. Therapeutic strategies that promote LVEF recovery have contributed to the recognition of HF with improved ejection fraction (HFimpEF), a distinct phenotype associated with better long-term prognosis [5] .
Angiotensin receptor–neprilysin inhibitors (ARNIs) have recently become the cornerstone of guideline-directed medical therapy for HF, exerting unique effects on LV reverse remodeling via dual neurohormonal modulation [6] . Previous studies in the era of angiotensin converting enzyme inhibitors (ACEIs), angiotensin receptor blockers (ARBs), beta-blockers, and mineralocorticoid receptor antagonists have demonstrated improved outcomes among patients undergoing EF recovery. However, evidence specifically addressing HFimpEF in patients treated with ARNIs remains limited [7,8] . Moreover, data from real-world Asian populations are scarce.
The present study aimed to investigate the clinical features, predictive factors, and outcomes of HFimpEF compared to persistent HF with reduced ejection fraction (HFrEF) among patients treated with ARNIs in the multicenter RECORD-SV registry.
METHODS
Study population
This retrospective cohort study analyzed the RECORD-SV registry dataset (https://cris.nih.go.kr; Identifier: KCT0005747). The dataset includes 251 HF patients with LVEF ≤ 40% who were treated with ARNIs from July 2017 to June 2020. The study protocol was approved by the Institutional Review Board of each participating center, including Soonchunhyang University Bucheon Hospital (approval number: 2020-11-010). In this study, we included patients aged ≥ 18 years who were hospitalized for acute HF with LVEF ≤ 40% and who newly initiated ARNI therapy during the index hospitalization. From the initially enrolled 251 patients, those without follow-up echocardiography data, those who discontinued ARNI therapy before undergoing follow-up echocardiography, and those with less than three months between ARNI initiation and follow-up echocardiography were excluded. These exclusion criteria were selected to eliminate patients undergoing early assessments wherein clinically meaningful reverse remodeling effects of ARNI therapy would not be expected to emerge within such a brief interval. Prior studies evaluating ARNI-associated LVEF improvement have typically used follow-up intervals of ≥ 6 months to ensure sufficient duration for structural changes to emerge [9,10] .
Consequently, a total of 198 patients were included in the final analysis cohort (Fig. 1). Based on their baseline and 1-year follow-up echocardiographic data, the patients were categorized into two groups: HFimpEF, comprising those who showed a follow-up LVEF of > 40% and an increase of ≥ 10% from baseline, and persistent HFrEF, comprising those who did not meet these criteria [5] .
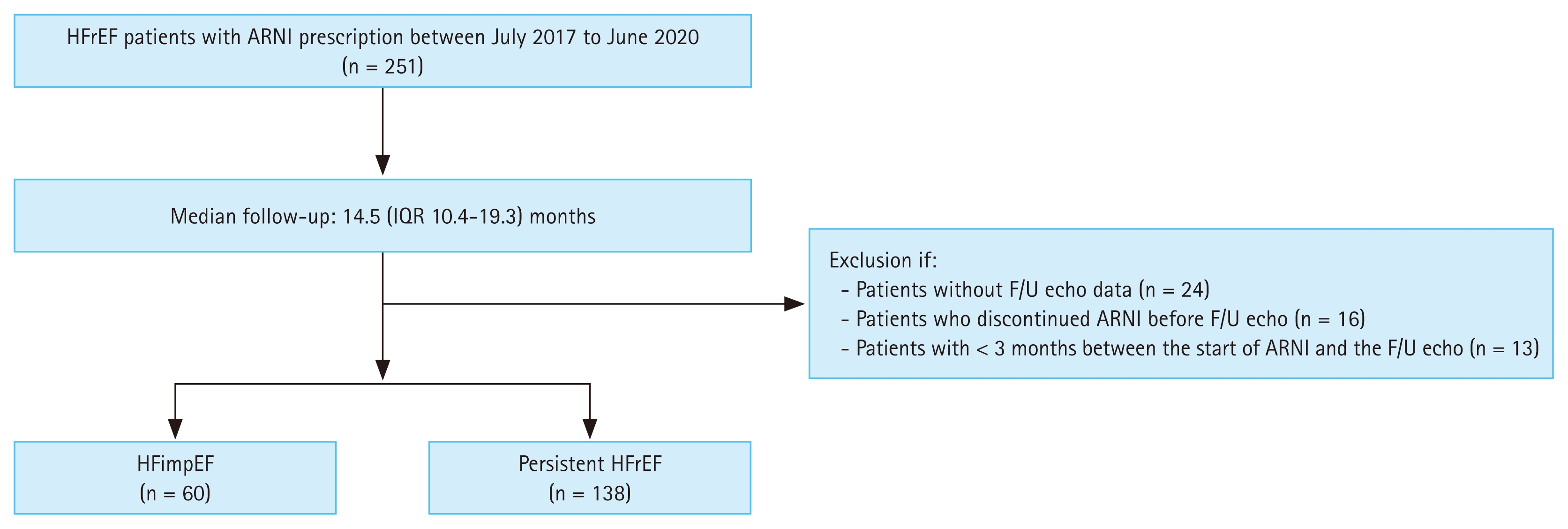
Study flow diagram. HFrEF patients who were prescribed ARNIs between July 2017 and June 2020 were identified. In total, 198 patients were included in our cohort. HFrEF, heart failure with reduced ejection fraction; ARNI, angiotensin receptor–neprilysin inhibitor; F/U, follow-up; HFimpEF, heart failure with improved ejection fraction.
Data collection
From the previously described RECORD-SV registry, demographic data including age, sex, clinical symptoms, medical history, comorbidities, current medications, laboratory test results and echocardiography reports were collected. Owing to the etiology of HF, its causes were classified into ischemic and non-ischemic causes. Ischemic etiology was defined as HF occurring in the context of documented evidence of myocardial ischemia, including a history of myocardial infarction (MI) or any form of coronary revascularization [11] . Non-ischemic etiologies comprised dilated cardiomyopathy, valvular heart disease, and tachycardia-induced HF. The criteria for distinguishing between prior and de novo HF were based on the duration from HF diagnosis to ARNI treatment initiation. De novo HF was defined as newly diagnosed HF within six months prior to ARNI initiation. The six-month threshold was selected to differentiate recent-onset, treatment-naïve HF from established chronic disease, while maintaining consistency with the definitions used in observational HF research [12] .
To minimize potential time-dependent bias, HFimpEF was defined solely based on the planned echocardiographic assessment performed approximately 12 months (within an allowable window of 6–18 mo) after ARNI initiation. Patients who showed no improvement on any earlier interim echocardiogram but fulfilled these criteria at the 12-month assessment were included as HFimpEF patients. LVEF was measured by the biplane Simpson technique, and other variable parameters were measured according to the standard protocol. The collected echocardiographic parameters were as follows: LV volumes and diameters, LV mass index, left atrial volume index, and early diastolic mitral inflow velocity/early diastolic mitral annular tissue velocity.
Outcomes
The primary outcome of this study was the composite outcome of all-cause deaths and hospitalizations for HF (HHF) over two years. HHF was defined as hospital admissions for symptom control and HF management. As for the secondary outcome, the following parameters were investigated over the same two-year period: all-cause deaths, HHF, urgent emergency room (ER) visits for HF management, and predictive factors for HFimpEF. Urgent ER visits were identified as instances wherein the patients required HF management via intravenous medical therapy without hospital admission. All clinical events were identified by analyzing the medical records.
Statistical analysis
For each treatment group, baseline characteristics were presented as numbers of patients and percentages for categorical variables, and as means with standard deviations or medians with interquartile ranges for continuous variables. In two-group comparisons, continuous variables were analyzed using either the independent t-test or Wilcoxon’s rank sum test, depending on the normality of the distribution. Categorical data were compared using the chi-square test or Fisher’s exact test. The software used for statistical analyses included SPSS version 25 (IBM Corp., Armonk, NY, USA) and R version 3.6.0.
Survival in the two groups during the follow-up period was analyzed using Kaplan–Meier curves with the log-rank test. The association between achieving HFimpEF and clinical outcomes was evaluated using the Cox proportional hazards model. For the secondary outcomes and subgroup analyses of the primary outcome, results were analyzed without adjusting for multiplicity. Subgroup analyses were conducted based on age, sex, comorbidities, renal function, etiology of HF, dose, and HF medication use.
To identify predictive factors for HFimpEF, a univariable logistic regression analysis was conducted, and variables with p < 0.2 were included in the multivariable analysis. Collinearity was considered as a basis for exclusion, and clinically established predictors were prioritized for inclusion.
RESULTS
Baseline characteristics
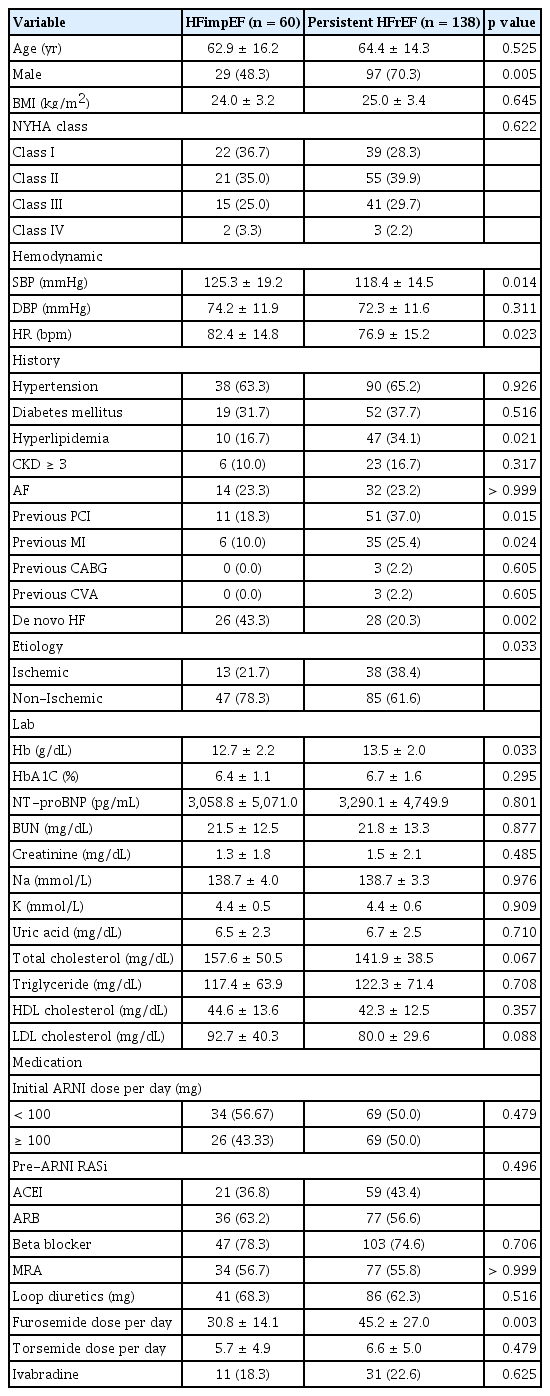
Among the 198 patients diagnosed with HFrEF, 60 patients (30.3%) achieved HFimpEF after one year of guideline-directed medical therapy, including ARNI, confirmed by follow-up echocardiography. The remaining 138 patients (69.7%), who did not meet the diagnostic criteria for HFimpEF, were classified into the persistent HFrEF group. The baseline characteristics of the two groups are presented in Table 1. The mean ages of the HFimpEF and persistent HFrEF groups were 62.9 ± 16.2 and 64.4 ± 14.3 years, respectively (p = 0.525). Compared to the persistent HFrEF group, the HFimpEF group had a significantly higher proportion of females (51.7% vs. 29.7%, respectively; p = 0.005), a significantly higher mean baseline systolic blood pressure (SBP; 125.3 ± 19.2 vs. 118.4 ± 14.5 mmHg, respectively; p = 0.014), a significantly higher heart rate (HR; 82.4 ± 14.8 vs. 76.9 ± 15.2 bpm, respectively; p = 0.023), and a significantly lower prevalence of hyperlipidemia (16.7% vs. 34.1%, respectively; p = 0.021). In addition, the HFimpEF group exhibited a significantly lower history of percutaneous coronary intervention (18.3% vs. 37.0%, respectively; p = 0.015) and MI (10.0% vs. 25.4%, respectively; p = 0.024), a significantly lower percentage of ischemic etiology (21.7% vs. 38.4%, respectively; p = 0.033), and a significantly higher proportion of de novo HF (43.3% vs. 20.3%, respectively; p = 0.002) compared to the other group. The HFimpEF group also required a lower dose of short-acting loop diuretics than the other group (30.8 ± 14.1 vs. 45.2 ± 27.0 mg, respectively).
Echocardiographic parameters
The initial EFs of the HFimpEF and persistent HFrEF groups were comparable at 28.5 ± 6.3% and 26.6 ± 7.3% (p = 0.081), respectively (Supplementary Table 1). However, volume parameters, including left ventricular end-systolic volume index (27.9 ± 9.9 vs. 60.3 ± 26.6 mL/m2, respectively; p < 0.001) and left ventricular end-diastolic volume index (56.7 ± 14.3 vs. 84.9 ± 30.9 mL/m2, respectively; p < 0.001), were significantly smaller in the HFimpEF group than in the persistent HFrEF group. At one-year follow-up, the mean EFs were 52.6 ± 7.5% and 29.7 ± 7.5% in the HFimpEF and persistent HFrEF groups (p < 0.001), with an increase of 24.0 ± 10.0% and 3.0 ± 6.0%, respectively. During the same period, both groups showed a decrease in volume parameters, with a more prominent reduction observed in the HFimpEF group.
Independent prognostic factors for HFimpEF
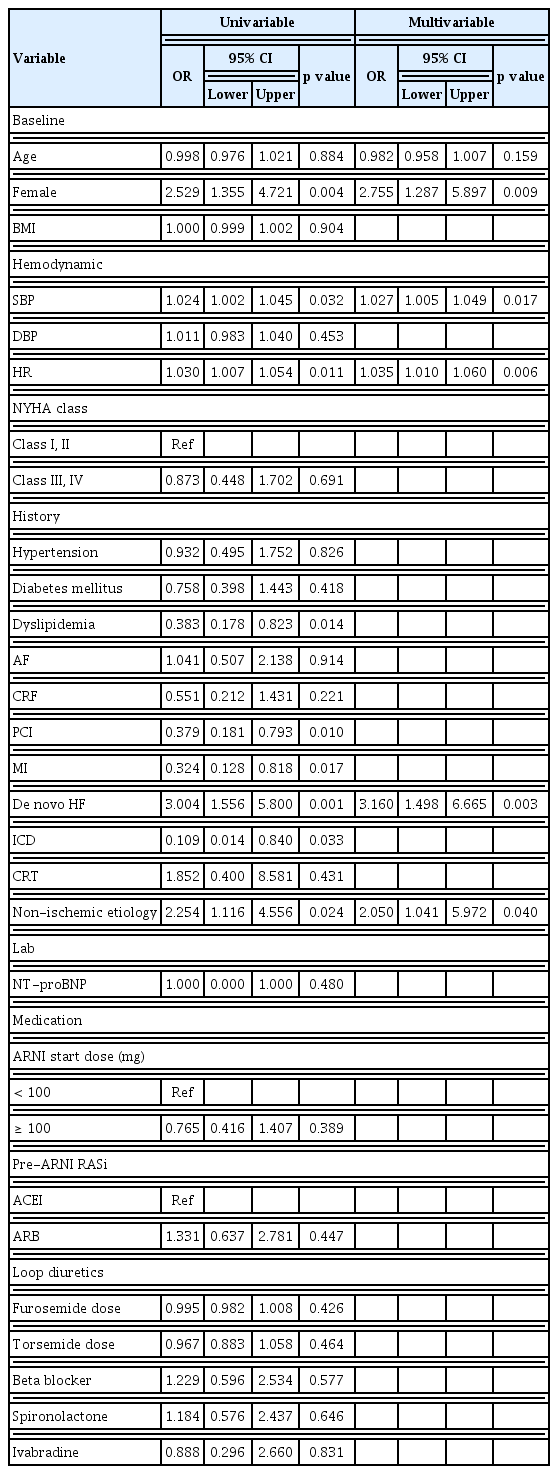
The multivariable logistic regression analysis conducted to identify the predictors of HFimpEF among HFrEF patients treated with ARNIs (Table 2) identified characteristics such as female sex, non-ischemic etiology, and de novo HF as independent prognostic factors. Higher initial SBP (per 1 mmHg) and HR (per 1 bpm) were associated with 2.8% and 3.0% higher likelihood of achieving HFimpEF (OR 1.027, 95% CI 1.005–1.049, p = 0.017 and OR 1.035, 95% CI 1.010–1.060, p = 0.006), respectively. There was no significant difference in the HFimpEF achievement among patients stratified by ARNI dosage (OR 0.765, 95% CI 0.416–1.407, p = 0.389).
Clinical outcomes
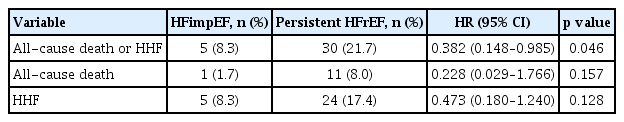
The median follow-up interval from ARNI initiation was 440.5 (IQR, 315.0–587.3) days. At two years, the primary outcome was observed more frequently in the persistent HFrEF group than in the HFimpEF group (8.3% vs. 21.7%, respectively; HR 0.382, 95% CI 0.148–0.985, p = 0.046; Table 3; Fig. 2). All secondary outcomes, including all-cause death (1.7% vs. 8.0%, respectively; HR 0.228, 95% CI 0.029–1.766, p = 0.157), HHF (8.3% vs. 17.4%, respectively; HR 0.473, 95% CI 0.180–1.240, p = 0.128), or urgent ER visit (10.0% vs. 16.7%, respectively; HR 0.686, 95% CI 0.277–1.700, p = 0.415), showed numerically lower event rates in the HFimpEF group than in the persistent HFrEF group (Supplementary Fig. 1).
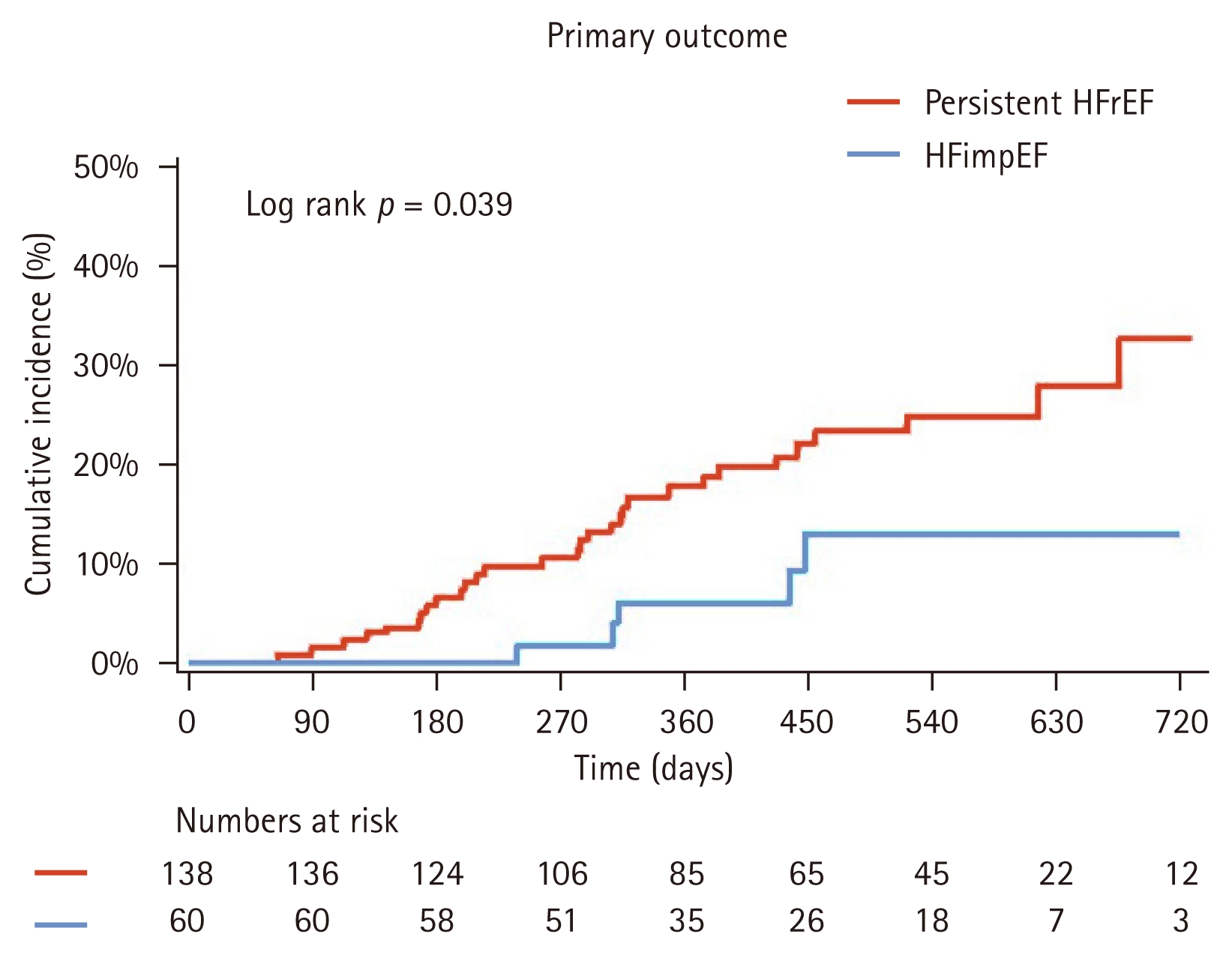
Cumulative incidence of primary outcome stratified by EF improvement. Kaplan–Meier curve comparing the primary composite outcome between the HFimpEF and persistent HFrEF groups over two years. HFrEF, heart failure with reduced ejection fraction; HFimpEF, heart failure with improved ejection fraction.
A prespecified subgroup analysis using the Cox proportional hazards model demonstrated that the favorable effect of HFimpEF on the primary outcome was consistent across a wide range of clinically relevant subgroups, including age, sex, ischemic etiology, baseline LVEF, and comorbid conditions (Fig. 3). In each subgroup, patients with HFimpEF exhibited a markedly lower incidence of the primary outcome compared to those who remained in persistent HFrEF. No significant interactions were observed between subgroup variables and group assignment.
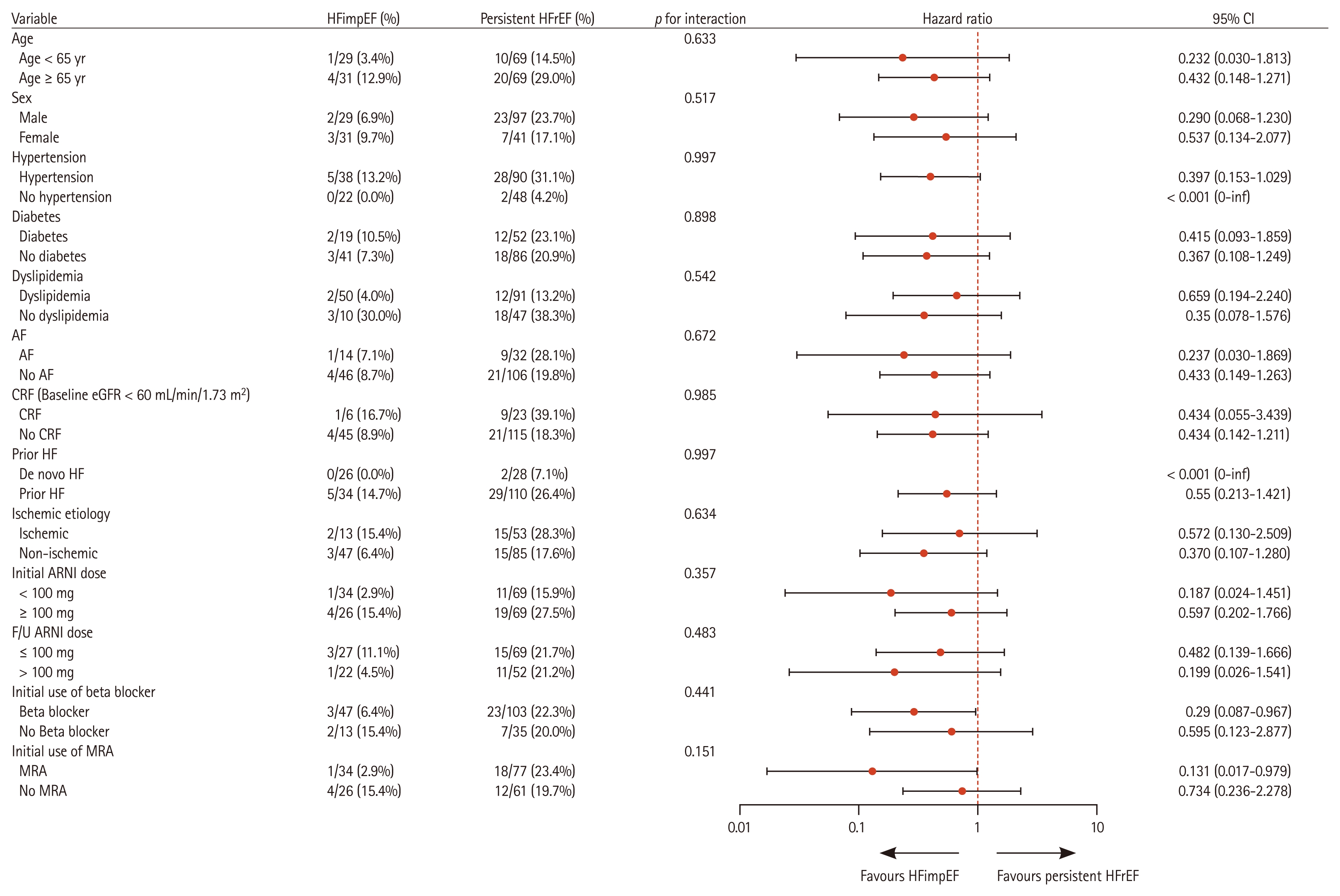
Subgroup analysis of the primary composite outcome. Forest plot showing the HR and 95% CI for the primary composite outcome across various subgroups. The vertical solid line at HR = 1 indicates the null value. Subgroup-specific HRs are plotted with respective CIs. p values for interactions indicate the consistency of effects among the subgroups. HFimpEF, heart failure with improved ejection fraction; HFrEF, heart failure with reduced ejection fraction; HR, hazard ratio; CI, confidence interval; AF, atrial fibrillation; CRF, chronic renal failure; eGFR, estimated glomerular filtration rate; HF, heart failure; ARNI, angiotensin receptor–neprilysin inhibitor; F/U, follow-up; MRA, mineralocorticoid receptor antagonist.
DISCUSSION
This study analyzed data from 198 HFrEF patients who were prescribed ARNIs and followed up for two years. The principal findings of this study were as follows: (1) among patients diagnosed with HFrEF, 30.3% achieved HFimpEF after one year of medical therapy involving ARNI; (2) Patients who achieved HFimpEF exhibited a lower incidence of the primary outcome (8.3%), which was defined as a composite of all-cause death and HHF; and (3) factors associated with HFimpEF achievement included female sex, non-ischemic HF etiology, and de novo HF.
The incidence of HFimpEF among patients treated for HFrEF has been reported to range from 10.4% to 52.1%, with an average of 22.6% [13] . It is well-established that patients whose systolic function recovered from severe dysfunction exhibit a better prognosis than those whose function did not recover. Although various studies have employed either “HFimpEF” or “HFrecEF,” with subtle variations in their definitions, numerous studies have consistently demonstrated that patients in these categories experience significantly reduced mortality and morbidity compared to those with persistently reduced EFs below 40% [13–17] . According to a study by Wang et al. [15], patients with HFrecEF had a lower risk of composite HF-related hospitalizations with mortality (adjusted HR 0.50, 95% CI 0.35–0.70) and all-cause hospitalizations with mortality (adjusted HR 0.59, 95% CI 0.49–0.73). Similarly, Nadruz et al. [16] reported that patients with HF with mid-range EF recovered from HFrEF (LVEF changed from < 40% to 40–55%) and exhibited a lower risk of composite events (death, LV assist device implantation, or transplantation) than the patients with HFrEF (HR 0.25, 95% CI 0.13–0.47). Our current findings also demonstrated an association between HFimpEF and a favorable primary outcome (a composite of all-cause death and HHF; HR 0.38, 95% CI 0.15–0.98), consistent with the findings from previous studies.
This study further elucidates significant characteristics of HFimpEF and its predictive factors. Patients with HFimpEF were more likely to be female, had a non-ischemic etiology or de novo HF, and exhibited lower echocardiographic volume parameters, indicating predictive factors for HFimpEF. The results aligned with the findings of previous studies [17–20] .
Female sex is a well-established predictor of HFimpEF, which might be attributed to less LV remodeling in response to volume and pressure overload and the ensuing enhanced myocardial recovery compared to males [19,21,22] . Similarly, non-ischemic etiology has been identified as an important determinant of EF improvement, as it is often characterized by potentially reversible myocardial dysfunction. In contrast, ischemic heart disease is frequently associated with irreversible scar formation and limited recovery. In line with these observations, our cohort showed that patients in the HFimpEF group commonly presented with de novo HF and exhibited smaller baseline echocardiographic chamber volumes. These findings supported that the likelihood of EF recovery diminishes as structural remodeling advances over a longer disease duration. Park et al. [23] reported that among HFrEF patients treated with ARNIs, those diagnosed with HFrEF more than six months before ARNI initiation showed less improvement in LVEF and poorer outcomes than those diagnosed within six months. A meta-analysis evaluating the effects of ARNIs on LV reverse remodeling demonstrated significant improvement during the 3-month follow-up [24] . This result underscores the importance of timely and appropriate intervention to alter disease progression before reaching the point of irreversibility. Taken together, these predictive factors highlighted the need for early identification of patients less likely to achieve EF recovery. In addition, these factors might aid in guiding risk factor modification as well as consideration of advanced therapeutic strategies.
On the other hand, there was no significant difference in the rate of HFimpEF achievement among patients stratified by ARNI dosage, suggesting that early ARNI therapy initiation following HF diagnosis, even at submaximal doses, could contribute to improved outcomes. These results were consistent with the findings of previous studies that evaluated ARNI administration at various doses, demonstrating that notable clinical and functional improvements were observed even in patients who received very low doses [24,25] .
In our study, the relative risk of the composite outcome of all-cause death and HHF in patients with HFimpEF was lower than that reported in previous studies examining similar composite outcomes [13,15] . Unlike previous studies on HF patients that predominantly utilized ACEIs or ARBs, the inclusion of patients exclusively using ARNIs in this study might have contributed to the more favorable outcomes observed. ARNI facilitates reverse LV remodeling via dual neurohormonal modulation, integrating the benefits of RAAS inhibition with neprilysin blockade. By preventing neprilysin-mediated degradation, ARNI enhances the natriuretic peptide system, further promoting its cardioprotective effects. It leads to an increase in the levels of natriuretic peptides, which promote vasodilation and sodium excretion, and also exert additional antifibrotic and antihypertrophic effects by modulating the extracellular matrix and reducing myocardial stiffness [26–29] . Such beneficial mechanisms validated our findings that ARNIs could provide superior outcomes compared to ACEIs or ARBs. Such beneficial mechanisms aligned with previous evidence from large-scale clinical trials, including PARADIGM-HF, which showed that ARNIs were superior to enalapril in reducing cardiovascular death and HHF rates [9] .
This study has several limitations. First, it was a retrospective cohort study. Thus, the potential for selection bias and residual confounding cannot be entirely excluded. Despite adjustments for known covariates, the unmeasured variables might have influenced the observed associations. Second, the total number of enrolled patients was relatively small, and the study population had a lower mean age compared to other HF registries. Third, the absence of sodium–glucose cotransporter 2 (SGLT2) inhibitors, now a key component of HF treatment, represents another limitation. Although the study period preceded the widespread adoption of SGLT2 inhibitors, the absence of these agents in the treatment regimen may provide a more lucid view of the isolated effects of ARNI therapy on EF recovery.
KEY MESSAGE
1. In this multicenter registry study (n = 198), we evaluated the incidence, clinical characteristics, predictive factors, and outcomes of HFimpEF among patients with HFrEF treated with ARNIs.
2. Approximately one-third of the patients achieved HFimpEF after one year of ARNI-based therapy. These patients were at significantly lower risk of all-cause death and HHF compared to those who remained in persistent HFrEF.
3. Female sex, non-ischemic etiology, and de novo HF were independent predictors of HFimpEF, suggesting that these clinical factors might help identify patients who are more likely to experience EF recovery with ARNI therapy.
Notes
CRedit authorship contributions
Dong-Eon Kim: investigation, data curation, formal analysis, writing - original draft; Min Gyu Kong: conceptualization, methodology, validation, writing - review & editing; Saerom Kim: investigation, data curation, formal analysis; Inki Moon: investigation, data curation; Hyun Woo Park: investigation, data curation; Hyung-Oh Choi: investigation, data curation; Hye Sun Seo: investigation, data curation; Yoon Haeng Cho: investigation, data curation; Nae-Hee Lee: investigation, data curation; Kwan Yong Lee: methodology, resources; Ho-Jun Jang: methodology, resources; Jae Sang Kim: methodology, resources; Ik Jun Choi: methodology, resources; Jon Suh: validation, writing - review & editing, supervision, project administration
Conflicts of interest
The authors disclose no conflicts.
Funding
This work was supported by the Soonchunhyang University Research Fund.