Eperisone-induced immediate hypersensitivity reactions
Article information
Abstract
Eperisone hydrochloride is a centrally acting muscle relaxant commonly prescribed for musculoskeletal pain, particularly in Korea and Japan. It induces relaxation of skeletal and vascular smooth muscle through central nervous system modulation and peripheral mechanisms, although its precise biochemical pathway remains unclear. While generally well tolerated, eperisone has been reported to cause immediate hypersensitivity reactions, including anaphylaxis. Because eperisone is frequently co-prescribed with non-steroidal anti-inflammatory drugs (NSAIDs), these reactions are often misdiagnosed as NSAID hypersensitivity, leading to unnecessary drug discontinuation and suboptimal pain management. A review of published case reports and pharmacovigilance data indicates that most eperisone-associated reactions are immediate-type, with anaphylaxis being clinically significant yet underrecognized. Skin prick tests and intradermal tests show variable diagnostic reliability, whereas drug provocation tests (DPTs) demonstrate the highest diagnostic accuracy and remain the gold standard. Basophil activation tests and serum tryptase measurement during DPT may serve as complementary diagnostic tools. Accurate identification of eperisone hypersensitivity is essential for preventing recurrent anaphylaxis and avoiding unnecessary NSAID discontinuation. This review summarizes the epidemiology, clinical features, and diagnostic strategies of eperisone hypersensitivity and emphasizes the importance of enhancing clinical awareness and referring affected patients to allergy specialists for proper evaluation.
INTRODUCTION
Eperisone hydrochloride, first introduced in Japan in 1983, is a centrally acting muscle relaxant widely used to alleviate skeletal muscle spasms. Unlike agents that act directly on neuromuscular junctions, eperisone primarily modulates central nervous system pathways, thereby reducing hyperactive reflexes and restoring the balance between excitatory and inhibitory neuronal impulses. These actions not only relieve muscle spasticity but may also produce mild sedative effects [1]. However, its exact biochemical mechanism remains unclear, although recent studies suggest that the microglial purinergic receptor 7 may be a potential pharmacological target for drug repositioning of eperisone hydrochloride [2]. Eperisone has also been reported to modulate sympathetic nervous system activity, further contributing to its muscle relaxant effects in skeletal muscles [3]. With these mechanisms, eperisone improves muscle tone by relaxing both skeletal and vascular smooth muscles and is used to treat spastic paralysis [4]. In addition to oral administration, a transdermal patch formulation of eperisone has been developed, providing a more potent and longer-lasting therapeutic effect than oral administration [5].
Clinically, eperisone is often co-prescribed with non-steroidal anti-inflammatory drugs (NSAIDs) to enhance its therapeutic effects. Combination therapy with eperisone and ibuprofen has been shown to produce superior pain relief and functional improvement compared with ibuprofen alone in patients with acute non-specific back pain and muscle spasm [6]. A pharmacokinetic study demonstrated no significant pharmacokinetic interactions between the two drugs, including the major active metabolites of pelubiprofen, when comparing monotherapy and combination therapy [7]. Despite its widespread use, especially among elderly patients in Korea [8], adverse drug reactions to eperisone are underrecognized. Because NSAIDs are well-known culprits of drug hypersensitivity [9], hypersensitivity reactions occurring during combined therapy are often misattributed to NSAIDs rather than eperisone. This diagnostic challenge leads to underreporting and mismanagement of eperisone-related hypersensitivity. In real-world practice, however, immediate hypersensitivity to eperisone appears to be relatively frequent, particularly in Korea, where prescribing patterns may further complicate timely and accurate diagnosis.
This review aims to summarize current evidence on eperisone-induced hypersensitivity, including its clinical presentation, diagnostic strategies, and management considerations, to improve recognition and ensure safer prescribing practices.
EPERISONE AND RELATED COMPOUNDS
There is currently no evidence of cross-reactivity with other muscle relaxants. Nevertheless, eperisone shares a close structural similarity with tolperisone hydrochloride (dimethyl-2,4′-piperidino-3-propiophenone), which may have clinical relevance. Two cases of tolperisone-induced anaphylaxis have been reported [10,11]. Recent findings suggest that reactive degradation products of tolperisone may contribute to hypersensitivity reactions, indicating that both the parent compound and its metabolites may play a role in tolperisone hypersensitivity [12]. Chemical structures of eperisone and related compounds are shown in Figure 1.
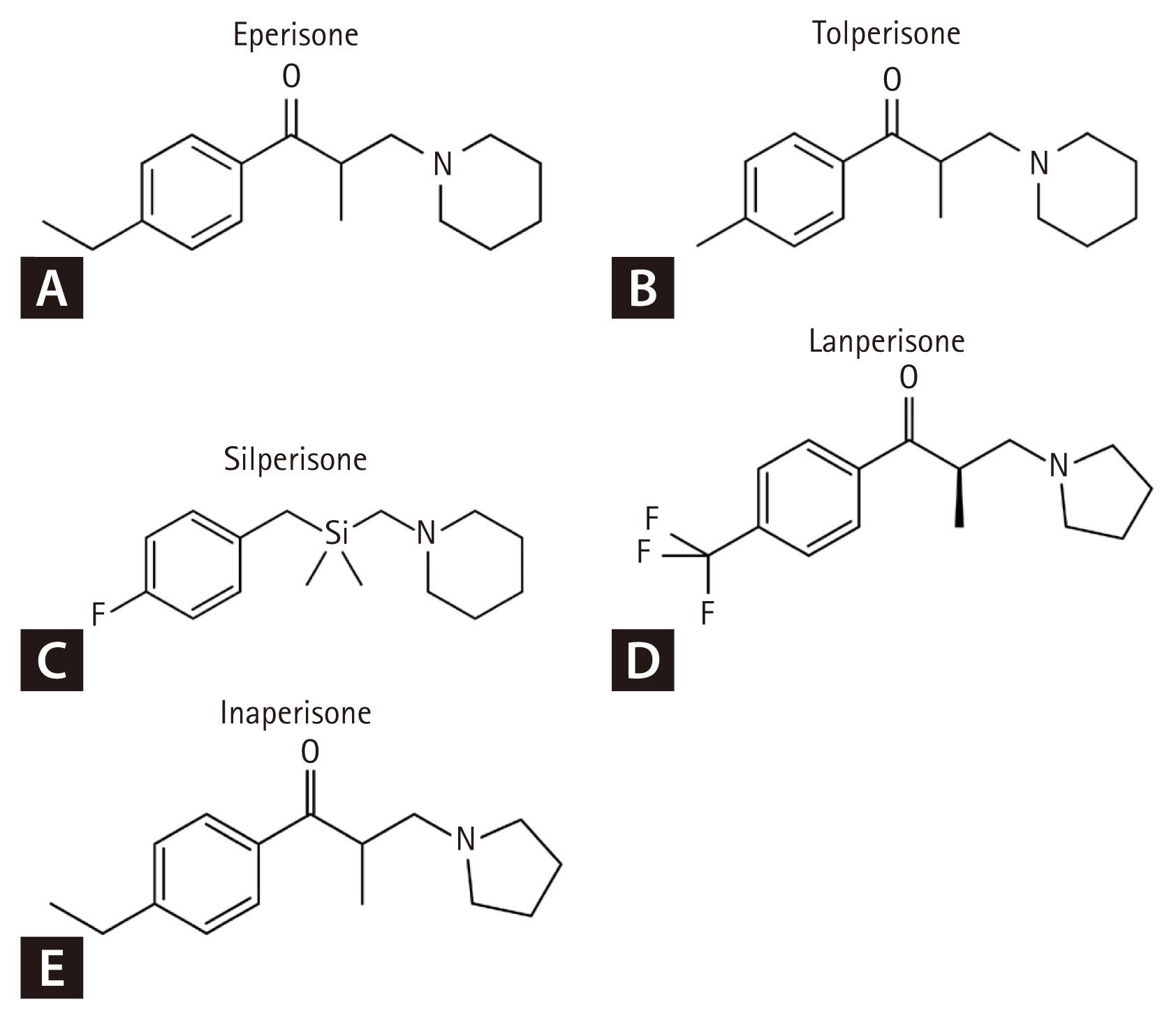
Chemical structures of eperisone (A) and related compounds (B–E). Panels A, C, D, and E were reproduced from Wikimedia Commons (Public Domain). Panel B was reproduced from Wikimedia Commons under CC BY-SA 3.0 license.
In Korea, eperisone is the only commercially available muscle relaxant of this class; therefore, clinical evidence of cross-reactivity with related compounds has not yet been documented. Although structural similarities suggest that cross-reactivity is theoretically possible, real-world clinical data confirming such reactions remain unavailable.
ADVERSE REACTIONS TO EPERISONE
Eperisone was originally developed by a Japanese company and is marketed only in a limited number of countries, including Japan, Korea, India, Bangladesh, and Italy. Consequently, reports of adverse reactions related to eperisone remain globally limited. Randomized controlled trials have generally demonstrated a favorable safety profile for eperisone, with gastrointestinal disturbances occurring in approximately 5% of treated patients [13,14]. However, several serious adverse reactions to eperisone have also been reported. Non-allergic reactions include a case of bilateral tonic seizure [15] and two cases of severe QT prolongation and Torsade de Pointes [16,17].
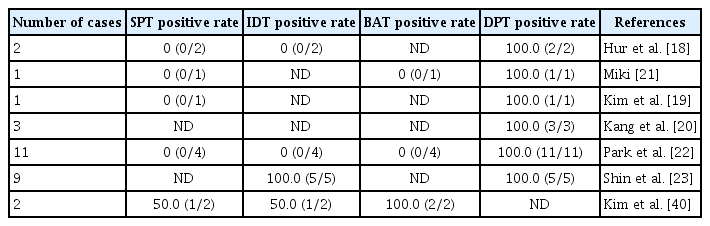
Immediate hypersensitivity reactions—including urticaria, angioedema, and anaphylaxis—represent the most clinically significant immunologically mediated adverse events. The first report of eperisone-induced immediate hypersensitivity reactions, involving angioedema and anaphylaxis, was published in Korea in 2012 [18]. As eperisone use has increased, subsequent case reports have documented additional anaphylaxis episodes, including one case in 2013 [19], three cases in 2017 [20], and another reported in Japan [21].
In a 2019 pharmacovigilance study from Korea, 207 patients with adverse reactions to eperisone were analyzed. Cutaneous manifestations such as urticaria, pruritus, and angioedema were most frequent (30.4%), and anaphylaxis accounted for the fifth most common reaction [22]. The investigators additionally enrolled and analyzed 11 patients with eperisone-induced anaphylaxis. Similarly, Shin et al. [23] found that 9 of 23 patients with eperisone-induced immediate hypersensitivity reactions met diagnostic criteria for anaphylaxis. In addition, data from a single tertiary hospital in Korea reported that 5.7% of drug-induced anaphylaxis cases were attributable to eperisone [24]. Collectively, these reports indicate that eperisone-induced immediate allergic reactions represent a relatively common and clinically significant problem in Korea.
CLINICAL FEATURES OF EPERISONE-INDUCED ANAPHYLAXIS
Review of published reports indicates that most eperisone-associated hypersensitivity reactions are immediate-type responses, including urticaria, angioedema, and anaphylaxis. Delayed-type reactions, including fixed drug eruption and acute generalized exanthematous pustulosis, have been reported only rarely. Based on the type of reaction, diagnostic tools may be selected, including skin prick test (SPT), intradermal test (IDT), basophil activation test (BAT), and drug provocation test (DPT).
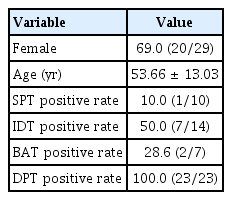
A pooled analysis of published case reports and observational studies identified 29 patients with confirmed eperisone-induced anaphylaxis. The majority were female (69.0%, 20/29), and the mean age was 53.66 ± 13.03 years (range, 28–80 yr). Their clinical features are summarized in Tables 1 and 2.
Among patients undergoing skin testing, SPT was positive in only one patient from pooled case reports (10.0%, 1/10), whereas IDT was positive in 50.0% (7/14). BAT was positive in 2 cases (28.6%, 2/7). Overall, IDT appears to have a relatively higher diagnostic yield (50% positivity) in patients with severe immediate reactions, although negative results do not exclude eperisone hypersensitivity. DPT demonstrated 100% positivity among those tested (performed in 23 of 29 patients), confirming its role as the gold standard diagnostic tool.
DIAGNOSTIC METHODS OF EPERISONE HYPERSENSITIVITY
Allergic skin tests
Skin testing remains a cornerstone in the evaluation of IgE-mediated drug hypersensitivity. However, the diagnostic utility of eperisone skin testing remains controversial. Detailed testing results from previously published case reports are summarized in Table 2. SPT was positive in only one patient in pooled case reports (10.0%, 1/10), whereas IDT was positive in 50.0% (7/14). However, IDT outcomes were not consistent across reports and varied substantially among investigators (Table 2). Furthermore, given that eperisone is an oral medication, IDT with this agent raises concerns regarding its appropriateness. For many drugs, there is insufficient evidence to recommend appropriate testing concentrations [25]. Regarding eperisone, a concentration of 10 mg/mL has been suggested as a non-irritating concentration for IDT based on a previous study by Shin et al., in which IDT was performed in 10 healthy volunteers without prior exposure to eperisone [23].
DPT
DPTs are regarded as the reference standard for confirming whether a patient can safely receive a suspected medication. Among the 29 published cases of eperisone immediate reaction, 23 underwent DPT and demonstrated 100% positivity, confirming its role as the gold standard diagnostic tool (Table 1, 2). Therefore, DPT should be recommended to confirm the diagnosis when clinical suspicion remains high. However, DPTs are generally contraindicated in severe hypersensitivity reactions, including severe non–IgE-mediated reactions such as severe cutaneous adverse drug reactions (e.g., Stevens–Johnson syndrome/toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis), drug-induced liver injury, or drug-induced cytopenia, as well as in cases of severe drug-induced anaphylaxis, particularly in the absence of reliable skin testing or when the potential benefit does not outweigh the risk [26].
DPT should be performed under medical supervision in a setting equipped for treating anaphylaxis, including resuscitation equipment, with well-trained medical staff who are proficient in performing DPT and in recognizing and treating hypersensitivity reactions, including anaphylaxis. Certain co-medications are contraindicated [26] if they may complicate emergency treatment. Concomitant use of β-blockers (BBs) and angiotensin-converting enzyme inhibitors has been associated with increased severity of anaphylaxis. BBs may blunt compensatory cardiovascular responses, enhance mast cell mediator release, and diminish the therapeutic effects of both endogenous and exogenous epinephrine.
Dosing and time intervals in DPT are determined by the clinical manifestations of the suspected drug. In a U.S. study [27], outcomes of patients with low-risk histories undergoing one- or two-step challenges (n = 456) were compared with those undergoing multistep (three- or four-step) challenges (n = 74). The frequency of reactions was similarly low across groups (11% vs. 12%), and most reactions were mild to moderate. However, in cases of severe immediate reactions or in patients with high-risk histories, the starting dose is suggested not to exceed 1:100 of the single target dose [28,29], depending on the severity of the reaction, and the time interval between doses should be at least 30 minutes, although certain drugs and specific situations may require longer intervals. Following the final dose, the observation period should be a minimum of 1–2 hours, depending on individual risk, as severe reactions such as anaphylaxis most commonly occur within this time interval [30,31].
A DPT can be considered positive if it reproduces objective signs and symptoms compatible with the original reaction. Subjective symptoms may precede objective ones. However, subjective symptoms without accompanying objective findings are more likely stress- or anxiety-induced reactions (nocebo reactions) and may be difficult to differentiate from drug hypersensitivity reactions [32,33]. In such equivocal cases, placebo-controlled DPTs are required to ensure accurate interpretation.
Serum tryptase may be helpful in distinguishing between hypersensitivity and anxiety reactions, especially in severe cases. Following allergen exposure, serum tryptase concentrations typically peak within 1–2 hours and gradually return to baseline within approximately 24 hours, coinciding with resolution of clinical symptoms [34]. The current gold-standard laboratory approach for confirming anaphylaxis consists of measuring total serum tryptase during the acute phase of the reaction and comparing it with a baseline value obtained at least 24 hours later [35]. Although a significant rise in serum tryptase strongly supports the diagnosis of anaphylaxis, not all cases exhibit elevated levels; therefore, a normal concentration does not exclude the condition. An increase that meets the formula (1.2 × baseline tryptase) + 2 μg/L within 0.5–2 hours after symptom onset is considered diagnostic for anaphylaxis [36,37]. In a retrospective study conducted in 2022, Liang et al. [24] analyzed 53 cases of anaphylaxis at a Korean tertiary hospital. Drugs were identified as the most frequent triggers (42 cases; 79.2%), with eperisone accounting for 3 cases (5.7%), emphasizing its clinical relevance as a potential culprit in drug-induced anaphylaxis. Among the 3 cases, two showed markedly elevated tryptase levels after provocation testing, whereas one did not meet the criteria for a positive tryptase response, suggesting that measurement of serum tryptase may serve as a valuable adjunct in confirming eperisone-induced anaphylaxis.
Recommended procedure of eperisone DPT
We suggest the following procedure for eperisone DPT.
1. The patient should be admitted to a hospital setting with close monitoring and resuscitation equipment available under the medical supervision of allergy specialists.
2. The patient’s health status should be stable on the day of testing, and intravenous access should be secured.
3. Verbal and written informed consent must be obtained before DPT.
4. The starting dose is suggested at 5 mg (1:10 of the single target dose) and increased to 10, 25, and 50 mg at 30-minute to 1-hour intervals. Dosing and time intervals may be adjusted according to the severity of prior anaphylactic reactions.
5. Serum tryptase levels may be measured as an adjunct to anaphylaxis diagnosis in cases of severe reactions.
6. The observation period should be a minimum of 1–2 hours after the final dose.
7. In the case of a positive DPT, clinicians should provide a drug allergy identification card and educate the patient to distinguish between the prohibited culprit drug and safe alternatives. The reaction should be reported in electronic drug hypersensitivity or allergy reporting systems linked to electronic medical records to prevent repeated reactions.
8. In the case of a negative DPT, clinicians should explain that the drug can be used and ensure that the allergy label is removed from all medical records.
BAT
Accurate diagnosis of immediate drug hypersensitivity remains a clinical challenge, underscoring the need for reliable and non-invasive diagnostic tools. BAT has gained attention as a complementary in vitro method that closely replicates the immune response observed in vivo [38].
Basophils, which express high-affinity IgE receptors, represent a convenient cellular model for evaluating IgE-mediated mechanisms. When exposed to an offending allergen, cross-linking of IgE on the basophil surface triggers cellular activation, leading to the upregulation of surface molecules such as CD63 and CD203c and the activation of intracellular pathways, including phosphorylation of p38 MAPK. BAT enables detection of these activation events at the cellular level through multicolor flow cytometry using fluorochrome-labeled monoclonal antibodies. Furthermore, by passively sensitizing donor basophils with a patient’s serum, the assay may help establish whether the reaction is IgE dependent [39].
Standardization of BAT and its analysis is essential to ensure consistent results across laboratories. Standardized BAT protocols have been proposed and validated, demonstrating acceptable intra- and inter-laboratory reproducibility based on recognized quality criteria.
Regarding the role of BAT in identifying culprit drugs in drug allergy, a Korean study by Kim et al. [40] reported an overall BAT positivity rate of 57.9% among 19 patients with drug-induced anaphylaxis. Notably, two patients had confirmed eperisone-induced anaphylaxis, and BAT yielded positive results in both cases, corresponding to a positivity rate of 100% for eperisone-related reactions in that cohort.
However, as summarized in Table 2, the reported positivity rates of BAT vary considerably across studies. Although BAT protocols are relatively well standardized, the limited number of reported cases of eperisone-induced anaphylaxis restricts the ability to draw definitive conclusions regarding its diagnostic accuracy. Larger, systematically designed studies are required to clarify the clinical utility of BAT in diagnosing eperisone hypersensitivity.
SPECIAL CONSIDERATIONS FOR EPERISONE ANAPHYLAXIS
Accurate identification of the causative drug is crucial for preventing recurrence. In patients with suspected eperisone hypersensitivity, confirmatory testing with in vivo methods (IDT and DPTs) and in vitro assays (e.g., BAT) by an allergy specialist is recommended. Patients should be provided with an individualized emergency action plan, including training on the use of epinephrine auto-injectors.
Importantly, eperisone is frequently co-prescribed with NSAIDs for pain management. Consequently, eperisone-induced anaphylaxis is often misattributed to NSAIDs. To accurately identify the causative agent, a diagnostic process—including IDT and DPTs—under the supervision of an allergy specialist is essential. A structured diagnostic approach is essential to correctly identify eperisone as the culprit drug and to avoid unnecessary NSAID restriction.
The mechanism of eperisone-induced anaphylaxis remains unclear. Elevation of serum tryptase levels after DPT, together with BAT findings, suggests that mast cell activation serves as the final effector pathway in eperisone-induced anaphylaxis. However, given the negative SPT results and only modest positivity on IDT, non-IgE-mediated mast cell activation may play a critical role in this condition. Further studies are warranted to elucidate the precise mechanisms underlying mast cell activation in eperisone-induced anaphylaxis.
CONCLUSION
Eperisone-induced hypersensitivity, including anaphylaxis, remains underrecognized despite its clinical significance. Given its frequent co-prescription with NSAIDs, eperisone-related reactions are often misdiagnosed as NSAID hypersensitivity. Careful diagnostic evaluation, including IDT and DPT, is therefore essential to accurately identify the culprit drug. Early recognition, appropriate patient education, and implementation of avoidance strategies are crucial for preventing recurrence and ensuring safe and effective pain management in patients requiring muscle relaxants.
Notes
CRedit authorship contributions
Gyu-Young Hur: conceptualization, data curation, writing - review & editing; Jung-Won Park: writing - review & editing, supervision
Conflicts of interest
The authors disclose no conflicts.
Funding
None