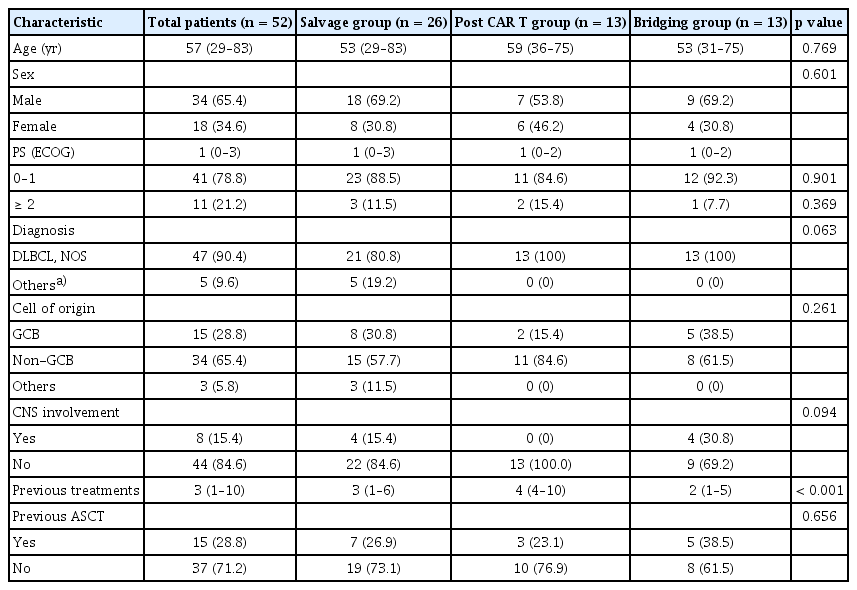
Real-world use of polatuzumab vedotin combined with bendamustine and rituximab for patients with relapsed or refractory large B-cell lymphoma
Article information
Abstract
Background/Aims
Polatuzumab vedotin combined with bendamustine and rituximab (Pola-BR) is a treatment option for relapsed/refractory diffuse large B-cell lymphoma (R/R DLBCL), particularly as bridging therapy before chimeric antigen receptor (CAR) T-cell infusion. However, real-world data regarding its feasibility, efficacy, and safety in Korean patients are limited.
Methods
We conducted a single-center retrospective study of 52 patients with R/R DLBCL treated with Pola-BR between April 2021 and April 2024. Patients were categorized into three groups: salvage (n = 26), post-CAR T (n = 13), and bridging (n = 13). The primary endpoints were objective response rate (ORR) and complete response (CR) rate; progression-free survival (PFS), overall survival (OS), and safety were secondary endpoints.
Results
The overall ORR was 51.9% (27/52), with 36.5% (19/52) of the patients achieving CR. The ORRs were 46.2%, 53.8%, and 61.5% in the salvage, post-CAR T, and bridging groups, respectively, with corresponding CR rates of 30.8%, 38.5%, and 46.2%. The bridging group achieved the highest response rates despite receiving a median of only one cycle, and patients with fewer prior treatment lines demonstrated superior responses. Grade 3–4 hematologic toxicities occurred in nearly all post-CAR T (100%) and salvage (92.3%) patients but were significantly lower in the bridging group (46.2%).
Conclusions
Pola-BR provided meaningful disease control in patients with R/R DLBCL. Its use as a bridging therapy before CAR T-cell infusion was associated with high response rates, favorable safety, and a successful transition to cellular therapy, underscoring its value as a practical option in this setting.
INTRODUCTION
Diffuse large B-cell lymphoma (DLBCL) is an aggressive subtype of non-Hodgkin’s lymphoma (NHL), accounting for 30–40% of NHL cases. The standard first-line therapy for newly diagnosed DLBCL consists of immunochemotherapy using rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP). A significant proportion of patients achieve long-term remission with R-CHOP; however, approximately 30–40% experience relapse following an initial response or have primary resistance to treatment [1].
For patients with relapsed or refractory DLBCL (R/R DLBCL), salvage strategies traditionally involve intensive chemotherapy followed by autologous stem cell transplantation (ASCT). However, this approach is only effective in a subset of patients, highlighting the need for alternative therapeutic options to treat this disease. Consequently, novel treatment modalities with mechanisms distinct from those of conventional cytotoxic chemotherapy have been developed, including antibody-drug conjugates (ADCs), bispecific T-cell engagers, and chimeric antigen receptor (CAR) T-cell therapies. Although these advancements have improved the outcomes of R/R DLBCL, challenges remain regarding accessibility, financial burden, and feasibility. In particular, CAR T-cell therapies are not universally applicable. These therapies present significant barriers, such as treatment-related toxicities and limited efficacy in patients experiencing disease progression after CAR T-cell therapy [2,3].
Polatuzumab vedotin is a CD79b-directed ADC consisting of a humanized immunoglobulin G1 monoclonal antibody conjugated to the antimitotic agent monomethyl auristatin E. It is approved for the treatment of DLBCL as both frontline and salvage therapy in combination with cytotoxic chemotherapy. Polatuzumab vedotin combination with bendamustine and rituximab (Pola-BR) is indicated as a salvage regimen for patients with R/R DLBCL. The U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) approved Pola-BR based on the promising outcomes of a Phase II clinical trial [4].
According to the National Comprehensive Cancer Network (NCCN) guidelines, Pola-BR is recommended as a second-line therapy for patients with R/R DLBCL who are ineligible for CAR T-cell therapy, particularly those who experience relapse within one year of first-line therapy or have a primary refractory disease. Additionally, Pola-BR may serve as a second-line treatment option for patients who relapse more than one year after completing first-line therapy and are not candidates for transplantation. Furthermore, Pola-BR is considered a potential bridging therapy prior to CAR T-cell therapy [5].
Despite the inclusion of Pola-BR in the treatment guidelines, data on its real-world feasibility, efficacy, and safety remain limited, particularly in Korean patients. Therefore, in this study, we retrospectively analyzed the therapeutic efficacy, survival outcomes, and adverse events (AEs) associated with Pola-BR in both salvage and bridging CAR T-cell therapy settings.
METHODS
Study design
This single-center retrospective study analyzed the data of 52 patients with R/R DLBCL who received Pola-BR at Samsung Medical Center (Seoul, Korea) between April 2021 and April 2024 (Fig. 1A). Among the patients, 47 had DLBCL, not otherwise specified (NOS) and the remaining five had other high-grade B-cell lymphoma subtypes, including double-hit lymphoma, primary mediastinal large B-cell lymphoma, Epstein–Barr virus-positive DLBCL, and post-transplant lymphoproliferative disorder. Based on the clinical setting in which Pola-BR was administered, patients were categorized into three groups: salvage therapy (salvage group, n = 26), post-CAR T-cell failure (post-CAR T group, n = 13), and bridging therapy before CAR T-cell therapy (bridging group, n = 13). This study aimed to assess the objective response rate (ORR), complete response (CR) rate, progression-free survival (PFS), overall survival (OS), and safety profile. Given the retrospective nature of the study, the requirement for informed consent was waived, and the study protocol was approved by the Institutional Review Board of Samsung Medical Center (approval number: IRB NO. 2025-03-119). This research was conducted in accordance with the World Medical Association’s Declaration of Helsinki.
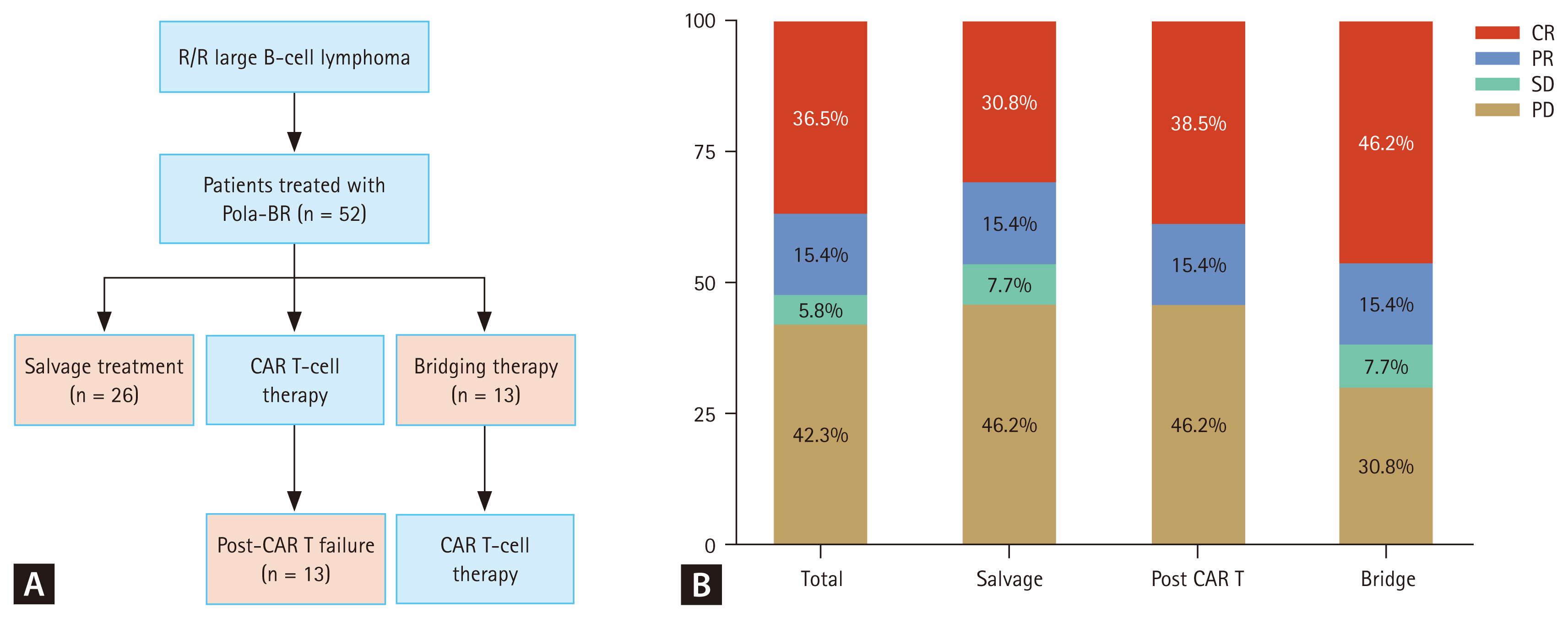
Characteristics and treatment outcomes of Pola-BR. (A) Patients treated with Pola-BR. (B) Overall objective response rates and subgroups. R/R, relapsed or refractory; Pola-BR, polatuzumab vedotin combined with bendamustine and rituximab; CAR, chimeric antigen receptor; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease.
Data collection
The electronic medical records of the patients with R/R DLBCL who met the study criteria were reviewed to collect data on demographics (age and sex), disease characteristics (stage, primary disease sites, presenting symptoms, and laboratory findings, including complete blood count, lactate dehydrogenase, and C-reactive protein), treatment history, and clinical outcomes. Relapsed disease was defined as disease progression after achieving a response to prior therapy, whereas refractory disease was defined as failure to achieve a response or progression within 12 months of the last therapy, including post-ASCT. All patients received Pola-BR, consisting of polatuzumab vedotin (1.8 mg/kg, day 2), bendamustine (90 mg/m2, days 2 and 3), and rituximab (375 mg/m2, day 1). Treatment responses were assessed using the Lugano 2014 criteria with positron emission tomography (PET)-based Deauville scores of 1–3 defined as complete metabolic response (PET-CR) [6]. AEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 [7].
Statistical analysis
The demographic and baseline characteristics were summarized using descriptive statistics. Continuous variables were compared using Student’s t-test or Wilcoxon rank-sum test, as appropriate, whereas categorical variables were analyzed using chi-square or Fisher’s exact tests. PFS and OS were estimated using the Kaplan–Meier method, and group comparisons were performed using log-rank tests. Univariate and multivariate Cox proportional hazards regression models were used to identify the prognostic factors for survival outcomes. Statistical significance was defined as a two-sided p value of < 0.05.
Primary statistical analyses were conducted using IBM SPSS Statistics software (version 26.0; IBM Corp., Armonk, NY, USA). Swimmer plots were generated using Google Colaboratory (Colab) and Python (version 3.10; pandas, Matplotlib). Kaplan-Meier survival curves and median survival estimates were generated using R (version 4.3.2; package survival, survminer, ggplot2, and ggtext). The distribution of the best responses according to prior treatment lines was visualized as 100% stacked bar charts using dplyr and ggplot2 in the same R environment.
RESULTS
Patient characteristics
A total of 52 patients were included in the analysis: 26 (50.0%) in the salvage group, 13 (25.0%) in the post-CAR T group, and 13 (25.0%) in the bridging group. The baseline characteristics were similar across the three groups (Table 1). The median age of all patients was 57 years (range, 29–83 yr), and the median Eastern Cooperative Oncology Group (ECOG) performance status was 1 (range, 0–3). Most patients (90.4%) were diagnosed with DLBCL (NOS), with the non-germinal center B-cell subtype being predominant (65.4%). Eight patients had a history of secondary central nervous system (CNS) involvement before Pola-BR treatment, and more than 90% were refractory to previous treatments. Because CAR T-cell therapy is generally administered as a third-line treatment, patients in the bridging group had a median of two prior treatment lines (range, 1–5), which was significantly fewer than that in the salvage or post-CAR-T groups. Additionally, 15 patients had a history of ASCT during treatment.
Response rate and survival outcome of Pola-BR
In the total cohort, the ORR following Pola-BR therapy was 51.9% (27/52), with 36.5% (19/52) achieving a CR (Fig. 1B). When analyzed by treatment intent, the salvage group had an ORR of 46.2% (12/26), with 30.8% (8/26) achieving CR. The post-CAR T group showed a similar ORR of 53.8% (7/13), with 38.5% (5/13) achieving CR. The bridging group exhibited the highest ORR of 61.5% (8/13), with 46.2% (6/13) achieving CR (Fig. 1B). The median PFS after the initiation of Pola-BR for the entire cohort was 2.9 months, with no significant differences among the three groups (Fig. 2A). In contrast, the median OS after Pola-BR was 6.2 months for all patients. Patients in the bridging group had a longer median OS than those in the salvage and post-CAR T groups (12.5 vs. 3.8 and 5.2 mo, respectively), although the difference was not statistically significant (p = 0.1, Fig. 2B). This observed difference in OS may be explained by the subsequent success of CAR T-cell therapy following Pola-BR bridging therapy.
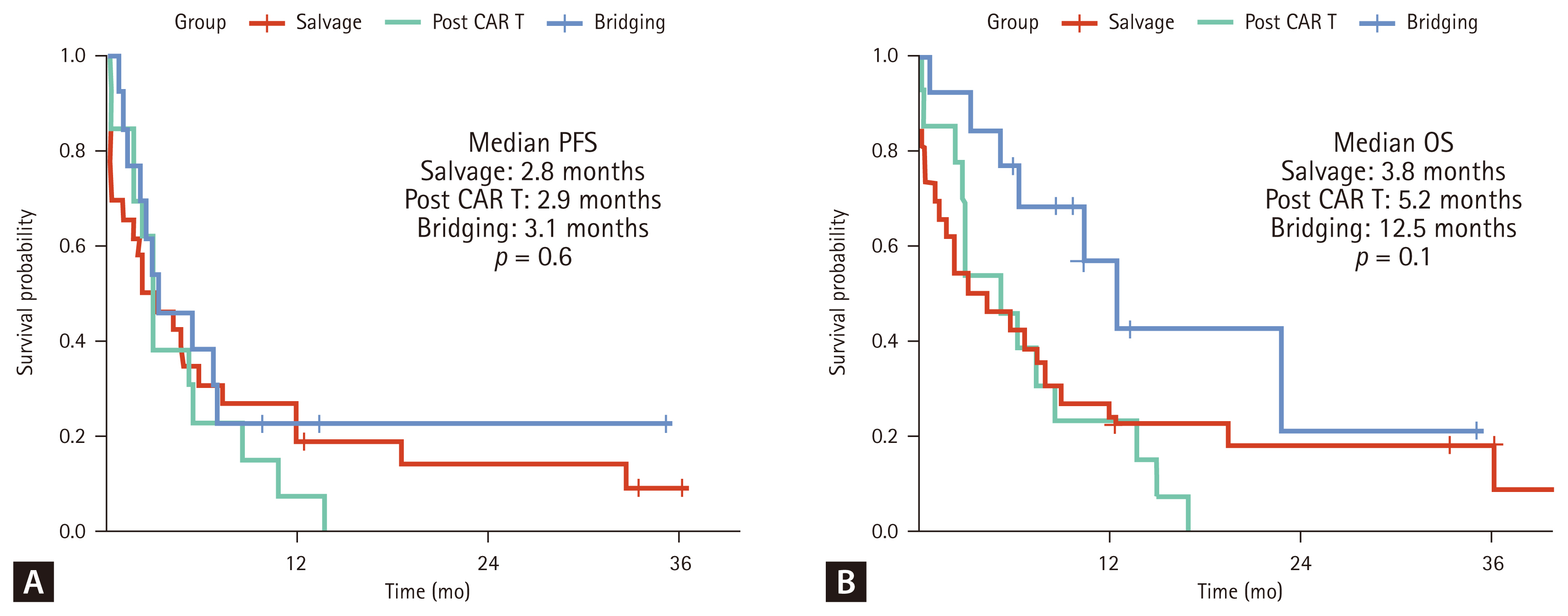
Comparison of survival outcomes by treatment intent. (A) PFS. (B) OS. PFS, progression-free survival; OS, overall survival; CAR, chimeric antigen receptor.
Notably, 12 of the 13 patients (92.3%) who received Pola-BR as bridging chemotherapy successfully proceeded to CAR T-cell infusion (Fig. 3A), suggesting that Pola-BR effectively stabilized the disease, ensuring that most patients reached infusion without significant progression. Furthermore, the response to Pola-BR bridging therapy influenced the survival outcomes after CAR T-cell therapy. Patients who achieved CR after Pola-BR bridging therapy had a median PFS of 7.0 months, which was significantly longer than that of the patients who did not achieve CR (2.4 mo, p = 0.010, Fig. 3B). Similarly, the median OS was significantly different between the patients who achieved CR and those who did not (22.9 vs. 5.4 mo, p = 0.003; Fig. 3C).
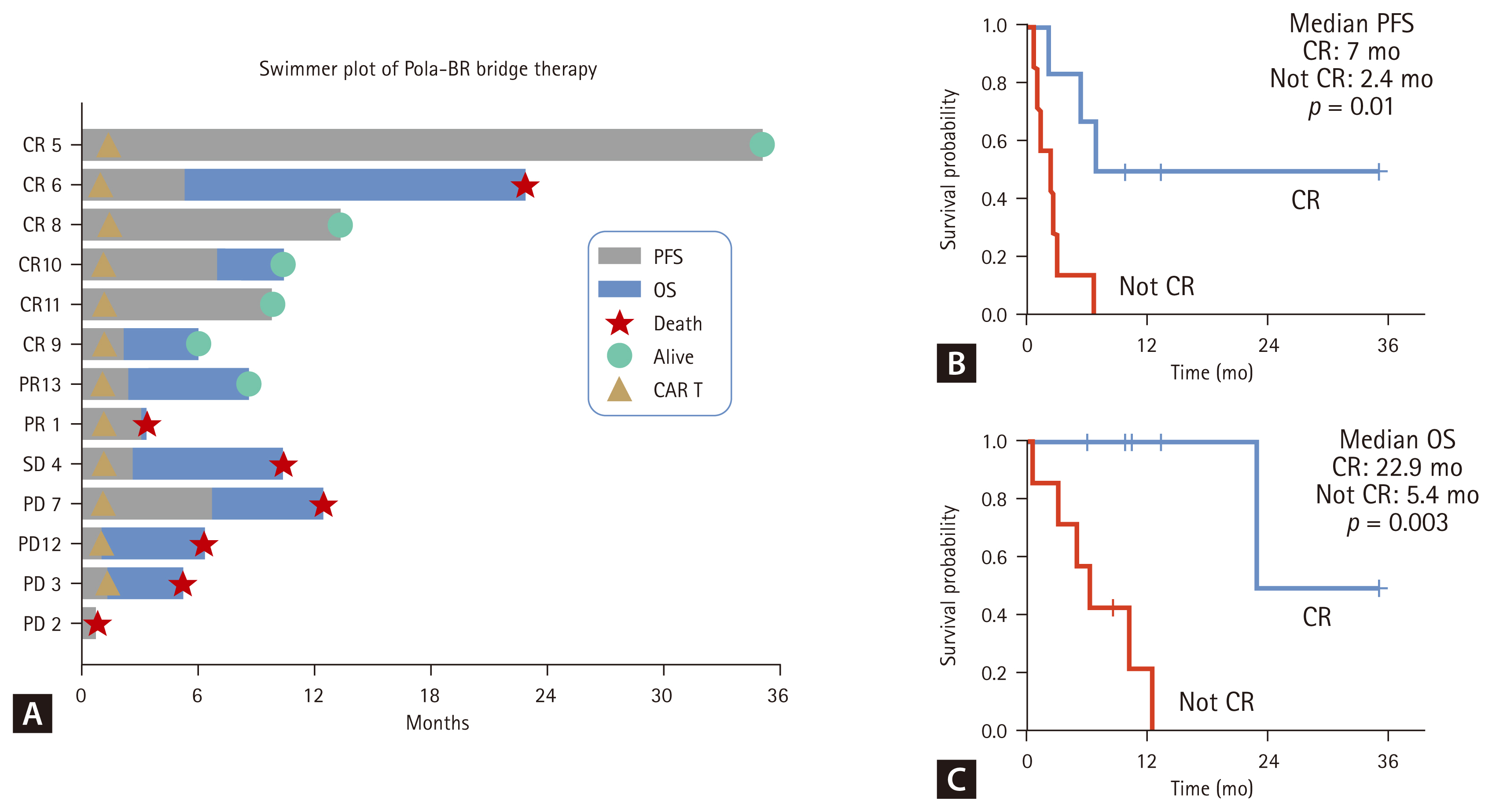
Outcomes of Pola-BR as bridging therapy. (A) Swimmer plot of patients who received Pola-BR as bridging chemotherapy. (B) PFS by response (CR vs. Not CR). (C) OS by response (CR vs. Not CR). Pola-BR, polatuzumab vedotin combined with bendamustine and rituximab; PFS, progression-free survival; OS, overall survival; CAR, chimeric antigen receptor; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease.
Prognostic factors associated with survival
Next, we investigated the baseline clinical factors associated with survival outcomes using univariate and multivariate analyses. In the univariate analysis for PFS, refractory disease was strongly associated with worse outcomes (hazards ratio [HR], 6.22; 95% confidence interval [CI], 1.76–22.00; p = 0.004). This finding remained significant in the multivariate analysis (HR, 7.67; 95% CI, 1.66–35.50; p = 0.009) (Supplementary Table 1). Female sex was independently associated with superior PFS (HR, 0.376; 95% CI, 0.163–0.871; p = 0.022). No other baseline variables, including age, intention to use Pola-BR, CNS involvement, or number of prior treatment lines, were significantly associated with PFS.
For OS, poor performance status (ECOG ≥ 2), refractory disease, and absence of prior ASCT were associated with inferior survival in univariate analysis (Supplementary Table 2). Specifically, patients with refractory disease had a significantly higher risk of death (HR, 6.55; 95% CI, 1.54–27.99; p = 0.011). Multivariate analysis confirmed refractory disease as an independent adverse prognostic factor for OS (HR, 5.80; 95% CI, 1.13–29.70; p = 0.035). Conversely, patients who underwent ASCT demonstrated improved OS (HR, 0.326; 95% CI, 0.115–0.921; p = 0.035).
Impact of prior treatment lines on response
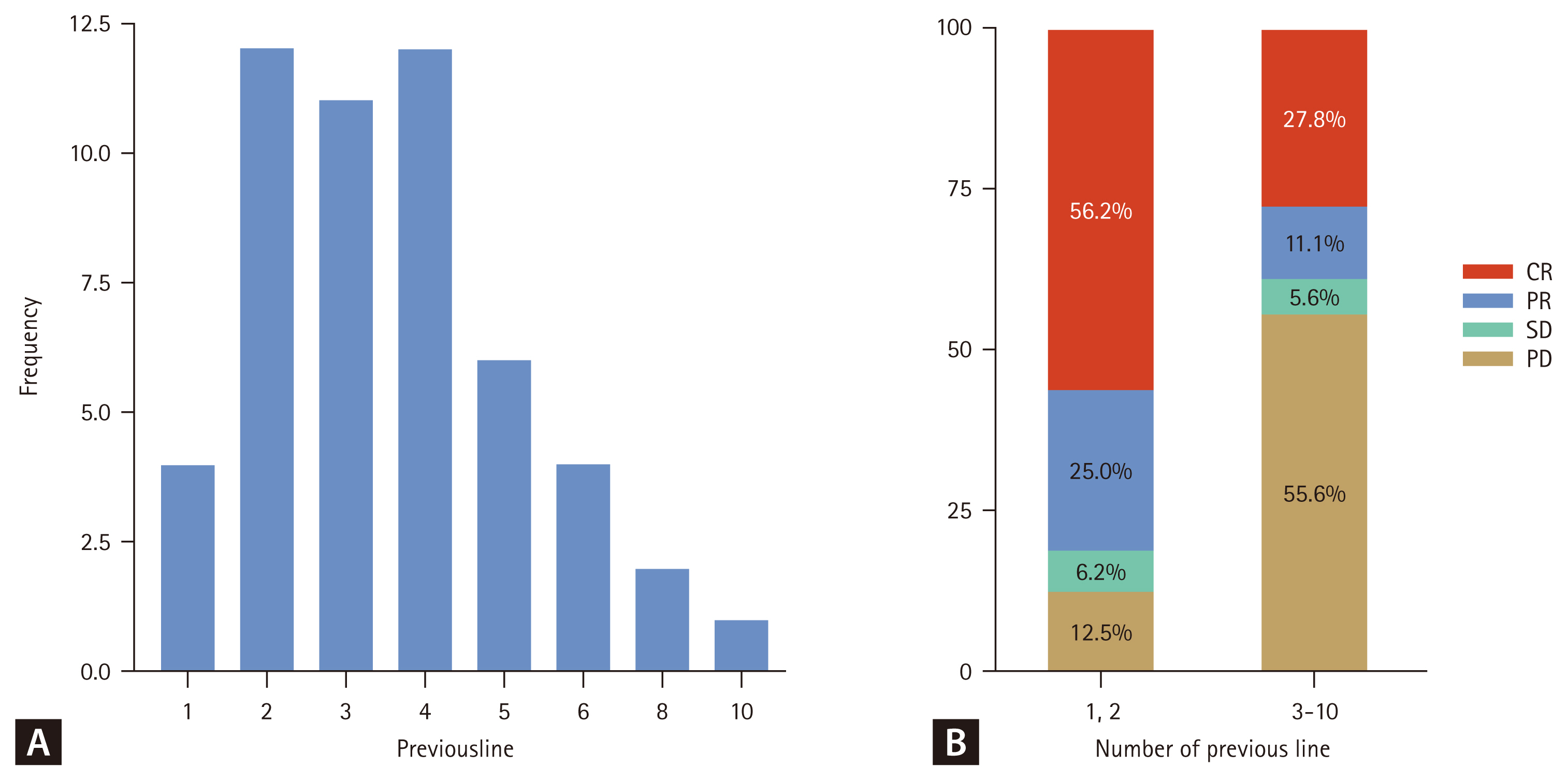
Given these heterogeneous outcomes, we evaluated whether the number of treatment lines before Pola-BR influenced the response. The median number of prior treatments before Pola-BR in the entire cohort was three (range, 1–10), indicating a diverse treatment history among the patients (Fig. 4A). Based on this data, patients were categorized into two groups: early use (≤ 2 prior lines of therapy) and late use (> 2 prior lines of therapy). Patients in the early use group demonstrated higher ORR and CR rates than those in the late use group (Fig. 4B). Specifically, the early use group had a CR rate of 56.2% and an ORR of 81.2%, whereas the late use group had a significantly lower CR rate of 27.8% and an ORR of 38.9% (Fig. 4B).
CNS involvement and outcomes
Of the 52 patients, 10 (19.2%) had a history of CNS involvement at some point during the course of the disease (Supplementary Table 3). At the time of Pola-BR initiation, five patients presented with active CNS disease, while two had a history of CNS involvement that had been controlled before Pola-BR. The remaining three patients developed CNS relapse after achieving a systemic response to Pola-BR. Overall, the outcomes of the patients with active CNS disease at baseline were poor. These patients predominantly achieved only transient or limited responses, with most experiencing progression within a short period (median PFS, 0.33–2.40 mo). In contrast, patients with prior CNS involvement who were in remission at Pola-BR initiation demonstrated more favorable outcomes, including durable systemic responses and controlled CNS status in some cases. Notably, three patients developed CNS relapse despite systemic disease control, highlighting the limited efficacy of Pola-BR in preventing CNS recurrence. Two patients in the bridging group experienced leptomeningeal or parenchymal relapse, while one patient in the salvage group had CNS relapse despite achieving systemic CR. In the bridging cohort, CNS outcomes were heterogeneous. Most patients without CNS involvement successfully proceeded to CAR T-cell infusion; however, those with CNS relapse after Pola-BR ultimately experienced inferior outcomes, highlighting the challenge of CNS disease, even in a bridging settings.
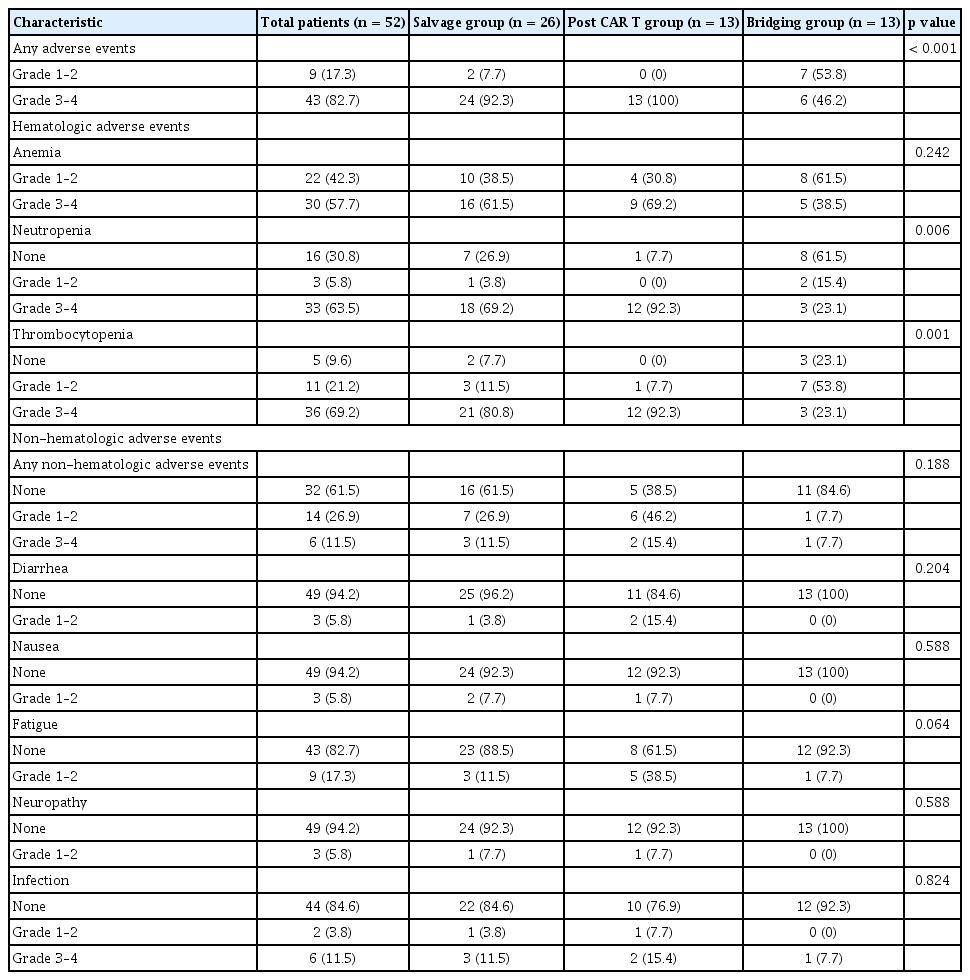
Treatment-related AEs
All patients experienced at least one AE, with most events (82.7%) classified as grade 3–4 (Table 2). The incidence of severe AEs was highest in the post-CAR T group (100%), followed by the salvage (92.3%) and bridging (46.2%) groups. Hematological AEs, including anemia, neutropenia, and thrombocytopenia, were also common. The incidences of grade 3–4 anemia were 61.5%, 69.2%, and 38.5% in the salvage, post-CAR T, and bridging groups, respectively (p = 0.242). Neutropenia was significantly more frequent in the post-CAR T group (92.3%) than in the salvage (69.2%) and bridging (23.1%) groups (p = 0.006). Similarly, thrombocytopenia was observed in 80.8%, 92.3%, and 23.1% of the salvage, post-CAR T, and bridging groups, respectively (p = 0.001). Non-hematological AEs were observed in all groups, with no statistically significant differences between the categories. However, grade 3–4 non-hematologic AEs such as severe fatigue and infections tended to occur more frequently in the post-CAR T and salvage groups. Despite these trends, the differences in most of the non-hematological AEs were not statistically significant.
DISCUSSION
This single-center retrospective study analyzed the data of 52 patients with R/R DLBCL who received Pola-BR in three distinct settings: salvage therapy, post-CAR T-cell failure, and bridging therapy prior to CAR T-cell infusion. The ORR was 51.9% and the CR rate was 36.5%, suggesting that Pola-BR can provide meaningful disease control in difficult-to-treat populations. These real-world outcomes are consistent with those reported in Western cohorts, in which ORRs ranged from 30% to 60% and CR rates were ≤ 50% [8–13]. Moreover, our outcomes fall between the higher ORRs but lower CR rates observed in Asian studies (ORR, ≤ 60–70%, CR, 15–30%) [14–16]. Thus, our results are broadly comparable across regions with only modest numerical differences. Such variations may be attributable to differences in patient characteristics (e.g., proportion of patients with refractory disease and prior ASCT), treatment timing (earlier vs. later line use), and small sample sizes, which limit statistical precision. Overall, although no major discrepancies were observed, our data provide additional evidence supporting the applicability of Pola-BR in Korean patients, a population that is often underrepresented in pivotal trials.
In the heavily pretreated salvage and post-CAR T-cell failure cohorts, the outcomes remained modest, with a median OS of only 3.8–5.2 months. These results are comparable to or slightly worse than, the 6.3 months median OS previously reported for R/R DLBCL patients treated with conventional chemotherapy before the immunotherapy era [1,17]. Cox regression analysis identified refractory disease as a consistent adverse prognostic factor, independently associated with inferior OS (HR, 5.80; 95% CI, 1.13–29.70; p = 0.035) and PFS (HR, 7.67; 95% CI 1.66–35.50; p = 0.009). Conversely, prior ASCT was associated with improved OS (HR 0.326, 95% CI, 0.115–0.921; p = 0.035). However, this finding should be interpreted with caution, as patients who undergo ASCT are typically a more favorable subgroup with better baseline fitness or disease biology, raising the possibility that a selection bias rather than a direct treatment effect explains the association [18]. Female sex was also associated with superior PFS (HR, 0.376; 95% CI, 0.163–0.871; p = 0.022), although this exploratory observation requires external validation. Collectively, these findings highlight the difficulty of achieving durable remission in multiple relapsed DLBCL and suggest that Pola-BR may serve as a tool primarily for temporary disease control rather than as a curative option when used as a salvage regimen.
In contrast, the bridging group achieved the highest ORR (61.5%) and CR rate (46.2%) despite receiving a median of only one cycle. This short-course treatment enabled 92.3% of the patients to successfully proceed to their scheduled CAR T-cell infusion, a rate somewhat higher than that reported in Western cohorts (70–85%) [10,12]. The ability to achieve rapid cytoreduction is clinically relevant because maintaining performance status and reducing tumor burden are essential prerequisites for CAR T-cell therapy [5,19,20]. Among the patients in the bridging group, those who achieved CR experienced longer post-CAR-T PFS than those without CR (7.0 vs. 2.4 mo, p = 0.010). However, given the small sample size, these observations should be regarded as exploratory rather than definitive. Overall, these findings suggest that the main value of Pola-BR in this context is to facilitate a successful transition to cellular therapy rather than to provide durable disease control as a monotherapy.
The timing of Pola-BR administration also appeared to influence the response outcomes. Patients who received Pola-BR after two or fewer prior lines of therapy achieved a substantially higher ORR (81.2%) and CR rate (56.2%) than those treated after three or more prior lines (ORR, 38.9%; CR, 27.8%). Although this association highlights the potential benefit of earlier use, it did not translate into improved OS in our cohort, as confirmed by Cox regression. Biologically, earlier administration may preserve the bone marrow reserve and reduce cumulative chemoresistance, thereby allowing Pola-BR to exert greater therapeutic activity [21–23].
CNS disease is a particularly challenging comorbidity in the management of R/R DLBCL, and our findings reaffirm the limited efficacy of Pola-BR in this setting. Patients with active CNS involvement at the time of treatment initiation showed only transient or minimal benefits, with rapid progression occurring despite therapy. These results are consistent with recent real-world data reporting CNS relapses despite the use of polatuzumab-containing regimens [24]. Although individuals with a history of CNS disease occasionally maintained systemic remission and stable CNS status, this was not consistent, and several patients experienced CNS relapse even after achieving a systemic response. Notably, relapse occurred in both the salvage and bridging groups, indicating that Pola-BR provided little protection against CNS recurrence. Among the bridging patients, CNS relapse was associated with inferior outcomes after CAR T-cell infusion, underscoring the persistent vulnerability of this subgroup. Collectively, these observations suggest that the principal value of Pola-BR lies in its systemic cytoreduction, although its impact on CNS activity is limited. Future studies incorporating dedicated CNS-directed strategies or novel agents with enhanced CNS penetrance are essential to improve outcomes in this high-risk subgroup.
The incidence of grades 3–4 cytopenias (anemia, neutropenia, and thrombocytopenia) differed markedly between the groups. Nearly all patients in the salvage (92.3%) and post-CAR T groups (100%) experienced severe hematological toxicity. This pattern likely reflects a combination of disease aggressiveness, cumulative marrow injury from multiple prior therapies, and intensive and prolonged administration of Pola-BR [25,26]. In contrast, the bridging group, treated earlier and with only one cycle, had a substantially lower rate of severe cytopenia (46.2%). This favorable safety profile is clinically relevant because maintaining patient performance status and hematopoietic function during the CAR T-cell manufacturing window is essential for treatment eligibility and better tolerance to eventual CAR T-cell toxicities. Therefore, our findings suggest that a single cycle of Pola-BR is a manageable and safe therapeutic option that can facilitate CAR T-cell therapy without compromising patient fitness.
This study had several limitations. The retrospective, single-center design and relatively small subgroup sizes limited the robustness of the survival analyses and the generalizability of the findings. Additionally, Pola-BR demonstrated limited efficacy in preventing or controlling CNS involvement; several patients experienced CNS relapse despite achieving systemic CR. This highlights the critical and unmet need for better strategies for managing CNS diseases in high-risk patients. Moreover, the cost of Pola-BR remains a major barrier, potentially limiting the number of cycles delivered in clinical practice, particularly in patients requiring intensive salvage therapy.
Prospective multicenter studies are needed to validate these findings, define the optimal timing and number of Pola-BR cycles, and directly compare their bridging efficacy and safety with those of other regimens. These studies should include a comprehensive evaluation of strategies to enhance CNS prophylaxis in high-risk patients. Furthermore, comprehensive cost-effectiveness analyses are essential to inform clinical guidelines and expand access to Pola-BR, particularly in healthcare systems with significant financial constraints.
In conclusion, our findings support Pola-BR as a clinically meaningful option for heavily pretreated R/R DLBCL, particularly as a bridging regimen for CAR T-cell therapy. By providing rapid cytoreduction with manageable toxicity, Pola-BR facilitates the successful transition to potentially curative cellular therapies, highlighting its practical value in contemporary treatment strategies.
KEY MESSAGE
1. Pola-BR provided meaningful but primarily temporary disease control in heavily pretreated R/R DLBCL, with its greatest utility as a bridging regimen enabling successful CAR T-cell therapy.
2. The timing and intensity of Pola-BR administration strongly influenced outcomes, with earlier use yielding higher response rates and limited-cycle bridging, thereby minimizing hematologic toxicity.
3. In heavily pretreated patients, Pola-BR offered limited survival benefits, underscoring its role as a cytoreductive option rather than a curative therapy.
Notes
CRedit authorship contributions
Changgon Kim: conceptualization, methodology, resources, investigation, data curation, formal analysis, validation, writing - original draft, writing - review & editing, visualization; Sang Eun Yoon: resources, investigation, data curation, formal analysis, validation; Hyun-Young Kim: resources, investigation, data curation, formal analysis, validation; Duck Cho: resources, investigation, data curation, formal analysis, validation; Junhun Cho: resources, investigation, data curation, formal analysis, validation; Won Seog Kim: resources, investigation, data curation, formal analysis, validation; Seok Jin Kim: conceptualization, methodology, resources, investigation, data curation, formal analysis, validation, writing - review & editing, visualization, supervision, project administration, funding acquisition
Conflicts of interest
The authors disclose no conflicts.
Funding
This work was supported by the Korea University Research Grant (No. O2514551).
Availability of data and materials
All data are available upon request from the corresponding authors.