Implementing antimicrobial stewardship: lessons and perspectives from a university-affiliated tertiary hospital in Korea
Article information
Abstract
Antimicrobial stewardship programs (ASPs) can lower antibiotic use, decrease medical expenses, prevent the emergence of resistant bacteria, and enhance treatment for infectious diseases. This study summarizes the stepwise implementation and effects of ASPs in a single university-affiliated tertiary care hospital in Korea; it also presents future directions and challenges in resource-limited settings. At the study hospital, the core elements of the ASP such as leadership commitment, accountability, and operating system were established in 2000, then strengthened by the formation of the Antimicrobial Stewardship (AMS) Team in 2018. The actions of ASPs entail key components including a computerized restrictive antibiotic prescription system, prospective audit, post-prescription review through quantitative and qualitative intervention, and pharmacy-based interventions to optimize antibiotic usage. The AMS Team regularly tracked antibiotic use, the effects of interventions, and the resistance patterns of pathogens in the hospital. The reporting system was enhanced and standardized by participation in the Korea National Antimicrobial Use Analysis System, and educational efforts are ongoing. Stepwise implementation of the ASP and the efforts of the AMS Team have led to a substantial reduction in the overall consumption of antibiotics, particularly regarding injectables, and optimization of antibiotic use. Our experience highlights the importance of leadership, accountability, institution-specific interventions, and the AMS Team.
INTRODUCTION
The inappropriate or unnecessary use of antibiotics constitutes a global threat that has led to increases in Clostridioides difficile infections, antibiotic resistance, and healthcare costs [1–4]. In an assessment of antibiotic use in U.S. medical institutions, 55.9% of cases were considered inappropriate, along with 33% of antibiotic prescriptions in tertiary hospitals [5,6]. In Korea, a nationwide assessment of antibiotic prescription adequacy, focusing on hospitals with > 500 beds, revealed an inappropriate prescription rate of 27.7% [7]. Furthermore, an evaluation of antibiotic prescriptions in Korean medical institutions with < 400 beds showed that the rate of inappropriate prescriptions was 30.6% [8]. In response, the Korean Ministry of Health and Welfare established a National Action Plan on Antimicrobial Resistance in 2016. This plan recognized the emergence of antibiotic-resistant bacteria as a threat at the level of emerging infectious diseases, and it highlighted the importance of antimicrobial stewardship programs (ASPs) within medical institutions.
An ASP comprises integrative interventions that promote optimal antibiotic use, including dosages, treatment durations, and routes of administration [9]. This approach has reduced antibiotic use, medical costs, and the incidence of C. difficile infection, along with mitigating antibiotic resistance in bacteria and contributing to effective treatment for infectious diseases [10–12]. There is a clear global emphasis on the importance of ASPs. A report from 2019 indicated that 88.9% of medical institutions in the United States had implemented ASPs [13]. The United Kingdom operates ASPs as part of its National Antibiotic Resistance Management Policy; it achieved a 7.3% reduction in the use of major antibiotics in humans from 2014 to 2017 [14]. In accordance with the National Antimicrobial Resistance Plan, Korean medical institutions have established ASPs, but the rate of antimicrobial use in Korea remained third-highest among Organisation for Economic Co-operation and Development (OECD) nations in 2020 [15].
The Catholic University of Korea, Seoul St. Mary’s Hospital (SSMH), is a university-affiliated tertiary hospital with 1,400 beds that performs approximately 500 hematologic stem cell transplantations annually. The precursor to this institution, Gangnam St. Mary’s Hospital, established the Antimicrobial Stewardship Committee (AMS Committee) in 2000; the activities of this committee are ongoing. Active ASP initiatives began in 2018, coinciding with the formation of the AMS Team under the AMS Committee. Here, we review the phased implementation of an ASP in a Korean tertiary hospital, highlighting core elements, presenting case studies, and discussing outcomes. Additionally, we describe future directions and challenges related to the implementation of ASPs in Korean medical institutions with limited resources.
STEPWISE IMPLEMENTATION OF CORE ELEMENTS AND OPTIMIZATION OF ANTIBIOTIC USE
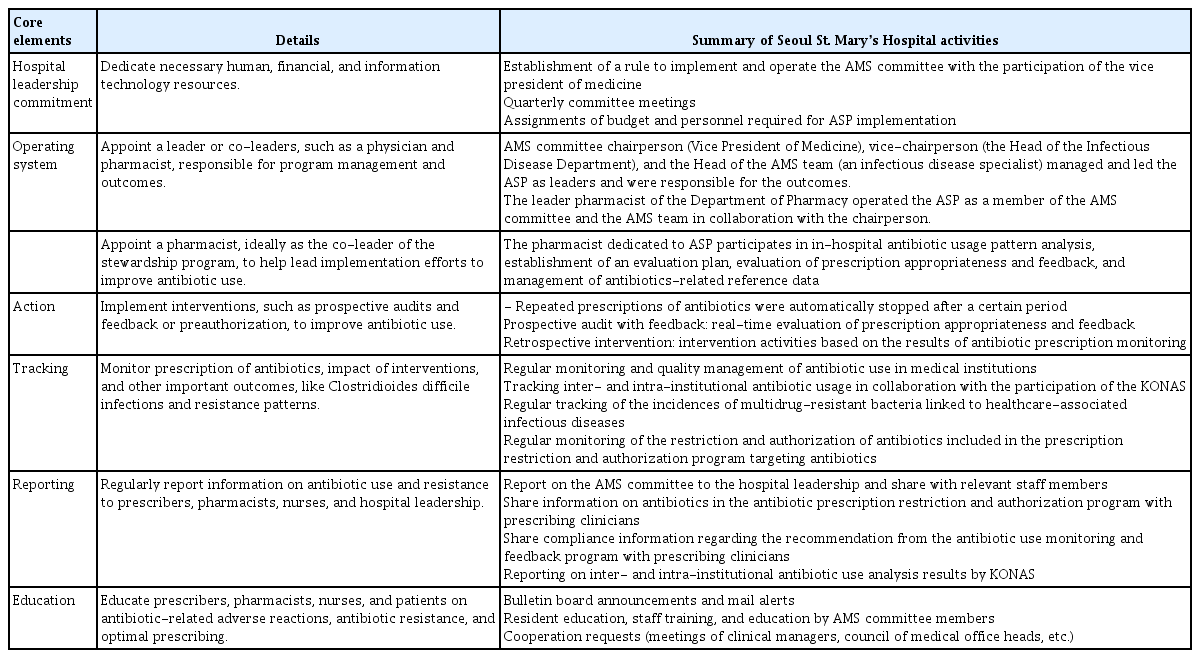
The core elements of an ASP—leadership commitment, operating system (running a dedicated multidisciplinary team that involves infectious disease specialists and pharmacists), action, tracking, reporting, and education—were systematically implemented in our hospital [16–18]. The following section outlines the implementation of these core elements (Table 1, Fig. 1).
The Catholic University of Korea, Seoul St. Mary’s Hospital activities corresponding to the core elements of the hospital antimicrobial stewardship program

Stepwise implementation of the antimicrobial stewardship program at The Catholic University of Korea, Seoul St. Mary’s Hospital. AMS, antimicrobial stewardship; CDSS, Clinical Decision Support System; KONAS, Korea National Antimicrobial Use Analysis System.
Hospital leadership commitment and operating system
In 2000, SSMH established the AMS Committee, which is currently led by the Vice President of Medicine and the Head of the Infectious Disease Department. The AMS Committee meets quarterly and comprises specialists in infectious disease, laboratory medicine, and other relevant disciplines; it collaborates with the Departments of Pharmacy and Nursing, as well as the Infection Prevention and Control Team, to formulate comprehensive ASPs. The AMS establishes policies and guidelines for proper antibiotic use, approves the use of restricted antibiotics, offers antibiotic-related consultations to other committees (such as advising the Pharmacy and Therapeutics Committee on the introduction of new antibiotics), appoints individuals responsible for antibiotic management in each department, and promotes ongoing activities (such as addressing practical challenges and proposing countermeasures).
A dedicated team of working personnel was needed to implement the antibiotic use policies and activities recommended by the AMS Committee. The AMS Team, established in the first quarter of 2018, initially was hindered by a shortage of dedicated personnel and limited leadership awareness of ASPs. By 2023, the AMS Team had evolved to include infectious disease specialists, pediatricians, and pharmacists, including a dedicated pharmacist (with a workload of 1.2 full-time equivalent [FTE], similar to the ASP infection specialist workload of 1.0 FTE) (Fig. 1).
The AMS Team actively conducted ASP activities by engaging in real-time communication. An infectious disease specialist assumed the leadership role in the AMS Team, chairing regular meetings, formulating improvement strategies based on restriction of antibiotic use, offering consultations to relevant committees and critical pathway (CP) teams, training clinicians, and proposing appropriate antibiotic use policies and guidelines. The ASP-dedicated pharmacist reviewed the appropriateness of antibiotic prescriptions, conducted therapeutic drug monitoring (TDM) of antimicrobials and antimicrobial-use-pattern analysis, provided medical information for antimicrobials and report writing for adverse drug reactions, and managed antimicrobial-related reference data.
Action
Computerized restrictive antibiotic prescription system
To ensure the appropriateness of antibiotic prescriptions, our hospital has implemented an antibiotic Clinical Decision Support System (CDSS) [19]. The antibiotic CDSS, a form of prospective antibiotic prescription intervention, provides antibiotic recommendations along with relevant information, based on the purpose of antibiotic use and the disease. It empowers physicians to select and prescribe antibiotics from the recommendations provided. Target drugs include injectable and oral antibiotics, antifungals, and antivirals. Among these, restricted antibiotics can only be prescribed through the antibiotic CDSS program. Upon identifying the prescription’s purpose (prophylaxis, empirical therapy, or targeted therapy) and selecting the corresponding infectious diseases, the program recommends hospital-owned antibiotics in line with established guidelines. In addition, the system imposes restrictions on selection beyond the pre-specified antibiotics. For targeted therapy, antibiotics are suggested based on the patient’s microbiological test results. As prophylactics, recommended antibiotics can be used for up to 7 days, compared with 14 days for empirical and targeted therapies. Subsequently, automatic cessation of repeated prescriptions is implemented.
Restricted antibiotics are defined as antibiotics with a high potential for resistance to develop as a result of widespread use; antibiotics with a high risk of adverse drug reactions; and antibiotics expected to require strict control (e.g., expensive or newly introduced broad-spectrum antibiotics). The decision to categorize an antibiotic as restricted is made by the AMS Committee or the Pharmacy and Therapeutics Committee. The prescription of restricted antibiotics requires prior authorization from an infectious disease specialist or a pediatrician. Authorization of the use of a restricted antibiotic is decided based on the indication, the reason for the request, the dosage of the antibiotic, and the duration of the prescription. As of October 2023, our hospital has listed as restricted 20 injectable antibiotic ingredients and 7 oral antibiotic ingredients (Supplementary Table 1).
Audit and feedback based on individual patients and overall antibiotic prescription pattern analysis
① Prescription review and intervention
The AMS Team monitors antibiotic prescriptions on a weekly basis, selects interventions at AMS Team meetings, and provides feedback. In 2018, an analysis of antibiotic use showed that moxifloxacin was being used as a prophylactic antibiotic during surgeries for chronic sinusitis, such as polypectomy, septoplasty, and turbinoplasty in otorhinolaryngology. Based on the recognition that moxifloxacin was inappropriate in this context, a targeted post-prescription review and intervention resulted in a decrease in the number of inappropriate prescriptions from 29 in June 2018 to 0 in August and September 2018.
② Continuous monitoring and feedback after quantitative evaluation of broad-spectrum antibiotic use
The AMS Team engaged in continuous monitoring and provided feedback on a quarterly basis, particularly in departments with a considerable increase in broad-spectrum antibiotic use. In the third quarter of 2021, an antibiotic-use-pattern analysis showed that the use of levofloxacin, meropenem, cefditoren, and cefepime had substantially and simultaneously increased, predominantly within certain departments. In response, several improvement initiatives were launched. The infection specialist and pharmacist on the AMS Team communicated the observed increases to the relevant departments, seeking explanations and proposing corrective actions. These actions comprised early cessation of antibiotic treatment, proactive de-escalation, and close collaboration with the Department of Infectious Disease. Consequently, there was a considerable decline in the use of certain antibiotics during the first quarter of 2022, particularly regarding meropenem (~45%).
Establishment of a target specific antibiotic restriction policy
An antibiotic-use-pattern analysis revealed the need to reduce the use of broad-spectrum antibiotics such as cefepime and carbapenem. The AMS Team, led by an infectious disease specialist, analyzed the hospital’s antibiotic protocol with a focus on these agents; this analysis identified overuse in empirical treatments, particularly among patients with hematologic malignancies [20]. Consequently, the AMS Team implemented ASP interventions, restricting the empirical use of cefepime to cases of neutropenia and limiting the empirical use of carbapenem to a third-line option for febrile neutropenia [21,22].
Provider-based intervention through regular reviews of prophylactic antibiotics for surgery
Considering that patients who undergo surgeries with ASP monitoring reportedly have reduced antibiotic use [23], an evaluation of the surgical CP protocol, including antibiotics, was conducted between 2018 and 2020; this evaluation led to 91 interventions. Beginning in 2020, the AMS Team instituted regular reviews when formulating or modifying CPs that involve antibiotics.
In 2023, the AMS Team expanded antibiotic guidelines for perioperative prophylaxis, covering surgeries outside of CPs. The recommendations were limits on third-generation cephalosporins, discouragement of vancomycin irrigation, completion of administration within 24 hours after surgery, and cessation of prescribed antibiotics at discharge. The AMS Team promoted compliance with the guidelines by engaging with non-compliant departments.
Pharmacy-based interventions through real-time review of antibiotic prescription appropriateness and administration
① Pharmacist-initiated interventions
The review of antibiotic prescription appropriateness was mainly conducted by the ASP-dedicated pharmacist; it included dosage according to indication, appropriateness of switching to a maintenance dose after a loading dose, ingredient/efficacy/spectrum overlap, equivalent doses according to formulation, drug interactions, and duration of administration for all inpatients. Relevant variables were extracted from electronic medical records using computer software.
The AMS Team also conducted interventions on administration by means of interdisciplinary discussions, as demonstrated by the standardization of administration timing for injectable antibiotics. Antibiotics with a time-dependent bactericidal effect require administration at regular intervals. Nevertheless, the actual administration time for injectable antibiotics often varies among patients, hindering compliance with the recommended administration time and leading to uncertainties about blood level-based dosing recommendations. Thus, the AMS Team promulgated recommendations focused on standard antibiotic administration times.
② Dose optimization
The AMS Team has engaged in activities designed to maximize treatment effects, such as guideline-based dosing, TDM of antibiotics, and use of dose-optimization software. Specifically, beginning in August 2020, the Team performed a pharmacokinetic consultation to change the criteria for TDM of vancomycin in patients with severe methicillin-resistant Staphylococcus aureus (MRSA) infections. The revision shifted the focus from the trough level to area under the curve, minimum inhibitory concentration, in accordance with the guidelines of the Infectious Diseases Society of America [24]. Additionally, the AMS Team conducted an intervention to establish the initial dose of vancomycin by screening patients with severe MRSA infections. Among patients with MRSA bacteremia, the intervention showed positive outcomes with a higher initial blood trough level in the therapeutic range, fewer days to the therapeutic range, and fewer days to negative conversion in the group that began with a loading dose and continued with a maintenance dose, compared with the usual dosing group. Based on these results, computer software was introduced in August 2023 to facilitate the calculation of loading and maintenance doses for vancomycin. Additionally, the AMS Team pharmacist recommended an optimized dose based on each patient’s renal function, using the pharmacy-based intervention computer software mentioned above.
Tracking
The ASP-dedicated pharmacist regularly conducted antibiotic-use-pattern analyses. Prior to 2023, the analyses were based on defined daily dose (DDD); after the first quarter of 2023, they were based on days of therapy to enable more precise evaluation of the duration of antibiotic use. The pharmacist analyzed the overall use of injectable and oral antibiotics, as well as trends in use according to class and ingredient, thereby identifying increases and decreases in antibiotic use. For antibiotics that showed a substantial increase in use compared with the previous quarter, the pharmacist reviewed prescription appropriateness for each department. Moreover, restricted antibiotics were monitored for use trends and for authorization/non-authorization.
The Infection Control Team systematically tracked the incidences of infectious diseases caused by multidrug-resistant pathogens such as MRSA, vancomycin-resistant Enterococci, multidrug-resistant Pseudomonas aeruginosa, multidrug-resistant Acinetobacter baumannii, and carbapenem-resistant Enterobacteriaceae. In accordance with established guidelines, these incidents were compared with the nationwide data for Korea. The Vice Chairperson of the AMS Committee also served as Head of the Infection Control Team and was continuously briefed on major matters. Furthermore, infection rates and antibiotic-use patterns were simultaneously reported and reviewed at meetings of the AMS Committee [18]. Additionally, the Team monitored surgical-site infection rates and compliance with prophylactic antibiotic administration, then compared those data with statistics from the Korean National Healthcare-associated
Infections Surveillance System [25,26]. Improvement activities for the above indicators were performed as necessary based on the monitoring results. Subsequently, outcomes before and after improvement activities were compared to evaluate their effectiveness. After modification of the vancomycin pharmacokinetic advisory criteria for patients with severe MRSA infections, the AMS Team has been monitoring pharmacokinetic advisory outcomes, along with related indicators such as the number of days to achieve negative conversion and the total duration of administration.
Reporting
The details of ASP activities, results from the Korea National Antimicrobial Use Analysis System (KONAS), and the outcomes of regularly monitored indicators have been systematically recorded in meeting minutes and reported to the president of the hospital. Furthermore, feedback (such as changes in antibiotic use and prescription errors) has been provided to relevant clinicians. To promote appropriate antibiotic use, our hospital has participated in the KONAS project since 2022, which involves monitoring and reporting of antibiotic use, trends, and inter- and intra-institutional comparisons based on the KONAS results [27].
Education
Education can promote appropriate antibiotic use, including reductions in prescription rates [28]. Accordingly, our hospital has conducted educational initiatives and sought cooperation from clinicians. Educational sessions covering fundamental antibiotic-use guidelines and ASP measures have been provided to physicians, including rotating residents and staff members twice annually. In cases where improvements were considered necessary, cooperation was solicited at meetings involving individuals such as clinical managers and heads of the council of medical offices. In particular, physicians of the Catholic Hematology Hospital have been educated on appropriate antibiotic use during each rotating resident shift (every 2 months), primarily because of their higher antibiotic use rates and burden of antibiotic-resistant bacteria. In contrast, nurses have undergone education related to antibiotic skin reaction tests and administration as part of their job training. The Department of Pharmacy has completed the standardization of guidelines for reviewing antibiotic prescription appropriateness; these standardized guidelines are used as educational materials for pharmacists.
IMPACT ASSESSMENT OF ANTIMICROBIAL STEWARDSHIP PROGRAMS
Achievements identified with quantitative evaluation of antibiotics
The hospital leadership’s commitment to ASP responsibilities, along with the appointment of the AMS team, enabled both quantitative (e.g., prospective interventions, automatic alerts on prolonged antibiotic use) and qualitative (e.g., reviews of broad-spectrum antibiotic appropriateness, tracking antibiotic usage, providing clinician feedback, and advanced TDM) evaluations. To assess the concrete accomplishments of ASPs at our hospital, we conducted an examination of the changes in antibiotic usage. The usage trend was examined by calculating DDD/1,000 patient-days based on antibiotic usage data by period (by quarter and year), followed by a comparison before and after the active ASPs intervention by the AMS team in 2018.
Overall antibiotic use trends over 10 years
Over the past decade, mean injectable antibiotic use was 668.8 DDD/1,000 patient-days. Notably, injectable antibiotic use increased until 2017 and substantially decreased after 2018, coinciding with establishment of the AMS Team and initiation of ASP activities (Fig. 2, Table 2, 3).

Annual trends in injectable and oral antibiotic use at The Catholic University of Korea, Seoul St. Mary’s Hospital (2013 to 2022). Arrow, period of organization by the Antimicrobial Stewardship Team; DDD, defined daily doses.

Injectable antibiotic use at The Catholic University of Korea, Seoul St. Mary’s Hospital over 10 years (2013 to 2022) (defined daily dose/1,000 patient-days)
Class-specific antibiotic use over a 10-year period
A class-specific antibiotic-use-pattern analysis enabled assessment of the effects of quantitative and qualitative interventions by the AMS Team. After the AMS Team limited the empirical use of broad-spectrum cephalosporins and carbapenems, the overall use of cephalosporins and carbapenems substantially decreased (Fig. 3A, B). Indeed, the use of cefepime decreased from 126.4 DDD/1,000 patient-days in the fourth quarter of 2017 to 60.2 DDD/1,000 patient-days in the first quarter of 2018.

Class-specific antibiotic use at The Catholic University of Korea, Seoul St. Mary’s Hospital over 10 years (2013 to 2022) (DDD/1,000 patient-days). (A) Injectable broad-spectrum antibiotics. a)Black arrow, reduction in Ceph(4) use. (B) Injectable antibiotics for multidrug-resistant pathogens. (C) Injectable non-broad-spectrum antibiotics and other antibiotics (clindamycin, colistin, linezolid, pentamidine, and daptomycin). b)Black arrow, reduction in Ceph(2) use and increase in Ceph(1) use. (D) Injectable non-broad-spectrum antibiotics-ii. (E) Oral broad-spectrum antibiotics. (F) Oral non-broad-spectrum antibiotics and others (clindamycin, fusidate, linezolid, rifaximin, fosfomycin, and nitrofurantoin). c)Black arrow, reduction in TC use. Ceph(1)/(2)/(3)/(4), first/second/third/fourth-generation cephalosporins; ePC, extended-spectrum penicillins; Q1/3, first/third quarter; DDD, defined daily doses; CB, carbapenems; GL, glycopeptides; AG, aminoglycosides; TC, tetracyclines; MC, macrolides; PC, penicillins; QN, quinolones.
The AMS Team reviewed the antibiotic appropriateness of CP, then restricted the unnecessary preoperative use of broad-spectrum prophylactic antibiotics and recommended first-generation cephalosporins in accordance with the guidelines. As a result, the use of second-generation cephalosporins decreased beginning in 2020, whereas the use of first-generation cephalosporins substantially increased (Fig. 3C). Although the use of flomoxef (frequently utilized as a preoperative prophylactic antibiotic) continuously decreased (60.4 DDD/1,000 patient-days in the fourth quarter of 2019 to 29.9 DDD/1,000 patient-days in the fourth quarter of 2020), the use of first-generation cefazolin increased (29.2 DDD/1,000 patient-days in the first quarter of 2020 to 44.9 DDD/1,000 patient-days in the third quarter of 2020).
Although the use of oral antibiotics did not substantially decrease compared with injectables (Fig. 2), ASP activities intended to limit the long-term use of broad-spectrum oral antibiotics via tracking and feedback resulted in a substantial reduction in the use of broad-spectrum antibiotics (Fig. 3E). The use of oral antibiotics not included in the intervention increased; this was caused by factors such as increased use of linezolid and fosfomycin (Fig. 3F).
Enhancement of antibiotic use
Continuous education of clinicians and other departments ensured the rapid adoption and effectiveness of the ASP. As a result of the intervention, coupled with advanced TDM for vancomycin, the program utilization rate increased to > 80% immediately after the introduction of a computerized loading dose.
In response to a suggestion to review patient eligibility for antibiotic skin testing and antibiotic concentrations for intradermal skin tests, a Task Force Team (TFT) was established. Based on discussions within the AMS Committee, the TFT comprised representatives from the Departments of Infectious Disease, Allergy Medicine, Pharmacy, and Nursing. After a review of the literature (including the guidelines of the Korean Academy of Asthma, Allergy and Clinical Immunology), investigation of status in other hospitals, legal review, and discussion, eligibility criteria for antibiotic skin testing were defined; guidelines were revised regarding drugs used in the skin reaction test (antihistamines, systemic corticosteroids, and imipramine), the intradermal test concentration of each drug, and the period of validity for the results. The revised guidelines, implemented in March 2022, have reduced the rate of unnecessary antibiotic skin tests and improved efficiency by enabling clear communication.
DISCUSSION AND PERSPECTIVES
We have summarized the stepwise adoption of an ASP and its status in a tertiary hospital in Korea. Increasing support from hospital leadership led to expanded authority and responsibilities for the AMS Committee. Consequently, a multidisciplinary AMS Team, led by an infectious disease specialist, was established; its members included a pediatrician and a dedicated pharmacist. ASP activities have expanded from computer-based interventions to quantitative and qualitative evaluations. To ensure the safe and effective use of antibiotics, the ASP-dedicated pharmacist has conducted real-time evaluations and given feedback regarding prescription appropriateness, regularly provided physicians with education, and requested cooperation from several departments.
Because quantitative and qualitative ASP interventions related to antibiotic prescription could reduce healthcare costs, hospitalization durations, and the incidences of bacterial resistance caused by antibiotics misuse and overuse, the importance of such interventions has been emphasized internationally and domestically [29,30]. Although medical institutions in Korea have made continuous efforts to implement ASPs, many have been hindered by limited medical resources [31–34]. In this context, we provide a summary of the accomplishments of and constraints faced by our hospital, which expanded ASP activities by gradually introducing intervention measures, even under conditions involving severe shortages of personnel and resources. We present our perspectives on the implementation of ASP activities in similar medical institutions worldwide.
ASP implementation requires a dedicated leadership and team. In our hospital, ASP awareness was cultivated among the hospital leadership by continuous reporting of ASP achievements. This elevated the status of the AMS Committee, the governing body responsible for ASP, thereby facilitating the expansion of ASP initiatives. The sustained involvement of hospital leadership laid the foundation for establishment of the AMS Team. When implementing an ASP, participation by hospital leadership (via continuous reporting and feedback) motivates the allocation of personnel and budget resources to the ASP; thus, the ASP becomes a top priority for the hospital [18].
The use of preauthorization in many institutions requires stepwise multidisciplinary implementation of ASP measures tailored to institutional needs. Korean medical institutions must introduce new antibiotics active against multidrug-resistant bacteria [35]. When introducing new antibiotics, the importance of the traditional antibiotic CDSS for restricted antibiotics will be further emphasized.
Nonetheless, it is important to tailor ASP interventions to institutional needs, with a focus on the operating team [36,37]. While our hospital implemented antibiotic computerized restrictive antibiotic prescription system from its inception, we observed no significant change in antibiotic use. In 2018, the AMS Team implemented further interventions, such as the establishment of institution-specific guidelines, monitoring, and feedback on antibiotic use for each department; restriction of unnecessary use of broad-spectrum antibiotics by prophylactic antibiotic interventions; and broad-spectrum antibiotic prescription intervention. These new interventions led to a substantial reduction in antibiotic use. Additionally, we initiated qualitative evaluations of long-term prescriptions of oral antibiotics and interventions to control the duration of injectable antibiotic administration based on each patient’s condition and indications. Moreover, biomarker-integrated ASP activities are in preparation in our hospital based on recent reports that a procalcitonin level-based ASP approach decreased antibiotic use [38–41].
The appointment of an ASP-dedicated pharmacist in 2023 could enhance the efficacies of ASP interventions. The interventions implemented in our hospital led to a substantial reduction in the use of broad-spectrum antibiotics, despite an increasing burden of resistant bacteria, distinguishing our hospital from comparable institutions in Korea [42].
An effective ASP requires interventions to be implemented based on qualitative evaluations [37]. Considering that Korean medical institutions lack medical resources, it is difficult to simultaneously implement qualitative evaluation-based ASP interventions. Qualitative evaluations and interventions based on the recently proposed quality indicators in Korea may be a sustainable alternative in such circumstances [36,37]. It is important to apply a stepwise implementation strategy—which should be led by a dedicated AMS Team—tailored to the institution. Indeed, quantitative evaluations should be combined with easy-to-implement interventions based on qualitative evaluations [43].
Continuous collaboration between clinical departments and support units, along with ongoing education, is crucial. ASP activities and interventions cannot be performed by the AMS Team alone. In our hospital, institution-specific guidelines were applied in collaboration with the Department of Hematology, which had the highest rate of broad-spectrum antibiotic use; this approach resulted in a substantial reduction in antibiotic use. Moreover, to enhance administration appropriateness, we revised guidelines for antibiotic skin testing and standardized antibiotic administration times in collaboration with the Pharmacy and Nursing Teams. Continuous education is also important; education and computerization of antibiotic loading doses resulted in an initial compliance rate of > 80% [44]. During the stepwise application of an ASP, it is important to involve other clinical departments and units as active (rather than passive) recipients by appointing individuals responsible for antibiotic management in each department. This involvement is crucial for efforts to track and report intervention outcomes, ensuring successful ASP implementation.
Clinical outcome tracking and reporting must be combined. ASPs are associated with decreases in antibiotic use, duration of administration, and duration of hospitalization [45]. However, few studies have focused on the relationship between an ASP and clinical outcomes, such as the incidence of antibiotic-resistant bacterial strains and changes in survival rates [46]. Although our hospital monitors the incidence of antibiotic-resistant bacterial strains, there has been insufficient analysis of its relationship with the ASP. Such data are also important to demonstrate the clinical need for the ASP, enhance understanding and interest among the leadership, and promote collaboration with other units relevant to the ASP. Beginning during the planning stages of the ASP, efforts should be made to monitor clinical outcomes.
Successful implementation of an ASP requires institutional efforts, as well as administrative and financial support. According to the antimicrobial stewardship staffing survey results reported by the KONAS in 2022, only four tertiary hospitals (7.0%) had staff dedicated to antimicrobial stewardship, with less than 1 FTE combined for the ASP infection specialist (0.69 FTE) and pharmacist (0.35 FTE). In terms of the ASP status of Korean medical institutions in 2020, the greatest barrier to ASP implementation and expansion was a lack of personnel, followed in order by a lack of tools, a lack of appropriate reward for operating an ASP, and a lack of education about ASPs [33]. Most medical institutions in Korea are facing a severe personnel shortage that hinders ASP implementation, and ASPs need structural and financial support [47]. However, fees for ASP implementation are not supported by the medical reimbursement system in Korea. The authors of a recent study suggested compensation through antimicrobial management fees to promote antimicrobial stewardship in Korean medical institutions [48], and there has been discussion about insurance coverage of ASPs. In 2018, the medical reimbursement system in Japan was revised to include fees for ASP implementation, which may have supported ASP activities [49]. Thus, it is important to build on the achievements of ASPs in Korea, which will promote recruitment of ASP staff and eventual reimbursement.
CONCLUSION
Our hospital has implemented ASP measures in a stepwise manner. Over the past decade, the results have included a considerable decrease in the overall use of injectable and oral antibiotics, including broad-spectrum antibiotics. To propagate the successful stepwise implementation model from SSMH to other institutions, leadership commitment and government support, including ASP fees will be needed. This is expected to limit the misuse and overuse of antibiotics, mitigate the emergence of antibiotic-resistant bacterial strains, reduce healthcare costs, and optimize therapeutic effects.
Acknowledgments
We acknowledge the members of the AMS Committee and the AMS Team for their unwavering dedication to ASPs in the hospital.
Notes
CRedit authorship contributions
Soo Jin Lee: conceptualization, methodology, data curation, formal analysis, writing - original draft; Raeseok Lee: conceptualization, methodology, investigation, data curation, formal analysis, writing - original draft, writing - review & editing; Sung-Yeon Cho: validation, writing - review & editing, supervision, project administration; Dukhee Nho: conceptualization, data curation, writing - original draft; Hye Lim Ahn: conceptualization, methodology, validation, writing - original draft, writing - review & editing, supervision; Dong-Gun Lee: conceptualization, methodology, validation, writing - review & editing, supervision, project administration
Conflicts of interest
The authors disclose no conflicts.
Funding
None

