Clinical Frailty Scale, K-FRAIL questionnaire, and clinical outcomes in an acute hospitalist unit in Korea
Article information
Abstract
Background/Aims
Frailty increases the risks of in-hospital adverse events such as delirium, falls, and functional decline in older adults. We assessed the feasibility and clinical relevance of frailty status in Korean older inpatients using the Clinical Frailty Scale (CFS) and Korean version of the Fatigue, Resistance, Ambulation, Illnesses, & Loss of Weight scale (K-FRAIL) questionnaires.
Methods
Frailty status was measured using the Korean-translated version of the CFS and K-FRAIL questionnaire within 3 days from admission in 144 consecutive patients aged 60 years or older. The correlation between CFS and K-FRAIL score was assessed. The criterion validity of CFS was assessed using receiver operating characteristic analysis. As outcomes, delirium, bedsore, length of stay (LOS), in-hospital mortality, and unplanned 30-day readmission were measured by reviewing medical records.
Results
The mean age of the study population was 70.1 years (range, 60 to 91), and 75 (52.1%) were men. By linear regression analysis, CFS and K-FRAIL were positively correlated (B = 0.72, p < 0.001). A CFS cutoff of ≥ 5 maximized sensitivity + specificity to classify frailty using K-FRAIL as a reference (C-index = 0.893). Higher frailty burden by both CFS and K-FRAIL was associated with higher LOS and bedsores. Unplanned readmission and in-hospital mortality were associated with higher CFS score but not with K-FRAIL score, after adjusting for age, gender, polypharmacy, and multimorbidity.
Conclusions
Frailty status by CFS was associated with LOS, bedsores, unplanned readmission, and in-hospital mortality. CFS can be used to screen high-risk patients who may benefit from geriatric interventions and discharge planning in acutely hospitalized older adults.
INTRODUCTION
Defined as a state of increased vulnerability to possible stressors with decreased physiological reserve associated with human aging, frailty is a common geriatric condition affecting up to > 20% of community-dwelling older adults in Korea [1,2]. Frailty increases the risks of developing adverse consequences including falls, functional decline, and mortality in community settings [3,4]. Frailty frequently accompanies multimorbidity and functional dependency and may deteriorate longitudinally in cycles of frailty [5]. However, frailty and associated geriatric conditions can be managed with appropriate intervention programs to prevent adverse outcomes such as functional decline even in resource-scarce public health settings [6,7].
Compared to young adults, older adults tend to have a larger burden of chronic diseases in addition to subclinical organ pathology that accumulates with aging [8]. Many older patients experience hospitalization for acute or chronic conditions [9]. In Korea, 16.8% of older adults aged 65 years or older reported at least one hospitalization in the past year in 2017 [10]. In older hospitalized patients, frailty was associated with increased in-hospital or subsequent mortality, higher length of stay (LOS), functional decline, delirium, and institutionalization [11,12]. To prevent these adverse outcomes associated with acute inpatient care of frail older adults, acute geriatric medicine services have been developed, with improved clinical outcomes compared to those for usual care [13].
Screening frailty might be an initial clinical step to identify high-risk older patients to receive these patients centered services [14]. Among numerous frailty screening tools, the Clinical Frailty Scale (CFS) is a quick measure using function-focused descriptions and pictures that has been validated in a wide range of care settings including nursing homes, emergency departments (EDs), acute wards, and intensive care units [15,16]. With its outcome prediction abilities, the CFS was also proposed as a guide to rationing scarce medical resources in the coronavirus disease 2019 (COVID-19) pandemic. However, to our knowledge, no studies have yet evaluated the feasibility and possible roles of the CFS in hospitalized older patients in Korea. Thus, this study assessed the feasibility and clinical relevance of frailty status in Korean older inpatients using the Korean-translated version of the CFS, with the Korean version of the Fatigue, Resistance, Ambulation, Illnesses, & Loss of Weight scale (K-FRAIL) questionnaire previously validated in Korean geriatric outpatients as a reference [17].
METHODS
Study design and participants
The internal medicine part of the hospital medicine center in Seoul National University Hospital, a tertiary teaching hospital, started frailty screening using the CFS and K-FRAIL in July 2020 as part of the process to identify candidate patients to receive inpatient medication reconciliation and patient-centered discharge planning services. A trained social worker measured the CFS, K-FRAIL within 72 hours of admission in patients aged 60 years older. We included patients admitted through both outpatient clinics and the ED. We excluded apparently terminal patients expected to die in fewer than 3 months due to cancer or uncontrolled underlying disease. For this study, we retrospectively reviewed the medical records of 144 patients admitted between July and November 2020, with available CFS and K-FRAIL scores.
Frailty assessments
We used the Korean-translated version of the CFS 2.0 (Supplementary Table 1), a screening measure with scores ranging from 1 (very fit) to 9 (terminally ill) that was previously validated with both frailty index and frailty phenotype in the Korean population and published elsewhere [18]. We considered CFS scores ≥ 5 and ≤ 4 to indicate frailty and non-frailty, respectively. We also used the K-FRAIL scale [17], a five-item questionnaire on fatigue, resistance, ambulation, illness, and loss of weight. We considered K-FRAIL scale scores with ≥ 3 positive items to indicate frailty. These measures were assessed by interviewing patients or their proxy, focusing on their health state before (i.e., 2 weeks before) the current acute clinical issue that resulted in the index hospitalization, by a single social worker (S.R.L.) throughout the study. Recorded CFS and K-FRAIL were cross-checked by a physician (S.J.H.).
Co-variables
As clinical co-variables, we recorded the vital signs on the day of frailty assessments. For comorbidities, histories of clinical diagnoses of angina, arthritis, asthma, cancer, chronic lung disease, congestive heart failure, diabetes, myocardial infarction, hypertension, chronic kidney disease, and stroke were assessed by medical record review. Multimorbidity was defined as the presence of two or more chronic diseases. The numbers of different medications the patients were taking at admission were recorded, with polypharmacy defined as taking five or more medications.
Outcome measures
As in-hospital complications, the incidence of clinically recorded delirium and bedsores either by physicians or nurses were reviewed. LOS was measured. Delirium, bedsore, in-hospital mortality, unplanned ED visits, and readmission within 30 days after discharge were assessed.
Statistical analyses
Continuous and categorical variables were expressed as mean ± standard deviation (SD) or numbers (%). To compare parameters across frail- and non-frail groups by CFS, we used t tests for continuous variables and chi-square tests for categorical variables. The correlations between age and CFS and K-FRAIL scale were evaluated by linear regression analysis to calculate the standardized beta (B) and were visualized using fractional polynomial regression analysis with 95% confidence intervals. The correlation between the CFS and K-FRAIL scale was also assessed by linear regression analysis. Receiver operating characteristic (ROC) analysis was performed for CFS, with frailty by K-FRAIL questionnaire as the reference. The sensitivity and specificity for individual cutoffs and C-index were also calculated. The associations between baseline frailty status and the incidence of clinical outcomes were evaluated using univariate and multivariate logistic regression analyses, with adjustment for age, gender, and multimorbidity in the multivariate model. Two-sided p values < 0.05 were considered statistically significant. The statistical analyses were performed using Stata version 15.0 (StataCorp., College Station, TX, USA).
Ethics statement
The study protocol was reviewed and approved by the Institutional Review Board of Seoul National University Hospital (H-2012-059-1179), which waived the need for informed consent due to the retrospective nature of the study.
RESULTS
Clinical characteristics
In the study population, the mean age was 70.1 years (range, 60 to 91; SD, 7.2) and 52.1% were men. The mean CFS score was 4.6 (range, 3 to 8; SD, 1.5) and 65 patients (45.1%) had a CFS score of ≥ 5. The mean K-FRAIL score was 1.7 (range, 0 to 5; SD, 1.4) and 45 patients (31.3%) were considered to be frail (K-FRAIL score ≥ 3). The distributions of CFS and K-FRAIL scores are shown in Fig. 1. The clinical parameters of the study population are shown in Table 1. When the population was grouped into frail (CFS ≥ 5) and non-frail (CFS ≤ 4) groups, the patients with frailty were older, were frailer by K-FRAIL scale, had low diastolic blood pressure at admission, experienced longer hospital stays, and were more likely to have bedsores and be re-admitted within 30 days after discharge.
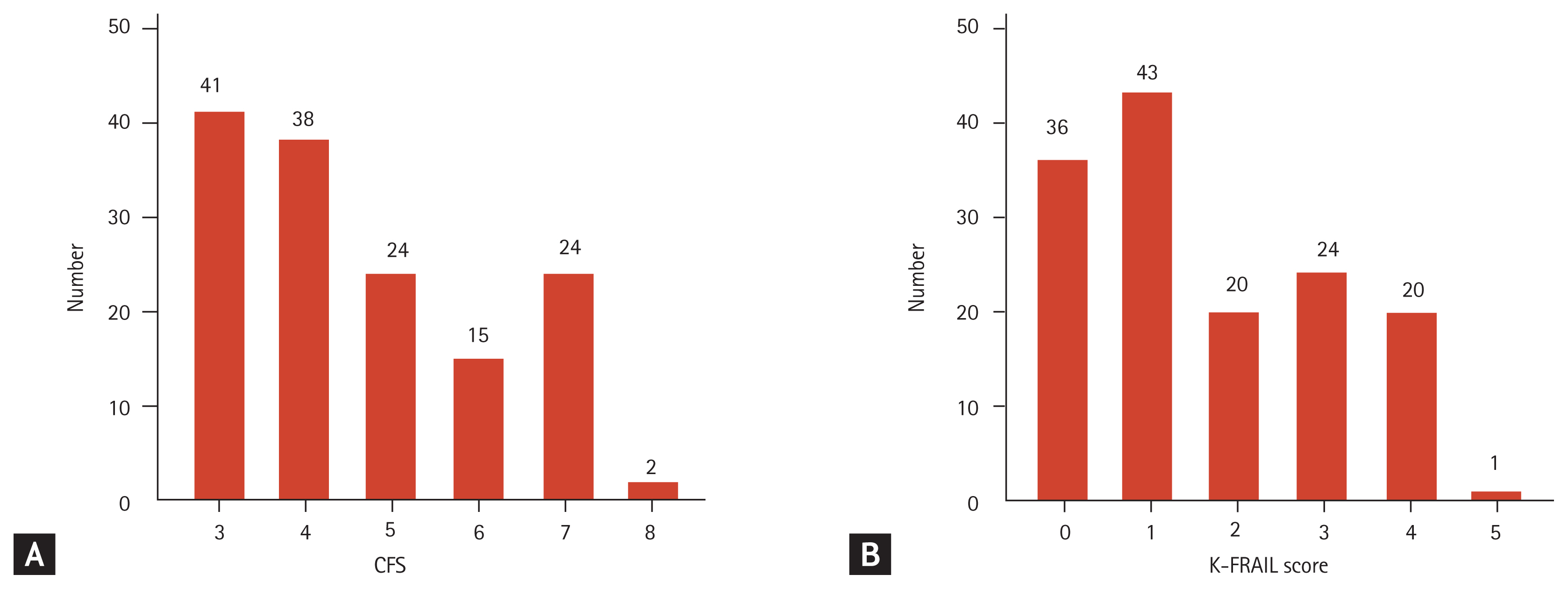
Distributions of (A) Clinical Frailty Scale (CFS) and (B) Korean version of the Fatigue, Resistance, Ambulation, Illnesses, & Loss of Weight scale (K-FRAIL) scores in the study population.
Content and criterion validity of CFS
The CFS and K-FRAIL were both significantly correlated with age (B = 0.35, R2 = 0.121, p < 0.001; and B = 0.25, R2 = 0.061, p < 0.001), respectively. The trends of the CFS and K-FRAIL scale and age are shown in Fig. 2. The CFS and K-FRAIL were correlated with each other (B = 0.72, p < 0.001, R2 = 0.525), as depicted in Fig. 3A.
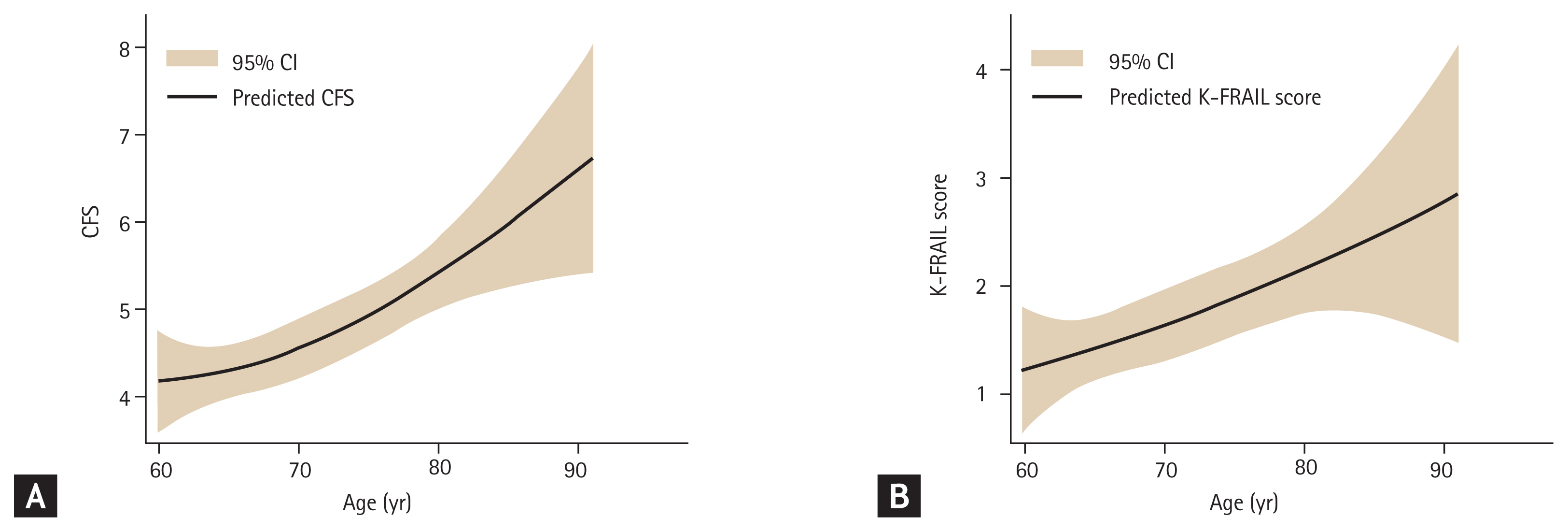
Trends of (A) Clinical Frailty Scale (CFS) and (B) Korean version of the Fatigue, Resistance, Ambulation, Illnesses, & Loss of Weight scale (K-FRAIL) scores by age, as mean (line) and 95% confidence interval (CI; shaded area) calculated by fractional polynomial regression analysis.
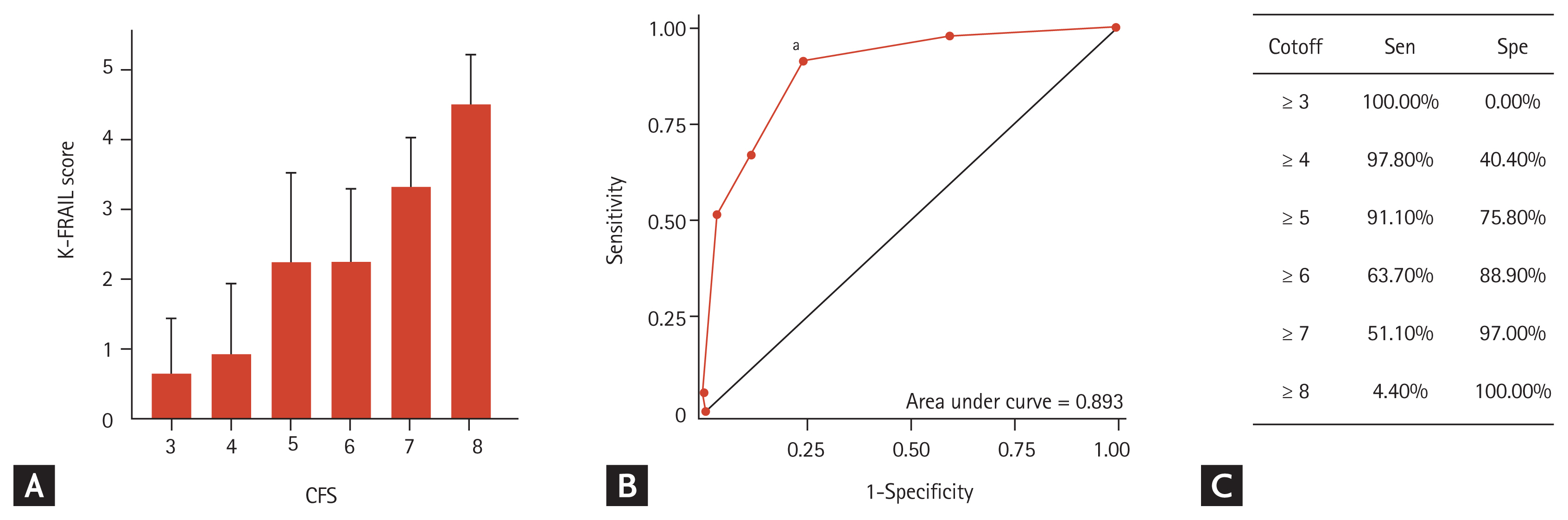
Means (bars) and standard deviations (whiskers) of the Korean version of the Fatigue, Resistance, Ambulation, Illnesses, & Loss of Weight scale (K-FRAIL) scores by corresponding Clinical Frailty Scale (CFS) scores (A), receiver operating characteristics (B) curve of CFS score with frailty by K-FRAIL (≥ 3) as the reference, and a sensitivity (Sen)/specificity (Spe) table for specific CFS scores to classify frailty by K-FRAIL (C). aCFS of 5 or higher maximized Sen + Spe in determining frailty by K-FRAIL.
The classification ability of the CFS was assessed using frailty by K-FRAIL scale as the reference. By ROC analysis (Fig. 3B), the C-index was 0.893 and a CFS cutoff of ≥ 5 maximized sensitivity + specificity (Fig. 3B and 3C).
Frailty status and clinical outcomes
The mean LOS was 10.4 days (SD, 12.2) in the study population. Higher frailty burden by both CFS (B = 0.47, p < 0.001) and K-FRAIL (B = 0.34, p < 0.001) was associated with longer LOS in age- and gender-adjusted multivariate linear regression analysis.
During hospitalization, 10 patients (6.9%) experienced delirium, 12 (8.3%) had bedsores, and four (2.8%) died. After discharge, 23 (16.8%) patients experienced unplanned ED visits and 14 (10.2%) experienced unplanned readmissions within 30 days. The presence of bedsores was associated with both CFS score and K-FRAIL, even after adjusting for age, gender, polypharmacy, and multimorbidity. In the univariate logistic analysis, unplanned readmission and ED visits were associated with higher CFS and K-FRAIL scores, respectively. When adjusted for age, gender, polypharmacy, and multimorbidity, the CFS remained significantly associated with unplanned readmission, while the association between K-FRAIL and ED visit became attenuated. In-hospital mortality was marginally associated with higher K-FRAIL scores in univariate logistic analysis, although this association was not significant after adjustment. In contrast, the CFS remained significant in predicting in-hospital mortality after adjustment, although this association was not significant in unadjusted analysis (Table 2).
DISCUSSION
In this study, we found that the CFS correlated with the K-FRAIL and could classify frailty status by K-FRAIL score, a tool previously validated in the Korean population in hospitalized older patients. A higher CFS score was associated with a longer hospital stay, bedsores, unplanned readmission, and in-hospital mortality even after adjusting for age, gender, polypharmacy, and multimorbidity. To our knowledge, this is the first study to report the clinical relevance of the CFS in hospitalized patients in Korea.
Our observations of the clinical outcomes with the CFS are consistent with those of previous studies performed in acute inpatients [12,16]. In the present study, the CFS predicted LOS and unplanned readmission after adjusting for age and gender, concordant with previous observations from other countries [19,20]. Although the associations of CFS and unplanned ED visits were attenuated after adjusting for age and gender, the general trends suggested a significant association of the CFS with outcomes in a larger population, as shown in a study in Canada [21]. While the CFS was associated with the detection of bedsores during hospitalization, outcomes such as deconditioning or subsequent institutionalization were not available in our study, although existing literature supports the use of the CFS to predict high-risk patients with functional decline [22].
The clinical importance of frailty as a risk-stratifying measure has been studied in patients with varying clinical conditions or in acute to chronic care settings, as well as community-dwelling older population [4]. The outcome prediction abilities of frailty in specialties entailing cardiology and cardiac surgery [23], general surgery [11], nephrology [24], oncology [25], hepatology [26], emergency medicine [27], and critical care [28] have been extensively reported, with specialties other than geriatrics now embracing frailty as an important parameter that deserves geriatric attention to improve clinical outcomes.
In Korean acute hospitals, however, assessments of frailty in inpatients are rarely performed except for a few hospitals with geriatric medicine services [29]. While measures focusing on older patients such as fall prevention, discharge planning, and social welfare services are available in some hospitals, case-finding and coordination are mostly performed ad hoc, without evaluation of frailty or other geriatric problems [29]. Consequently, many older, vulnerable patients are currently left with unmet needs in the medical and functional domains. Therefore, frailty screening measures that have been validated in acute hospital settings may help identify high-risk patients who may benefit from inpatient-focused services [14,30,31].
The results of the present study demonstrated that the CFS can be used as a quick screening measure of high-risk older patients in hospitalist units. Since hospital medicine provides global medical care and functional demands of hospitalized patients, adopting the concept of frailty may facilitate the coordination of various patient-centered programs including risk prevention measures for adverse outcomes such as falls, delirium, and bedsores [32]; translation care planning services involving pharmacists and social workers [33]; and care planning entailing advance directives and deprescribing [34]. With hospital medicine in Korea rapidly expanding to address the soaring demand for in-hospital care with population aging, adopting frailty-based care coordination may also alleviate the ever-increasing and unmet geriatric care needs [29].
Our study has several limitations. The study was performed in a single tertiary center, in which a high proportion of patients have cancers as key clinical problems [35,36], limiting the generalizability of our results to other acute or chronic care settings across Korea. Also, data on outcomes such as longer-term mortality and functional deterioration were not available in the current study design. Larger, longitudinally designed studies in hospitalized older patients with detailed functional measurements are warranted. We used the K-FRAIL, another screening tool, as a reference measure of frailty in this study, rather than the frailty index, since comprehensive geriatric assessment (CGA) was not available in the study setting. Consequently, a detailed analysis of the content and construct validities of CFS was not possible in our study. However, an ongoing study comparing the Korean version of the CFS to geriatric parameters using the CGA in another population may overcome this weakness of the present study. In this study, due to the retrospective nature, we had to rely on information on sore and delirium using medical records documented by physicians and nurses. Therefore, whether recorded sores were newly developed or already existed are not clearly assessible in the present analysis, thus limiting its value as outcomes. Also, without structured delirium screening or surveillance measures, deliriums could be under detected in the current retrospective study. Surveillance on geriatric outcomes such as delirium and sore using validated instruments in the future prospective study might be helpful to alleviate possible limitations of this study.
In conclusion, frailty status according to CFS score was associated with LOS, bedsores, unplanned readmission, and in-hospital mortality among inpatients of a hospitalist unit in a tertiary hospital. The CFS can be used to screen high-risk patients who may benefit from geriatric interventions and discharge planning in acutely hospitalized older adults.
KEY MESSAGE
1. The Korean-translated Clinical Frailty Scale (CFS) determined frailty status according to Korean version of the Fatigue, Resistance, Ambulation, Illnesses, & Loss of Weight scale (K-FRAIL) score in hospitalized older patients.
2. Worse baseline CFS was associated with longer length of stay, increased risk of in-hospital mortality, and unplanned 30-day readmission after discharge.
3. The CFS may be used to screen high-risk older patients in acute-care hospitals.
Notes
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This study was supported by grant No.0420202070 from the Seoul National University Hospital Research Fund and a grant from the Korea Health Technology R&D project through the Korea Health Industry Development Institute (KHIDI) funded by the Ministry of Health & Welfare, Republic of Korea (grant no. HI18C2383).


