Endoscopic Cryotherapy of Lung and Bronchial Tumors: A Systematic Review
Article information
Abstract
Background/Aims
We made a systematic review and evaluation of endoscopic cryotherapy of endobronchial tumors, investigating safety and efficacy.
Methods
Qualified studies regarding endoscopic cryotherapy of lung tumors were systemically evaluated using available databases according to predefined criteria.
Results
In total, 16 publications were included in the final assessment. A narrative synthesis was performed because a formal meta-analysis was not viable due to the lack of controlled studies and study heterogeneity. Overall success rates for significant recanalization of the obstruction were approximately 80%, although they varied, depending on disease status in the patient population. Complications from the procedure developed in 0-11.1% of cases, most of which were minor and controlled by conservative management. Although limited data were available on comprehensive functional assessment, some studies showed that respiratory symptoms, pulmonary function tests, and performance status were significantly improved.
Conclusions
Endoscopic cryotherapy was found to be a safe and useful procedure in the management of endobronchial tumors although its efficacy and appropriate indications have yet to be determined in well-designed controlled studies.
INTRODUCTION
Despite recent development of therapies and anti-cancer drugs, lung cancer does not yet respond well to treatments and continues to have a poor prognosis. Accordingly, it is ranked first in the world in terms of mortality among malignant tumors [1-3].
Two-thirds of all lung cancers are already in a stage where surgery is difficult by the time of diagnosis, and conservative treatments play an important role. In approximately 30% of lung cancer patients, central airway obstruction is accompanied by symptoms such as dyspnea and hemoptysis. Local treatment for these symptoms plays an important role in alleviating patient symptoms and improving their quality of life [4]. In progressive lung cancers, accompanied by central airway obstruction, various interventional methods for improving airway obstruction have been tried, including Nd:YAG laser therapy, electrocautery, brachytherapy, photodynamic therapy and cryotherapy [4,5]. Nd:YAG laser therapy, which has been widely used, and electrocauterization are effective and instantly open up the airway, but are more likely to result in such complications as airway perforation [5]. Also, photodynamic therapy is effective on small tumors, but is expensive, and may result in complications, including hemorrhage or secondary skin burns [5-7].
Cryotherapy uses flexible or rigid bronchoscopy to quickly freeze cells to -70℃, destroying them. Although it has a relatively long history, it has been used less than other treatment methods [5,8]. This is believed to be attributed to the fact that its effect is more delayed and its indication narrower than the more popular Nd:YAG laser therapy. Several case studies have shown that endoscopic cryotherapy is fairly effective and safe, but its objective efficacy has not been proven through large-scale controlled studies [9,10]. However, based on available research reports, endoscopic cryotherapy has a number of advantages: it is less expensive, and less likely to result in complications, such as perforation or hemorrhage. If it is used for appropriate indications, it is expected to establish itself as a useful interventional treatment method.
In this study, we systematically analyzed and evaluated research data on endoscopic cryotherapy, and discuss reference materials useful in the treatment of airway obstruction caused by tumors.
METHODS
Literature search strategy
The strategy to review the literature for 'endoscopic cryotherapy performed on lung and bronchial tumors' focused on studies that used the bronchoscope, and included studies that conducted a comparative analysis of laser therapy, electrocauterization, brachytherapy, stent insertion, and photodynamic therapy. After analysis, operation-related factors, such as safety, response, relapse, survival, and patient condition improvement factors, such as symptom improvement, pulmonary function, performance, quality of life, degree of bronchial obstruction, and oxygen saturation, were selected as major results.
Regarding 'endoscopic cryotherapy for lung and bronchial tumors,' eight domestic databases including KoreaMed and foreign databases like Ovid-Medline, EMBASE, CINAHL, and the Cochrane Library were used. The search strategy integrated 'lung neoplasm, bronchogenic carcinoma, bronchial neoplasm and tracheal neoplasia' and 'cryotherapy, cryosurgery, cryoablation, cryocoagulation, cryodestruction and cryoextraction.' In total, 664 documents in Korean and English were identified initially. Publications that were review articles, editorials, non-human experiments, preclinical studies, and documents containing abstracts only were excluded. In total, 648 documents, including 210 overlapping documents, were excluded, leaving 16 documents in the final evaluation. Each step, from literature review through application of selection criteria to data extraction, was carried out independently by a subcommittee and two evaluators. Scottish Intercollegiate Guidelines Network (SIGN) methodology was used to evaluate the quality of the literature, and levels of evidence and grades of recommendation were selected accordingly [11].
Study inclusion criteria
Studies on lung and bronchial tumor patients.
Studies on cryotherapy using bronchoscopy.
Studies in which more than one appropriate medical outcome was reported.
Study exclusion criteria
Non-human and pre-clinical studies.
Studies that were not original articles (non-systematic reviews, editorials, letters, opinion pieces).
Studies not published in Korean or English.
Cases where the effects of other therapies were mixed with that of cryotherapy.
Studies that published abstracts only.
Effectiveness assessment of cryotherapy
The effectiveness of endoscopic cryotherapy for lung and bronchial tumors was evaluated on the basis of discussions of the subcommittee in terms of operation-related factors and influences on the results of medical treatment. Operation-related factors included response, relapse, survival, and success rate, whereas influences on the results of medical treatment included improvement in clinical symptoms, pulmonary function, performance, quality of life, degree of bronchial obstruction, and the increase in oxygen saturation.
RESULTS
Literature search results
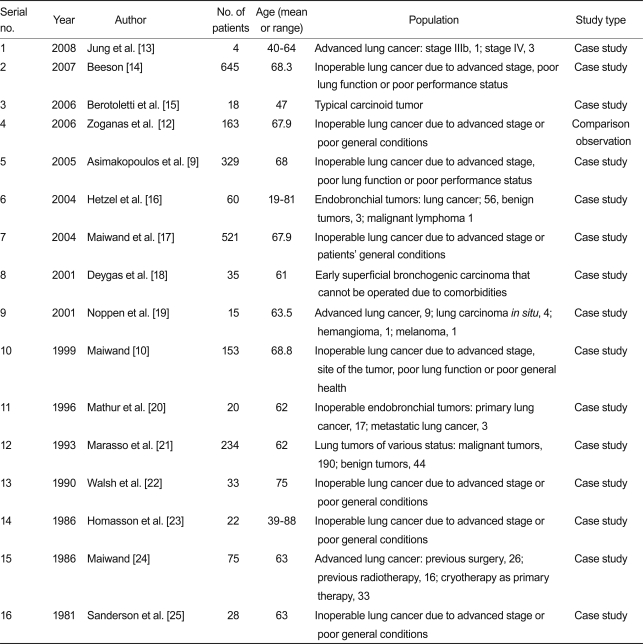
In total, one domestic report and 15 foreign reports were used for the evaluation. The safety and effectiveness of endoscopic cryotherapy for the lung and bronchus were discussed in all 16 documents. Of the 16 selected reports, one was a comparison observation study with evidence level 2 [12], and the remaining 15 were case studies [9,10,13-25]. General characteristics of the studies are presented in Table 1.
Clinical data and outcomes
Safety
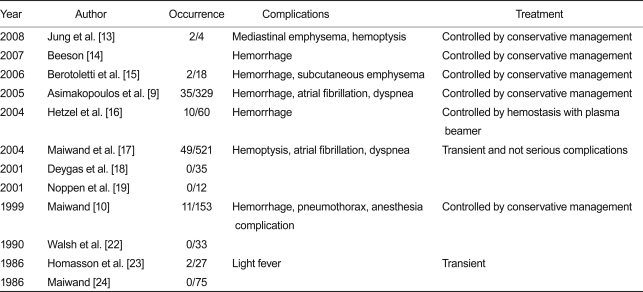
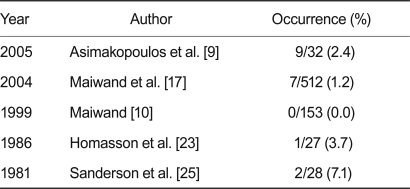
The safety of endoscopic cryotherapy for lung and bronchial tumors was evaluated on the basis of one comparison observation study, and 15 case studies with regard to deaths and complications within 30 days. Except for the small-scale study involving four subjects, complications, such as hemorrhage, mediastinal emphysema, atrial fibrillation, and dyspnea, occurred in 11.1% of all cases in 10 of the 16 studies. Most of the complications were controlled with simple conservative treatments (Table 2). In five of the 16 studies, mortality occurred in 7.1% of cases within 30 days of the operation. Causes of death were hemoptysis and respiratory failure. However, it was considered that most of the mortality was more likely to be associated with disease progression rather than a direct consequence of the procedures (Table 3).
Response rate
The case study by Hetzel et al. [16] evaluated the response rate of endobronchial tumors accompanied by a high level of stenosis after cryotherapy; the complete response rate was 61% (37/60), the partial response rate was 22% (13/60), and the total response rate was 83%. Deygas et al. [18] performed cryotherapy in treating 35 early superficial bronchogenic carcinoma patients who were inoperable due to comorbidities. The process was repeated 10-15 days later. They also followed 22 of the 35 patients; as a result, the complete response rate after 1 year was 91% (32/35). Homasson et al. [23] performed three cycles of -80℃ cryotherapy in 27 lung cancers including patients who underwent preoperative radiation treatment (n = 5), chemotherapy (n = 3), and chemoradiation therapy (n = 3). The procedure was repeated 4-6 days layer and the response rate was evaluated. The results showed that the response rate of malignant tumors was 61.9% (13/21), and that of benign tumors was 100% (5/5). In the case study by Jung et al. [13], endoscopic cryotherapy was performed on one stage IIIb, and three stage IV lung cancer patients, with a success rate of 75% (3/4). Noppen et al. [19] performed 3 × 20-s cycles of -80℃ cryotherapy in five invasive lung cancer patients, four carcinoma in situ (CIS) patients, two metastatic cancer patients, and one hemangioma patient. The process was repeated 1-2 weeks later, and the success rate was evaluated. Results showed the success rate to be 80% (4/5) in lung cancer, and zero in metastatic lung cancer. In all cases where the operation needed to be repeated, the second operation was a success. In the case study by Beeson [14] -70℃ cryotherapy was performed on 645 patients who were inoperable for various reasons. They reported that the tumor in the bronchus was reduced, and the airway was opened in most cases. Walsh et al. [22] performed three cycles of -70℃ cryotherapy for 33 inoperable patients, and evaluated the degree of bronchial obstruction. The degree of bronchial obstruction improved in 77% (20/26) of the patients, and, radiologically, atelectasis improved in 24% (7/24) of the patients.
Relapse rate
Hetzel et al. [16] reported a relapse rate of 24.6% (14/57) 10-24 weeks after cryotherapy in endobronchial tumors with obstruction. Deygas et al. [18] performed two successive sessions of cryotherapy for 35 superficial endobronchial tumors, and the relapse rate was 28% (10/35) after 13-45 months. According to the case study by Berotoletti et al. [15], three cycles of -70℃ cryotherapy was performed for 18 typical carcinoid tumor patients. They were followed up for 44.5 months, and there was no relapse. However, after 7 years, two of the patients had relapsed (11.1%).
Survival rate
In the case study by Beeson [14], -70℃ cryotherapy was performed on 645 patients who were inoperable. Among them, squamous cell carcinoma patients accounted for 68.3%, adenocarcinoma 15.2%, large cell carcinoma 2.6%, undifferentiated carcinoma 5.2%, and small cell carcinoma 8.7%. Patients in stage II accounted for 6.7%, stage IIIa 21.0%, IIIb 23.9%, and stage IV 48.4%. This case study did not present comprehensive data on survival rates, but suggested the possibility of an increase in the survival rate due to the procedure. In the case study by Zoganas et al. [12], cryotherapy was performed for inoperable cancer patients, and the survival rate was analyzed 2 years later. The result showed that the survival rate was 19.3% in the group to which only cryotherapy was performed, and 25% in the group to which anticancer treatment was performed concurrently, but the difference was not statistically significant (p = 0.388).
In the case study by Asimakopoulos et al. [9], cryotherapy was performed more than twice for advanced lung cancer patients. This group survived for 15 months, on average, whereas the other group for whom cryotherapy was performed only once survived for 8.3 months, on average. In the case study by Hetzel et al. [16], cryotherapy was performed on patients with protruding tumors accompanied with a high level of stenosis, and their survival rate after 36 weeks was 52.6% (30/57). Maiwand and Asimakopoulos [17] performed two sessions of cryotherapy for 521 cancer patients who were inoperable, and observed them for 18 months (4 to 84 months). Average survival was 8.2 months, and according to the stage, the average survival period of the patients was 15.1 months in stage IIb, 8.5 months in stage IIIa, 9.0 months in stage IIIb, and 6.6 months in stage IV. Also, the 1-year survival rate was 38.4%, while the 2-year survival rate was 15.9%. Deygas et al. [18] followed up 22 of 35 early superficial cancer patients, and the 2-year survival rate was 62.5% (20/32), and after 48-89 months the survival rate was 50% (11/22). Six of the 19 died from other causes, six died of a relapse in the same location, and the remaining seven died of metastasis in other locations. Maiwand [24] performed two cycles of -70℃ cryotherapy on 75 lung cancer patients with endobronchial tumors, who had undergone pneumonectomy (n = 18), preoperative radiation treatment (n = 16), and pneumonectomy and radiation treatment (n = 8), repeated for 2, 4, and 8 weeks, and followed them for 12 months. Results showed that the survival period of 19 patients was less than 1 month, 35 patients survived for 1-5 months, six patients survived for 6-11 months, and the remaining 16 patients survived for 12 months or longer.
Improvement in clinical symptoms and pulmonary function
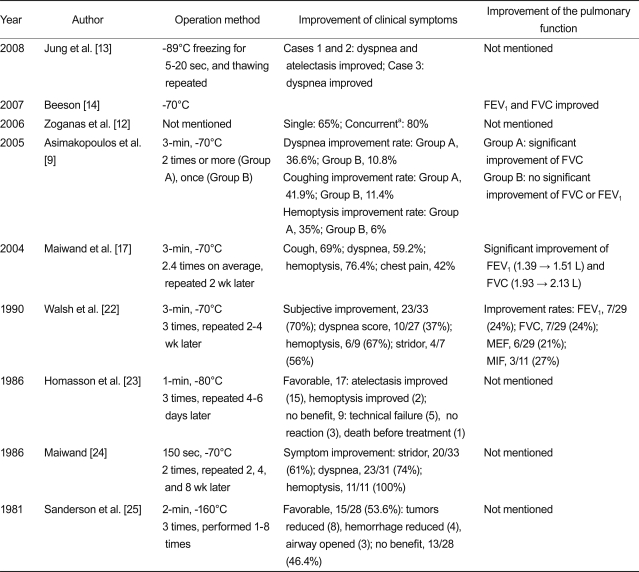
In most studies, the procedure was performed for advanced lung and bronchial cancer patients who were inoperable. Following the procedure, symptoms, such as dyspnea, cough, hemoptysis, and stridor, showed statistically significant improvements. This was also the case for pulmonary function; forced expiratory volume at 1 second (FEV1), forced vital capacity (FVC), and maximal flow rates showed statistically significant improvements after cryotherapy. The results of the studies are summarized in Table 4. Walsh et al. [22] reported that oxygen saturation (SaO2) improved in 77% (20/26) of patients.
Improvement in performance and quality of life
In the study by Maiwand and Asimakopoulos [17], performance status and quality of life were assessed before and after cryotherapy in 521 inoperable cancer patients. Results showed that the Karnofsky score improved significantly, from 60 to 75, and the WHO score was reduced significantly, from 3.04 to 2.20. Walsh et al. [22] reported that in 33 inoperable patients, the performance of 27% (6/22) improved in the 6-minutes walking test. In the case study by Asimakopoulos et al. [9], cryotherapy was performed twice or more for Group A of advanced cancer patients, and once for Group B. The Karnofsky scores of Group A were 67.7, 72.2, and 74.6 before cryotherapy, after the first cryotherapy and after the second cryotherapy, respectively, whereas the Karnofsky scores in Group B was 67.5, 74.6, and 73.6, respectively. Both groups showed statistically significant improvements.
DISCUSSION
We have discussed the available safety and efficacy data of cryotherapy in the treatment of the endobronchial tumors, although detailed and comprehensive analysis was not possible due to the variability of methodologies and lack of standardization of the procedures. According to this study, endoscopic cryotherapy generally showed high treatment efficiency in approximately 80% of cases, although there was variation depending on operation methods or target patient groups. It was also effective in improving quality of life, improving symptoms, like dyspnea, and improving pulmonary function. In lung tumors, airway obstruction is one of the symptoms seen in patients, but it has an important influence on quality of life, so appropriate treatment is required on many occasions, and although it may be difficult to expect that such local treatment will ultimately improve the survival rate, it will play an important role in patient management and conservative treatment. Laser therapy has been the preferred treatment for airway obstruction due to tumors. This is perhaps because laser therapy can instantly and powerfully open up even severe obstructions [26-28]. Laser therapy is known to result in more complications, including hemorrhage and perforation. Cryotherapy can be said to be superior in terms of safety [5]. Photodynamic therapy is also a useful local treatment method for various purposes, including curative treatment of an early cancer in the airway or alleviation of airway obstruction, but it is relatively expensive, and pre-and post-operation management is cumbersome [29]. Other endobronchial local treatment methods, including brachytherapy and electric cauterization, are used, and these methods have their own strengths and weaknesses. Accordingly, several factors, such as the status of the patient's disease, experience of the practitioner, and financial considerations, must be taken into account when any of these methods are selected.
As described previously, endoscopic cryotherapy may still be less researched and used than other treatment methods, including laser therapy. Because cryotherapy does not affect the cartilage or collagen in the bronchus, there is almost no risk of perforation, and, in this sense, it is safer than other methods, but it has less destructive power and its effect is delayed. Thus, its efficacy may be considered to be limited in cases with severe airway obstruction due to a large and extensive tumor [5]. As the spectrum of cryotherapy is about 5 mm, it is limited, and though it is possible to overcome this with repetition, there are other limitations. As laser therapy is more effective in opening up severe obstructions, it may be inappropriate to select cryotherapy as an initial treatment in such a case. It is recommended to perform laser therapy or cauterization first, and then cryotherapy as an additional treatment for the remaining tumor. It can also be a useful treatment method in removing early tumors limited to the airway [5]. As reports on the supplementary efficacy and synergistic effect of using cryotherapy together with anticancer chemotherapy or radiation treatment are published, the possibility of using it as part of concurrent treatments is also suggested [30,31].
Limitations of the analysis in this study relate to cryotherapy not being standardized; individual studies differed in research methods and frequencies, and the characteristics of patient groups varied. Thus, comprehensive statistical analysis was difficult. Accordingly, comprehensive statistical analysis was avoided in the results section, and we focused on describing the characteristics and results of each study. Additionally, both rigid and flexible bronchoscopy can be used for cryotherapy; depending on the choice, efficacy can vary. Because there are many different types of probe, there are slight differences in the mechanism of action and efficacy. Thus, there are limitations in comprehensively analyzing multiple studies [5].
In conclusion, endoscopic cryotherapy performed for endobronchial tumors is a safe and effective treatment method that will improve the symptoms, pulmonary function, and performance in patients with endobronchial obstruction, especially in inoperable cases. Given the lack of well-designed studies comparing it with other treatment methods, it must be objectively compared with other treatment methods and more cases need to be analyzed in future studies.
Acknowledgements
This study was supported by 2008 grant from the Ministry for Health, Welfare and Family Affairs' research fund.
Notes
No potential conflict of interest relevant to this article was reported.