Clinical Aspects of Pregnancy and Delivery in Patients with Chronic Idiopathic Thrombocytopenic Purpura (ITP)
Article information
Abstract
Background
Idiopathic thrombocytopenic purpura (ITP) is a condition that often develops in young women and, consequently, physicians should frequently manage and monitor pregnant patients with this disorder.
Methods
We reviewed the charts of 30 women with chronic ITP delivered in 31 pregnancies from January 1995 to December 2003.
Results
Fifteen patients were diagnosed with ITP before pregnancy and sixteen patients were diagnosed during pregnancy. The mean platelet counts before pregnancy, during pregnancy, and at delivery were 70,040/mm3, 83,960/mm3, and 62,680/mm3, respectively. The symptoms of hemostatic impairment were not noted in most of the pregnancies (77%, 24/31). During pregnancy and at delivery, most of the women (61%, 19/31) received various kinds of treatment to raise platelet counts. At delivery, the most commonly used therapy was platelet transfusion (48.4%, 15/31). Seven pregnancies (22.6%) were treated with corticosteroids during pregnancy and at delivery. Five pregnancies (16.1%) were treated with IV IgG during pregnancy and at delivery. Fifteen deliveries (51.7%) were performed by cesarean section and fourteen (48.3%) with vaginal delivery. Bleeding was uncommon at delivery. There were no cases of infants with any clinical signs of hemorrhage.
Conclusion
Our current results suggest that ITP in pregnancy can proceed safely with low hemorrhagic risk in both infants and mothers, and that mothers with ITP can deliver healthy infants without serious hemorrhagic complications
INTRODUCTION
Idiopathic thrombocytopenic purpura (ITP) is an autoimmune disorder characterized by a low platelet count and mucocutaneous bleeding. The estimated incidence is 100 cases per 1 million persons per year1). ITP in adults is generally chronic, the onset is often insidious, and approximately twice as many women as men are affected. It is estimated that ITP occurs in 1 or 2 cases out of every 1,000 pregnancies, accounting for approximately 3 percent of women who have thrombocytopenia at the time of delivery2). Although pregnancy is not discouraged in women with preexisting ITP, maternal and fetal complications can occur, and additional monitoring and therapy may be needed3).
In the past decade, our understanding of ITP in pregnancy has improved. However, most of the previous studies have focused on the outcomes of the infants. There has been limited information regarding ITP outcomes in obstetric patients, including hemostatic risk in delivery and the likely necessity of treatment during pregnancy. For this reason, we reviewed the outcomes and clinical aspects of pregnant patients with ITP.
MATERIALS AND METHODS
Data collection and patient selection
A retrospective chart review was performed for all obstetric patients with ITP who were treated and delivered at Hanyang University Medical Center from January 1995 to December 2003. We observed a total of 31 pregnancies in 30 patients. Patients were eligible for the study if they met the inclusion criteria of pregnancy with a diagnosis of ITP or pregnancy with a previous history of ITP. In each case, the diagnosis of chronic ITP had been established based on standard criteria: thrombocytopenia for >6 months associated with normal white and red blood cells, and exclusion of other known causes of thrombocytopenia. Other causes of thrombocytopenia included sepsis, pregnancy-induced hypertension, disseminated intravascular coagulation, drug-induced thrombocytopenia, and thrombocytopenia associated with autoimmune diseases such as systemic lupus erythematosus, thrombotic thrombocytopenic purpura, hemolytic uremic syndrome, and hereditary forms of thrombocytopenia, as well as gestational thrombocytopenia (also referred to as incidental or benign thrombocytopenia of pregnancy)4), which accounts for approximately 74 percent of cases of thrombocytopenia at full-term in healthy women who have had an uneventful pregnancy. Characteristic features include mild thrombocytopenia and normalization of platelet counts within 6 weeks after delivery5).
The following information was obtained from the chart of each eligible patient: age, date of ITP diagnosis, underlying medical conditions, medications, platelet count before and during pregnancy and at delivery (before pregnancy: before diagnosis of pregnancy; during pregnancy: from diagnosis of pregnancy to delivery 1 week ago; at delivery: from delivery 1 week ago to the time of delivery), signs and symptoms of hemostatic impairment during pregnancy, treatment received to raise platelet count during pregnancy or at delivery, gestational age at delivery, type of delivery, estimated blood loss at delivery, blood products transfused, and complications at delivery and in the postpartum period. Information collected for each infant included platelet count at birth, complications at birth, and treatment received.
Statistics
All data were analyzed using the SPSS version11.5 for Windows. Most of the data were not normally distributed. Therefore, nonparametric descriptives and tests were used. The Mann-Whitney test was performed to compare the mean platelet counts. Statistical significance was set at p<0.05.
RESULTS
Patient characteristics and clinical manifestation
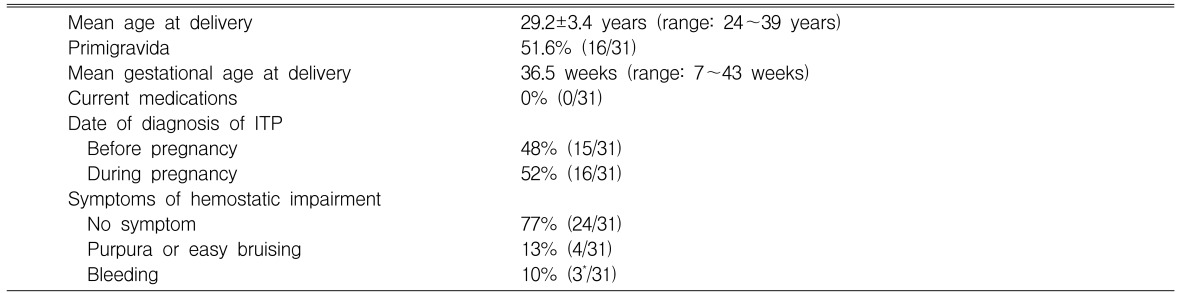
During the study interval, 30 women with ITP delivered 29 children in 31 pregnancies. The mean age of the women at delivery was 29.2 years (range: 24~39 years) and primigravida was 51.6% (16/31). The mean gestational age at delivery was 36.5 weeks (range: 7~43 weeks). All women were not currently using any medications. Of the cases, fifteen (48%) were diagnosed as ITP prior to pregnancy and sixteen (52%) during pregnancy. Two of the 31 pregnancies were complicated by missed abortion and intrauterine growth restriction. The symptoms of hemostatic impairment were not noted in most pregnancies (77%, 24/31). Among the remaining seven pregnancies, four pregnancies (13%) developed purpura and easy bruising. Three pregnancies (10%) had bleeding symptoms such as epistaxis, vaginal bleeding, or gastric ulcer bleeding (Table 1).
Maternal platelet counts
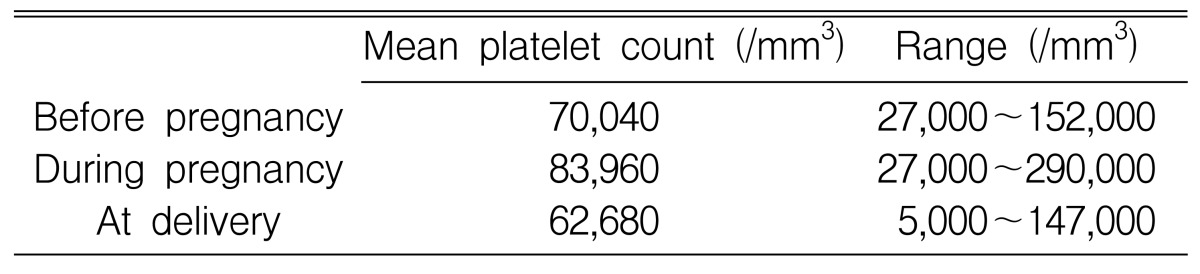
The mean platelet counts before pregnancy, during pregnancy, and at delivery were 70,040/mm3, 83,960/mm3, and 62,680/mm3, respectively. These results show that platelet counts tend to fall in the third trimester (Table 2). At delivery, two women (6.5%) had platelet counts of less than 20,000/mm3 and 9 women (29.0%) had platelet counts between 20,000/mm3 and 49,000/mm3. Women in 17 pregnancies (54.8%) had platelet counts from 50,000/mm3 to 100,000/mm3 and women in 3 pregnancies (9.7%) had platelet counts higher than 100,000/mm3 at delivery (Table 3). One woman with a platelet count lower than 20,000/mm3 at delivery experienced complication with gastric ulcer bleeding. The patient died of acute pulmonary edema after cesarean section. Another woman had no hemorrhagic symptoms during pregnancy or at delivery.
Medical intervention to raise platelet counts and type of delivery
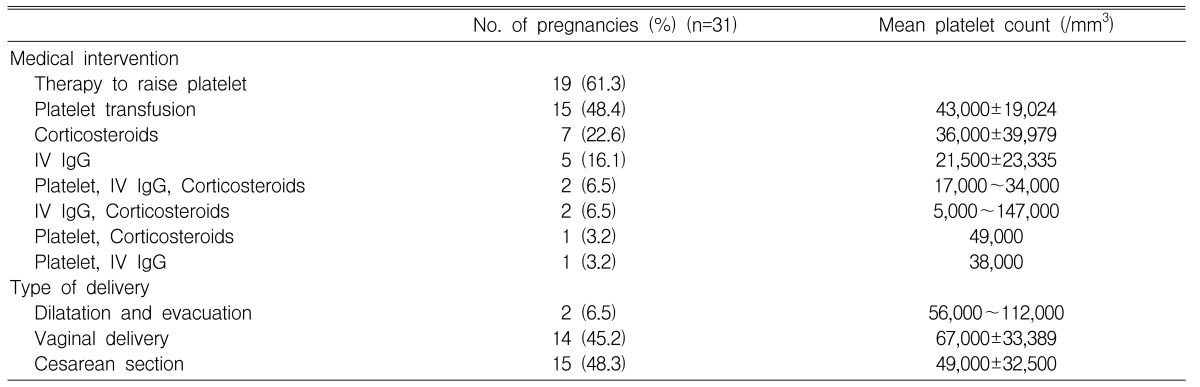
During pregnancy, most of the women (61.3%, 19/31) received therapy to raise platelet counts. At delivery, the most commonly used therapy was platelet transfusion. In the case of 15 pregnancies (48.4%), the women were treated with platelet transfusions. The mean number of transfused platelet concentrates was 9 units. The mean platelet counts of women receiving platelet transfusions at delivery were 43,000/mm3 (range: 28,000~76,000/mm3) compared with 73,000/mm3 (range: 5,000~140,000/mm3) in the group without transfusion. These differences were statistically significant (p=0.02). In seven pregnancies (22.6%), the women were treated with corticosteroids during pregnancy and at delivery. The mean platelet counts of women who received corticosteroids during pregnancy and at delivery were 78,500/mm3 (range: 40,000~121,000/mm3) and 36,000/mm3 (range: 5,000~97,000/mm3), respectively. An initial dose of 1 mg/Kg (based on pre-pregnancy weight) of prednisone was administered, and was subsequently tapered to the minimum hemostatically effective dose. Five pregnancies (16.1%) were treated with IV IgG during pregnancy and at delivery. The dose of IV IgG was 1g/Kg (pre-pregnancy weight) for 2 days. The mean platelet counts of women receiving IV IgG during pregnancy and at delivery were 74,000/mm3 (range: 27,000~121,000/mm3) and 21,500/mm3 (range: 5,000~38,000/mm3), respectively (Table 4).
Two of 31 pregnancies were terminated by dilatation and evacuation. One patient underwent dilatation and evacuation at 7 weeks gestation due to missed abortion. Pregnancy in the other patient was terminated due to intrauterine death of unknown origin and intrauterine growth restriction at 16 weeks gestation. There were no hemorrhagic complications during the termination procedures. Fifteen of 29 deliveries (51.7%) were performed by cesarean section, and fourteen (48.3%) with vaginal delivery. The mean platelet counts of the women who delivered vaginally and by cesarean section were 67,000/mm3 (range: 28,000~140,000/mm3) and 49,000/mm3 (range: 5,000~97,000/mm3), respectively. There was no statistically significant difference in platelet count between the two groups (p=0.85).
Most deliveries (90%) were successfully completed without hemorrhagic complications. Although two vaginal deliveries (7%) were complicated by third degree laceration, this complication was not attributed to the underlying ITP. One woman, who had gastric ulcer bleeding with a very low platelet count < 20,000/mm3, died of acute pulmonary edema after cesarean section. Another woman with a platelet count lower than 20,000/mm3 at delivery was treated with platelet transfusion (platelet concentrates, 60 units), IV IgG (1 g/Kg, 2 days), and corticosteroids (prednisone, 1 mg/Kg) and no hemorrhagic complications at were observed at vaginal delivery.
The mean estimated blood loss was 600 mL in the case of cesarean section and 393 mL in the case of vaginal delivery. The mean estimated blood loss for all of the women at delivery was 483 mL. In ITP pregnancies, the estimated blood loss during the delivery process was no different from that of normal pregnancies6).
Neonate outcomes
Since this study was performed retrospectively, a platelet count measured at the time of birth was available for only one neonate. Therefore, the incidence and severity of thrombocytopenia in neonates could not be evaluated. Nevertheless, 28 healthy babies were born from 28 full term pregnancies. None of the full-term newborns showed any signs of hemorrhagic diathesis, nor did they require any treatments to raise platelet counts. Three fetuses in 31 pregnancies died without hemorrhagic complications. Two fetuses died because of intrauterine death of unknown origin at 7 and 16 weeks gestation. Both fetuses had no signs of hemorrhage after evacuation. One fetus that was delivered by cesarean section due to bleeding from a gastric ulcer of the mother died because of premature delivery and respiratory failure at 27 weeks gestation (Table 5).
DISCUSSION
In this study, about half of the pregnancies in ITP patients were diagnosed during pregnancy. Patients occasionally present for the first time with severe thrombocytopenia during pregnancy. When pregnant women present with thrombocytopenia, it is often impossible to distinguish underlying ITP from incidental thrombocytopenia of pregnancy. Often, one must wait until the postpartum interval to determine whether the patient had true ITP or incidental thrombocytopenia of pregnancy. Incidental thrombocytopenia of pregnancy resolves itself following delivery, whereas ITP typically does not.
We observed that the mean platelet counts before pregnancy, during pregnancy, and at delivery were similar. However, the platelet counts tended to fall at the third trimester. This finding was consistent with previous studies reporting that women with previously diagnosed ITP may experience an exacerbation in pregnancy4), with the nadir platelet count usually occurring in the third trimester2). Regardless of platelet counts, most of the pregnancies did not show hemostatic impairment during pregnancy or at delivery.
During pregnancy, most of the patients received therapy to raise platelet counts. The decision to treat a woman for thrombocytopenia was made by the attending physician and was based on several factors including platelet counts, signs and symptoms of bleeding, and need for invasive intervention. The therapeutic strategies in this study seem to involve overtreatment because platelet transfusion was performed even in patients with appropriate platelet counts. In the past decade, it was commonly assumed that many pregnant women with ITP would experience complications and would sometimes suffer from fatal thrombocytopenic disorders during pregnancy7,8). These conclusions led to aggressive interventions that ranged from the administration of multiple medications to the pregnant mother with ITP or the performance of splenectomy during pregnancy, and often included delivery of the infant by cesarean section. Although the understanding of thrombocytopenia in pregnancy has evolved considerably over the last decade, the optimal diagnostic and treatment strategies for ITP in pregnancy remain controversial. The lack of prospective trials investigating ITP in pregnancy has meant that physicians frequently rely on personal experience and anecdotal evidence to manage these patients. However, recent studies have clarified several important aspects of ITP in pregnancy. Although the platelet count often drops in pregnant ITP patients, serious morbidity and mortality are distinctly uncommon for the mothers9,10). We also noted that most of the pregnancies did not show hemostatic impairment during pregnancy. However, in this study, the incidence of severe thrombocytopenia (below 20,000/mm3) at delivery was 6.5% (2 patients). One of two patients experienced complications with major bleeding, which suggests that hemorrhagic complications do occasionally occur which require careful monitoring and management.
It has been reported that, ideally, maternal platelet counts should be maintained above 30,000/mm3 throughout pregnancy and above 50,000/mm3 near full-term to minimize the need for platelet transfusions11). Asymptomatic patients with platelet counts above 20,000/mm3 do not require treatment until delivery is imminent, but should be carefully monitored, both clinically and hematologically. It is also known that platelet counts of above 50,000/mm3 are regarded as safe for normal vaginal delivery. A platelet count of above 50,000/mm3 is also safe for cesarean section, but would preclude the use of epidural anesthesia, for which the platelet count should be above 80,000/mm3 11). In this study, the most commonly used therapy at delivery consisted of platelet transfusion. Because of rapid correction of platelet counts after platelet transfusion and lack of time from recognition of thrombocytopenia to delivery, the attending physician tended to overuse platelet transfusion. There was no significant difference in hemorrhagic complications between the platelet transfusion group and non-transfusion group.
Generally, cesarean section was preferred against among the general population in pregnancies with ITP. The strategy behind delivery by C-section was the theoretical reduction in the risk of intracranial hemorrhage in infants born to mothers with ITP. However, Cook and colleagues reviewed literature that included 474 pregnant women with ITP and reported that the incidence of intracranial hemorrhage among neonates with severe thrombocytopenia (below 50,000/mm3) was 4% after cesarean section and 5% after vaginal delivery12). The occurrence of postpartum anemia requiring transfusion was higher among those undergoing cesarean section13). Therefore, the mode of delivery should be determined by obstetric indication only. The use of delivery method was similar for that of both cesarean section and vaginal delivery in our study and there were no differences in hemostatic complications between the two groups.
Because this study was retrospectively analyzed, the incidence and severity of thrombocytopenia in neonates could not be evaluated. However, none of the full-term newborns showed any hemorrhagic complications or needed any treatment to raise their platelet counts. It has been reported that the overall frequency of thrombocytopenia in infants is quite low, and about 4% of infants will have a birth platelet count of less than 20,000/mm3, while approximately 1% will have significant bleeding complications. Other studies reported that 9% of infants born to women with ITP are born with platelet counts below 50,000/mm3, and counts often decrease during the first few days of life2,14). Numerous studies over the past two decades have documented that neonatal thrombocytopenia is both uncommon and unpredictable. Data from both prospective studies and retrospective studies give relatively consistent results15-20). Consequently, the risk of intracranial hemorrhage or other major bleeding complications is also uncommon.
In conclusion, our current results regarding ITP in pregnancy confirm the low hemorrhagic risk in infants and mothers. Most mothers with ITP can proceed with their pregnancies and deliver infants without complications. Since fetal risks from severe thrombocytopenia are rare, the delivery mode should be determined strictly by obstetric indications. ITP in pregnancy necessitates the management of two patients, the mother and her baby. Hence, the close collaboration of a multidisciplinary group composed of a hematologist, obstetrician, and pediatrician is essential.