A Case of Bronchiolitis Obliterans Organizing Pneumonia Associated with Adenovirus
Article information
Abstract
A 58-year-old man described a short history of dyspnea and a preceding flu like illness with roentgenographic features of an interstitial lung disease. An open lung biopsy specimen from him showed bronchiolitis obliterans with organizing pneumonia (BOOP). Adenovirus was isolated from a throat swab. There was both clinical and radiographic improvement with supportive care. We herein report a first case of BOOP associated with adenovirus in Korea.
INTRODUCTION
Idiopathic bronchiolitis obliterans with organizing pneumonia (BOOP) is a specific clinicopathologic entity of uncertain etiology. The clinicopathologic entity of BOOP has been observed in the context of collagen vascular diseases and other auto-immue diseases like SLE; secondary to treatment with gold, cephalosporin, amiodarone, and other drugs; following inhalation of cocaine; after cytomegalovirus pneumonia in patients following allogeneic bone marrow transplantation; and associated with human immunodeficiency virus infection1). Previous reports2)–5) suggested that BOOP occurs with adenovirus. However, well documented reports of BOOP in patients with adenovirus infection were uncommon. Especially, in Korea, we cannot find a case report regarding BOOP associated with adenovirus. We report a first case of BOOP associated with adenovirus proven by open lung biopsy and culture of adenovirus in Korea.
CASE REPORT
A 58-year-old man was admitted because of acute onset of non-productive cough, fever, myalgia and progressive dyspnea over a one-week period. This was associated with bilateral pulmonary infiltrates on chest PA. Prior to his illness, the patient had been completely well, without past history of either cardiac or respiratory disease. Of particular note, he was an ex-smoker who was unemployed and did not have hobbies which might have exposed him to possible harmful antigens. Neither the patient nor any of his close associates suffered from tuberculosis, and he denied taking drugs or exposing himself to noxious gases prior to his illness.
On admission to hospital, he was distressed, tachypneic, and pyrexial with a respiratory rate of 35 per minute and a body temperature of 38°C. He was normotensive and his pulse rate was 90 beats per minute. The patient was not jaundiced. The head and neck were entirely normal.
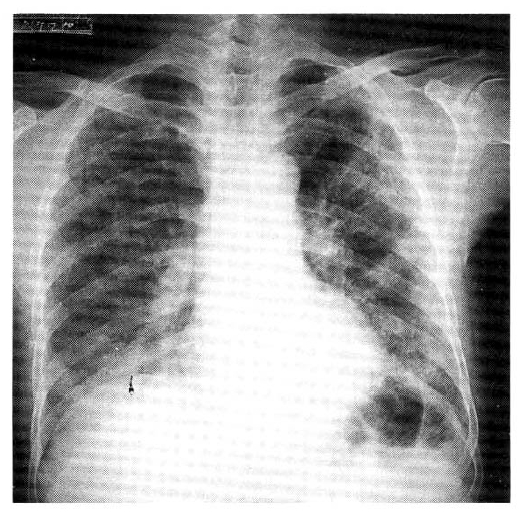
Examination of the chest revealed intercostal recession, but the respiratory excursion was equal bilaterally. The chest was resonant to percussion. The most striking feature on auscultation was the presence of extensive, bilateral, late inspiratory crepitations predominantly over the middle and lower lung zones. Vocal resonance was normal. Cardiac and abdominal examination were within normal limits. The chest radiographs showed multifocal and patchy distribution of opacities in the periphery of both lung zones (Fig. 1). Serial radiographs showed migratory natures of consolidations. High resolutional computed tomographic scan (HRCT) revealed nonsegmental and peribronchial distribution of alveolar consolidation and ground glass attenuation in the subpleural area, predominantly (Fig. 2). But, the consolidation had vertical zonal or anterior-posterior predominance. Several poorly defined centrilobular nodules and bronchial wall thickening with dilatation in the area of consolidation were seen. There was no evidence of pleural effusion or lymphadenopathy. Also, noted irregular lines were seen in both upper lung zones, suggestive due to previous centrilobular emphysema. Pulmonary function studies revealed an FVC of 3.28 L (88 percent predicted) and an FEV1 of 2.62 L (86 percent predicted) with an FEV1/FVC ratio of 98 percent. The patient’s arterial blood gas levels (at room air) were as follows: pH; 7.48, PaCO2; 27.8mmHg, PaO2; 43.8mmHg and HCO3-; 21.1mmol/L. The ECG was normal. Laboratory findings disclosed the following values: erythrocyte sedimentation rate (ESR), 60mm/h; hemoglobin level 13.8g/dl; leukocyte count 22,100/mm3, with a normal differential cell count; platelet count, 473,000/mm3; BUN, 6mg/dl; creatinine, 0.8mg/dl; sodium 140mEq/L; potassium 3.8mEq/L; lactate dehydrogenase, 263.6 IU/L; alkaline phosphatase, 84.4 IU/L; GOT 16.2 IU/L; GPT 9.7 IU/dl. Levels of antinuclear antibody, rheumatoid factor and anticardiolipin antibody were all normal and complement levels were normal. Mycoplasma serology was negative. Cultures of blood and sputum were negative. Results of the urinalysis were normal. He underwent bronchoalveolar lavage that revealed 61×104/ml with 63 percent macrophages, 6 percent lymphocytes and 31 percent neutrophils. An open lung biopsy was performed on the fifth hospital day which showed BOOP. The section showed well demarcated areas of fibrosis involving air spaces in patchy distribution (Fig. 3). Fibrosis consisted of young fibroblasts and inflammatory cells in myxoid-matrix-forming fibroblast polyps. These fibroblast polyps occluded distal bronchioles, alveolar ducts and adjacent alveolar spaces and they were sometimes covered by a lining of bronchiolar or alveolar epithelial cells. Most bronchiolar lumens or air spaces were filled with fibrinous exudate containing granular eosinophilic necrotic cellular debris (Fig. 4). On the second hospital day, viral culture was performed from his throat swab in the National Institute of Health, Korea and adenovirus was isolated. He received supportive treatments including oxygen inhalation with partial rebreathing mask, without application of mechanical ventilation and PEEP. One day later, dyspnea subsided gradually. There was improvement in gas exchange and clearing of roentgenographic infiltrates without corticosteroid and antibiotics therapy. At discharge, arterial blood gas levels (at room air) were as follows: pH 7.46, PaCO2 37.8mmHg, PaO2 83.4mmHg; HCO3-, 23.8mmol/L. He has continued to do well two months following discharge and the follow-up HRCT scan after 2 months showed almost disappeared subpleural air-space consolidation with poorly defined nodule and nearly normalized bronchial wall thickening with dilatation.
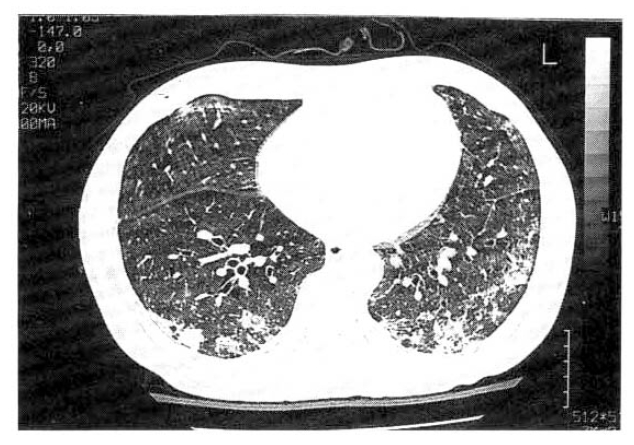
High resolution computer tomography (HRCT) shows bilateral patchy involvement of alveolar consolidation in mainly both lower lobe and focal ground-glass lesion is also noted.

Low power view shows elongated or serpiginous polypoid plugs of fibroblastic tissue filling air spaces (H-E, × 40).
DISCUSSION
BOOP is a clinicopathologic entity characterized by the following; the clinical presentation with a preceding flu-like illness and a short history of progressive dyspnea associated with patchy infiltrates on chest roentgenogram and/or computed tomographic (CT) scan and the pathohistologic pattern of intraluminal organization predominantly within the alveolar ducts6). Although the precise mechanism of injury leading to the formation of BOOP is unclear, recent pathologic studies suggest that injury to the alveolar epithelium may play an important role2). Lange described bronchiolitis obliternas in 2 patients in 1901. Autopsy of both of these patients revealed lungs studded with small miliary nodules8). Although this pathologic pattern has been described since 1901, it was rarely separated from other fibrotic lung disease until the report of 67 cases of BOOP by Epler and his colleagues in 19856). Their description of the clinical findings and prognosis of BOOP, including 50 patients with idiopathic BOOP, suggested that it should be distingushed from other interstitial diseases such as usual interstitial pneumonia. BOOP has been confirmed by many investigators since 1985. Subsequent papers have confirmed this finding, particulary emphasizing a much better prognosis in BOOP than in idiopathic pulmonary fibrosis7).
Pathologic confirmation is important, as it implies a benign course and therapeutic response to corticosteroid therapy. Performing an open lung biopsy is usually recommended but, in some cases, transbronchial biopsy as a diagnostic tool for BOOP may be considered11). In our case, we have done an open lung biopsy for differential diagnosis of interstitial lung diseases. The pathologic findings disclosed the filling of the alveolar and bronchiolar lumina by buds of loose connective tissue containing inflammatory cells and fibroblasts, which are associated with interstitial inflammation. This finding is the major histologic feature of BOOP.
Viral induced BOOP may be difficult to prove diagnostically but may be the cause of BOOP4, 5). Epler and his colleagues reported that a persistent, nonproductive cough was the most common symptom and a flu-like illness with fever, sore throat and malaise occurred in about one third of 50 patients with idiopathic BOOP6). This systemic flu-like symptom which is of short duration suggests that a portion of BOOP cases may represent an organizing bronchopneumonia caused by an undiagnosed viral infection7). Adenoviruses are most important clinically because of their capacity to cause acute infections of the respiratory system and conjunctivae. Pulmonary complications from adenovirus infection in healty adults are rare. The patients had a variety of influenza-like syndromes grouped under the term acute respiratory disease. Therapeutic agents for adenovirus pneumonia (including corticosteroids for pulmonary fibrosis) are of unproven benefit and support of ventilatory function until resolution of infection is the only known mode of management.
The adenovirus is an attractive candidate in the pathogenesis of BOOP because it has the ability to produce bronchiolitis and bronchopneumonia. Bronchiolitis obliterans, reported after adenovirus 21 infections in children2), suggests that this virus might cause some cases of BOOP in adults. Hogg et al in 1989 reported that in situ hybridization was a useful technique for demonstrating active adenovirus infection of the lung, and showed that the virus had disappeared to a very low number of copies per infected cell during recorevry from an acute infection3). Because of the high incidence of bronchiolitis obliterans reported after adenovirus 21 infections in children, a Vancouver study was designed to test this hypothesis using in situ hybridization techniques for adenovirus. Tissue obtained by open lung biopsy from 20 patients with BOOP showed 4 with the presence of adenovirus by in situ hybridization technique. The investigators conclude that the clinicopathological entity characterized as BOOP has several causes and that adenoviral infection may explain the etiology of a proportion of these cases4).
In the pathogenesis of BOOP, the role of adenovirus is not well known. The present study is that latent adenoviral infection may have the capability of amplifying the inflammatory response of airways. A current model is that adenoviral integration into the host cell establishes the presence of the E1A DNA of adenovirus and the E1A region may contribute to the pathogenesis of chronic airways obstruction10). This might contribute the connective tissue proliferation in the terminal and respiratory bronchioles and the histologic picture of acute bronchiolitis and to adult respiratory distress syndrome into one of BOOP.
The efficacy of corticosteroid therapy in patients with BOOP remains unclear. Although most patients are treated with corticosteroids and do well, there have been no prospective trials. In the series of Epler et al, patients with BOOP did well regardless of therapy, and of 11 untreated patients, only one developed progressive pulmonary disease6). Another study of patients with BOOP has suggested that patients with patchy lung involvement fare better than patients with diffuse disease9). In our patient with multiple patchy involvement, we did not use corticosteroid and improvement was noticed.
In cases of adenoviral infection, cells containing large basophilic, intranuclear inclusions giving the appearance of smudge-cells have been described in some cases. These histologic features were not found in our patient and we did not use in situ hybridization techniques for adenovirus on lung tissue. We could say that there is a strong association between adenovirus and BOOP if we had proven multinucleate giant cell, intranuclear inclusion, adenoviral DNA copies and viral particles on lung tissue obtained by open lung biopsy from our patient.
In summary, this case shows the association between BOOP and adenovirus in a Korean adult man. Adenoviral infection should be considered in the differential diagnosis of pulmonary opacities in patients with BOOP. We think it is very important to endeavor to isolate adenovirus for the evaluation of the underlying cause of BOOP.
Acknowledgments
We thank Joo Sung Kim and his colleagues in the Division of Respiratory Viruses, NIH, Korea, for their isolating of adenovirus.