INTRODUCTION
SweetŌĆÖs syndrome, or acute febrile neutrophilic dermatosis, is characterized by fever, neutrophilia, multiple painful asymmetric erythematous cutaneous plaques, dermal infiltrates consisting of mature neutrophils and rapid response to steroid therapy1). SweetŌĆÖs syndrome is associated with malignancy, most typically with acute myelogenous leukemia, in almost 10ŌĆō20% out of all its cases2).
All-trans retinoic acid (ATRA) induces complete remission out of a large proportion of patients suffering from acute promyelocytic leukemia (APL)3). Compared with conventional cytotoxic anticancer drugs, ATRA is considered relatively safe with only a few side effects. Retinoic acid syndrome is well known as the most serious adverse reactions of ATRA3). Cases of SweetŌĆÖs syndrome associated with ATRA have scarcely been reported worldwide, however4ŌĆō10). The cases of SweetŌĆÖs syndrome related with ATRA mean a big challenge to physicians. It is because SweetŌĆÖs syndrome appearing with ATRA treatment should be differentiated from the possibility of infectious conditions11).
We report a patient with APL who developed fever and multiple painful erythematous skin lesions during ATRA treatment. A wide range of evaluation found no source of infection. SweetŌĆÖs syndrome was diagnosed on the result of skin biopsy. As steroid therapy was introduced, fever and skin lesions abated promptly.
CASE REPORT
A 35-year-old Korean woman presented with dizziness, dyspnea on exertion, ecchymoses and menorrhagia. Investigation showed a hemoglobin of 3.5 g/dL, a hematocrit of 10.8%, a white blood cell count of 18.08├Ś109/L (70% promyelocytes, 14% band and polymorphonuclear cells, and 11% lymphocytes), and a platelet count of 29├Ś109/L. Bone marrow aspirate and biopsy showed hypercellular marrow with 31% myeloblasts and 38.5% hypergranulated promyelocytes containing Auer rods with faggot formation. By flow cytometric analysis, leukemic cells expressed CD13 and CD33. Cytogenetic analysis showed the characteristic t (15;17) translocation in 12 of 18 metaphases. The diagnosis of APL was made. PML/RARA mRNA was also detected by the reverse transcription polymerase chain reaction (RT-PCR) method. Fever developed during the evaluation period and empirical antibiotics, including a third generation cephalosporin combined with an aminoglycoside were started.
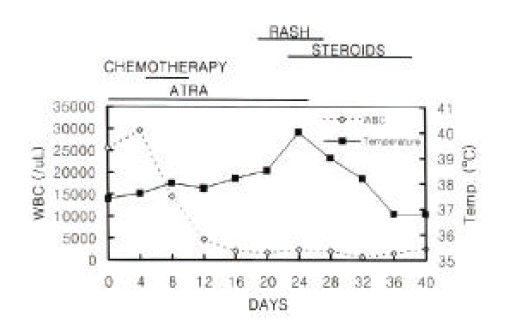
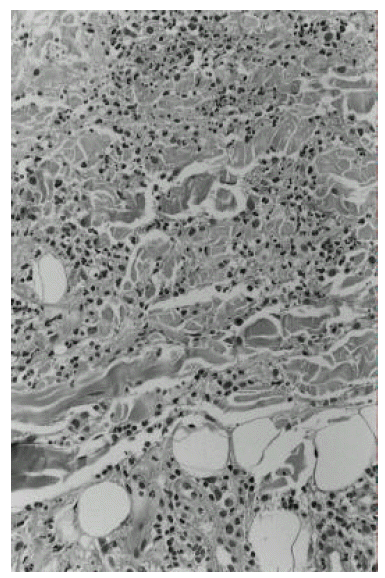
The clinical course of the patient is summarized in Figure 1. Induction therapy with ATRA was initiated at a dose of 70 mg/day (45 mg/m2/day). On the 4th day from the start of ATRA, white blood cell count increased up to 28.3├Ś109/L and chemotherapy with idarubicin (12 mg/m2/day for 3 days) and cytarabin (100 mg/m2/day for 7 days) was instituted. On day 24, multiple painful erythematous plaques appeared on both cheeks, right wrist and both shins and the temperature rose to 40┬░C. At that point, her white cell count was 2.5├Ś109/L, with 80% neutrophils and 0% blasts. All blood cultures and urine cultures were negative; her chest X-ray was unremarkable. Skin biopsy was performed from both cheeks and left shin, which revealed a dermal infiltrate of mature neutrophils and subepithelial edema (Figure 2). There was no fibrinoid necrosis or intramural cellular infiltrate suggesting vasculitis.
Special stains (Gram and fungal) were negative. The findings were compatible with SweetŌĆÖs syndrome. All-trans retinoic acid was discontinued and oral prednisone (1 mg/kg/day) started to be administered. As a result, her lesions were outstandingly improved within 48 hours without recurrence after tapering the corticosteroids.
On day 37, bone marrow aspiration and biopsy with cytogenetic analysis and RT-PCR were repeated and complete remission was confirmed based on the above studies. Thereafter, she completed two courses of consolidation chemotherapy. ATRA was cautiously retried as a maintenance therapy after the completion of consolidation chemotherapy. No skin lesions developed subsequently. She keeps on ATRA medication and remains in remission for 15 months.
DISCUSSION
SweetŌĆÖs syndrome develops under lots of conditions such as autoimmune disorders, infections, hematologic malignancies and solid tumors12). There are some differences between idiopathic SweetŌĆÖs syndrome and the malignancy-associated one in terms of clinical characteristics. Idiopathic SweetŌĆÖs syndrome is mainly found in middle-aged women with a prior upper respiratory tract infection13). It is characterized by fever, neutrophilia, an elevated erythrocyte sedimentation rate and a neutrophilic dermal infiltrate. The skin lesions are mostly found on the upper extremities, head and neck in the forms of red pseudovesicular plaques and nodules. In contrast to idiopathic SweetŌĆÖs syndrome, SweetŌĆÖs syndrome shown in patients with hematologic malignancy is characterized by no sexual predilection, typical and/or more severe (vesicular, bullous, ulcerative) cutaneous lesions, extracutaneous involvement in nearly 50% of the patients, absence of neutrophilia in over one-half of the patients, frequent anemia and abnormal platelet counts and a high rate of recurrence which often herald a tumor relapse1). Importantly, the onset of SweetŌĆÖs syndrome occurred prior to or coincident with that of the associated malignancy in nearly two-thirds of these patients2). Corticosteroids are the treatment of choice for both idiopathic and malignant-associated SweetŌĆÖs syndrome2, 12).
Besides many conditions related with SweetŌĆÖs syndrome, only a few drugs seem to be associated with it. These drugs include minocycline, lithium, furosemide and ATRA12). Less than 10 cases of ATRA-related SweetŌĆÖs syndromes were reported worldwide4ŌĆō10).
ATRA revolutionized the therapy of APL. ATRA causes the brief proliferation of the malignant cells through maturation, terminal differentiation and ultimately apoptotic death of the cell via binding to RAR╬▒. By the introduction of induction therapy with ATRA into the treatment of APL, high complete remission rates (80% to 90%) in APL patients could be attained without the toxic effects of the standard chemotherapy3). However, there are side effects with ATRA; headache, bone pain, intracranial hypertension, hypertriglycemia, lymphadenopathy, hypercalcemia, liver function impairment, skin pruritus, bone marrow necrosis, thromboembolic events, retinoic acid syndrome and rarely, in a few cases, SweetŌĆÖs syndrome3ŌĆō10).
The cause of SweetŌĆÖs syndrome is uncertain. But the role of the immunologic mechanism can be inferred from the following three: the clinical response to corticosteroids, systemic organ manifestation, and the similarity to other hypersensitivity reactions4). Also, the role of G-CSF in pathogenesis is suspected by the fact that SweetŌĆÖs syndrome may develop during G-CSF administration for granulocytopenia14, 15). ATRA was found to up-regulate IL-1╬▓ and G-CSF protein expression. The increase of IL-1╬▓ and G-CSF production in the presence of ATRA was highly correlated to the increase in APL cell count16). In this case, the leukocyte count was increased from the initial state during ATRA treatment. Cases with increased white blood cell counts during ATRA administration were reported, and even the occurrence of retinoic acid syndrome following on SweetŌĆÖs syndrome was reported8, 10). The association of G-CSF level with SweetŌĆÖs syndrome and leukocytosis is not proved in these cases because serum G-CSF measurement was not done. To elucidate the pathogenesis, it is recommended that various cytokines, including G-CSF, be measured when SweetŌĆÖs syndrome is suspected.
One of the most important clinical differential diagnoses of SweetŌĆÖs syndrome is an infectious condition. In this case skin lesions and fever developed in a neutropenic state induced by remission induction chemotherapy. The authors launched prompt and thorough investigation for all possible foci of infection because neutropenia is strongly correlated with an increased risk of infection, and delay in therapy may even be fatal. Especially, it is very important to discern the characteristic skin lesions associated with infections, like ones by Clostridium spp, Candida spp, and Pseudomonas aeruginosa, in febrile neutropenic patients, for it makes the appropriate therapy to bo taken promptly17). In this regard, skin lesions in febrile neutropenic patients provide important clues to accurate diagnosis and warrant immediate biopsy18). In addition to infectious conditions, SweetŌĆÖs syndrome should be one of the differential diagnoses in febrile neutropenic patients with skin lesions. When SweetŌĆÖs syndrome is not considered, it is so difficult to differentiate infection from SweetŌĆÖs syndrome that the use of antibiotics could be continued too long and the use of corticosteroid could be delayed11).
In conclusion, it is important to notice the side effects and complications caused by the treatment of APL with ATRA because SweetŌĆÖs syndrome is not simply differentiated from infection. Unexplained fever and skin lesions, shown in neutropenic patients during ATRA administration, require us to be alert to the possibility of infection and ATRA-induced side effects, including SweetŌĆÖs syndrome. Immediate identification of infection foci and prompt skin biopsy are also required.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





