INTRODUCTION
Helicobacter pylori infection is now recognized as the major cause of chronic gastritis throughout the world1ŌĆō3). A fraction of infected persons developed peptic ulcer disease4) or gastric cancer2, 5, 6), accounting for its clinical significance. The pathophysiology of this infection can be better understood by several concepts, such as heterogeneity of strains7), persistence of infection8), immunological down regulation9), physiological consequences and variability in outcome10). Microbial, host and environmental factors must contribute to the outcome variation, respectively. Especially, the relapse of H. pylori has been reported after antimicrobial therapy11ŌĆō13), and its transmission is still unknown. The fact that H. pylori can convert under unfavorable conditions into a metabolically active but non-culturable coccoid state has stimulated speculation about its role in transmission and reinfection14, 15). Some investigators16ŌĆō18) suggested that the coccoid forms are degenerative and have no potentiality of infection, like Campylobacter jejuni, and others14, 19ŌĆō21) supposed that they are dormant and one stage of the biological cycle of H. pylori, like Vibrio vulnificus. Some reported some studies22, 23) have suggested that the coccoid forms of H. pylori could be potentially viable. However, regrowth of H. pylori from the coccoid forms could not be possible. In the present study, to elucidate the viability of coccoid forms, we induced conversion from bacillary to coccoid forms and studied the morphological changes and antigenic evolutions during coccoid conversion.
MATERIALS AND METHODS
1. Strain and culture conditions
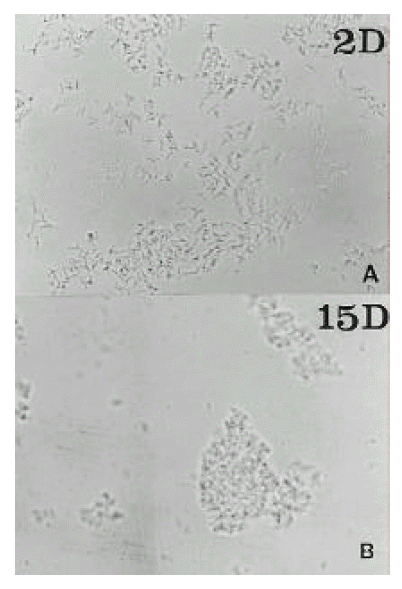
The H. pylori strain used (C001) was isolated in the Research Institute for Gastroenterology of Dankook University (Chunan, Korea) from a gastric biopsy of a patient with gastric cancer. Cells were grown at Brucella blood agar with 5% horse serum and antimicrobial agents, at 37┬░C under microaerophillic conditions with 10% CO2 and 5% O2. Added antimicrobial agents were vancomycin (10 mg/L), colistin (5 mg/L), trimethoprim (5 mg/L) and amphotericin B (5 mg/L). The cells of a 2day culture of H. pylori C001 were harvested from one plate, suspended in phosphate-bufferd saline (PBS, pH 7.4), and adjusted to a turbidity of 1 MacFarland unit. After Gram staining, it was microscopically observed that this suspension contained only bacillary forms. The suspension was then used to innoculate 10 plates of Brucella blood agar (0.1 ml/plate). The plates were incubated at 37┬░C under microaerophillic conditions. The cells of one plate were harvested daily and suspended in 1 ml of PBS (pH 7.4). Enumeration of colony forming unit were performed by standard serial dilution and plate count procedures. Total cell (bacillary or coccoid forms) counts were assessed by turbidimetry at 540 nm. To each suspension, Gram-stained smears were used to assess the relative percentages of coccoid and bacillary forms. These measurements were made blindly by 30 different persons, and the averages of their determinations were considered as the final results. Also, the suspensions of day 2, 7, 9 and 15 were used for electron microscopic examinations.
2. Preparation for electron microscopic examination
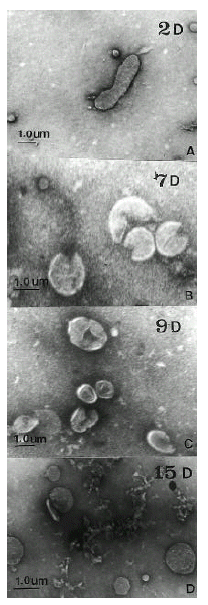
Bacteria were harvested from plates, fixed for microscopy with 2.5% glutaraldehyde in 100mM sodium cacodylate buffer. Cells were concentrated by centrifugation and samples were removed for examination by differential interference contrast light microscopy. To assist in identification, selected samples were also negatively stained with 1% uranyl acetate for transmission electron microscopy (TEM).
3. Western blotting
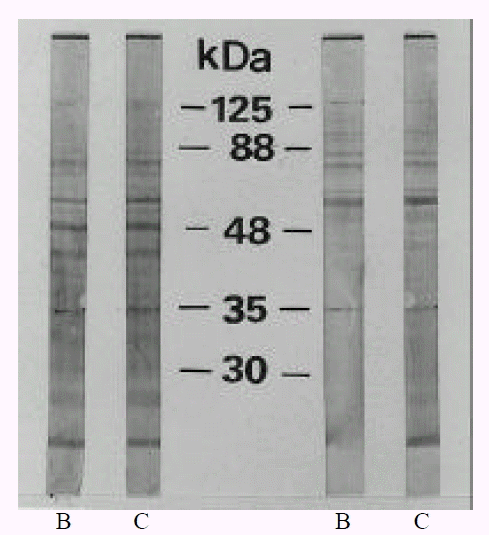
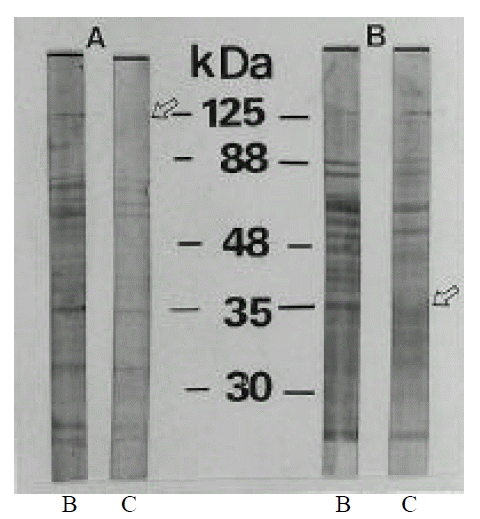
The evolution of the antigenic profiles during coccoid conversion of H. pylori C001 was studied by Western blotting, using sera from thirty patients known to be colonized by H. pylori. Total antigens of H. pylori C001 were obtained by sonification of bacterial cells harvested at different stages of rod and coccoid, respectively, and suspended in PBS (pH 7.4). After sonification, the unlyzed bacteria were eliminated by centrifugation (3000 rpm, 65 min), and the supernatants were saved, adjusted to 1 ml of protein per ml, and frozen at ŌłÆ70┬░C until use. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was performed according to the method of Laemmeli24) with a 4% stacking gel and a 10% separating gel. Prior to electrophoresis, the protein solutions were heated at 100┬░C for 5 min in a sample buffer containing 5% (wt/vol) SDS and 0.42% (wt/vol) 2-mercaptoethanol. Electrophoresis was conducted for 1 hour under a constant voltage (15 v/um) using the minigel system (Bio-Rad). Proteins were blotted onto a pre-wetted nitrocellulose membrane (Bio-Rad) by using a Mini Trans-Blot electrophoretic transfer cell (Bio-Rad) under a constant current of 200 mA for 1 hour. The blots were incubated for 1 h with human sera diluted at 1/100; they were then rinsed and incubated in goat serum anti-human IgG conjugated with alkaline phosphatase (Dakopatts, Copenhagen, Denmark). After a final wash, the nitrocellulose filters were developed with 5-bromo-4-chloro-3-indolylphosphosphate (BCIP) as a substrate, and nitroblue tetrazolium as a chromogenic indicator. The reactions were stopped after 20 min by washing the filters extensively with distilled water.
RESULTS
1. Examination of morphological changes
As assessed by turbidimetry, the bacterial mass increased from day 1 to day 3. The maximal active growth was observed on day 2ŌĆō3 (36 hours). The stationary phase was day 4ŌĆō6. The proportion of coccoid forms increased from 0 to 100% from day 2 to day 15 (Fig. 1). Between day 5 and day 9, the fraction of coccoid forms on the plates was increased. Transmission electron micrographs at day 2 showed spiral rod forms. At day 7, the relative number of bacillary forms decreased and U-shaped forms became predominant, and they looked like invaginated bacilli. At day 9, U-shaped forms were converted to doughnut-shaped forms. At day 15, only globular full coccoid forms were observed (Fig. 2). The ultrastructure of the inner cell side was not observed.
2. Evolution of antigens of H. pylori during coccoid conversion
The evolution of the antigenic profiles during coccoid conversion of H. pylori was studied by Western blotting, using different sera from culture positive patients. These sera were used to reveal the total antigens of the strain cultured for 2 day (0% coccoids) and 15 days (>99% coccoids). SDS-PAGE analysis of whole cell preparations of H. pylori showed numerous bands between 30 and 125 kDa. These proteins included CagA at 125 kDa, VacA (vacuolating cytotoxin) at 88 kDa, an adhesin and porin at 35 kDa and urease subunit at 30 kDa25, 26). The antigenic profiles were not changed in 46.7% (14/30 cases) (Fig. 3) and were changed in 53.3% (16/30 cases) during coccoid conversion. Antigenic fractions changed during coccoid conversion were protein band at 125 kDa and band at 35 kDa, which were intensively detected in bacillary forms. Those proteins which disappeared included CagA and porin, outer membrane (Fig. 4). Disappearance of CagA protein (125 kDa) during coccoid conversion was observed in 68.8% (11/16 cases) and disappearance of porin (35 kDa) in 62.5% (10/16 cases). The rest of the profiles were identical between rod and coccoid forms.
DISCUSSION
Helicobacter pylori, a gram negative spiral bacterium, can survive in such diverse environments as the human stomach of low pH23), gut of high osmolarity27) and water28). In order to adapt to such crucial conditions, the bacteria should carry systems that respond to changes in nutrient, osmolarity, temperature and other external factors. The organisms exist in two forms, an actively dividing spiral forms and a coccoid form of arrested growth under various stress, including the starvation for nutrients29), extended incubation28), accumulation of metabolic products14), pH alteration14) and exposure to antimicrobial agents30). The viability of the coccoid form of H. pylori, the possible role of this form in transmission and as a cause of reinfection is controversial14, 15). Although the mode of transmission still remains unclear, oral-oral and oral-fecal transmission have been suggested31, 32). If H. pylori follows the latter route, they must pass through the anaerobic atmosphere of the alimentary tract which is an adverse situation for H. pylori. Under these stressed conditions, infected bacillary forms might be changed to coccoid form. Shirai et al.20) have reported that almost 100% cells changed to coccoid-like bodies within 24 hr of anaerobic incubation, and 0.1ŌĆō1% produced colonies on Brucella agar. Also, they revealed that the colonies appeared from coccoid bodies which remained viable under anaerobic conditions, although some of them appeared from a few spiral bodies, thereby they assisted H. pylori in passing the adverse anaerobic route of the human alimentary tract by changing their morphology. However, it is still unknown whether coccoid forms of H. pylori can revert to vital organism in vivo or if they are of any patho-physiological significance at all. According to some reports16ŌĆō18), the coccoid forms are degenerative and incapable to form complex adhesions and, hence, are of low pathogenic potential. But others14, 19ŌĆō21) suggested that coccoid forms are dormant cells and viable cells. Vijayakumari et al.19) revealed that specialized attachment sites were seen in the interaction between coccoids and epithelial cells of KATO III cell, and these adherence patterns were similar to those observed with spiral forms in vivo, suggesting a possible pathogenic role for the coccoids of H. pylori. Also, with antigens prepared from both coccoid and spiral forms, immunoreactive protein bands of 128, 116, 110, 95, 91, 66, 60, 54, 50 and 33 kDa were conserved in both the coccoid and spiral forms by the results of Western blotting. Vijayakumari et al.19) suggested that the coccoids could be differentiated infective form of H. pylori, and that they could evoke an immune response from the host after attachment to gastric epthelial cells. We found the morphological changes during coccoid conversion by the transmission electron microscopy. Spiral bacilli forms at day 2 converted to full coccoid forms at day 15, through the U-shaped forms at day 7, and doughnut-shaped forms at day 9. Although we could not detail the change of ultrastructure during coccoid conversion under our transmission electron microscopy, coccoid forms were considered via U-shaped and doughnut-shaped forms. Bena├»ssa et al.21) revealed that initiation of conversion from bacillary was the formation of dense periplasmic material, followed by an inwardly curved formation of bacilli as an intermediate step between the bacillary and coccoid forms and, then, a change in the protoplasmic cylinder was evoked to full coccoid forms, strongly suggesting a transition of bacillary to full coccoid forms via U-shaped forms. The extracytoplasmic side of the invaginated membrane might serve as a site for the oxidation of toxic material, which has been described for Ecthiorhodospira mobilis and other spirilloid or vibroid gram negative organism29, 33), just like H. pylori. Therfore, similar invagination of the membrane could consolidate the potential viability of H. pylori coccoids. On the antigenic evolution, we observed the identical antigenic profile between the bacillary and coccoid forms in 46.7%, and changed antigenic profile in 53.3%. Interestingly, disappeared protein bands during coccoid conversion were CagA and porin or adhesin, which were more intensively detected in bacillary forms. Those findings could suggest that virulent factors of H. pylon might vanish in some coccoid forms. Vijayakumari et al.19) and Bena├»ssa et al.21) have reported that the protein bands were the same pattern in both the coccoid and bacillary forms, which were a little bit different from our results. We think the same protein bands between the bacillary and coccoids highlight the significance of these antigens. However, we cannot clarify the role of the antigenic components absent in the coccoid forms. In conclusion, these results showed that coccoid forms of H. pylori retain antigenic characteristics similar to bacillary forms, and some of the antigens disappeared in coccoid forms. Therefore, coccoid forms might be viable, and represent one of the stages of the H. pylori biological cycle. However, it is still unclear whether the coccoid forms can revert to infective bacillary forms in vivo, whether they represent a temporary adaptation to a particular environment, and whether they are actually involved in the transmission of the bacterium.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





