INTRODUCTION
Behcet's disease (BD) is a chronic, relapsing, multisystemic inflammatory disease that's characterized by recurrent oral and genital ulceration and skin and ocular lesions1). Cardiovascular involvement occurs in 7~31% of the BD patients2, 3). There may be different types of vascular involvement, yet the majority of the vascular involvement is in the venous system and arterial involvement is rare. In general, BD patients with major vessel involvement have a poor prognosis.
Chronic inflammation is a non-traditional risk factor in the pathogenesis of atherosclerosis4), and accelerated atherogenesis has previously been shown in inflammatory rheumatic disease such as rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE)5, 6). Acute systemic inflammation and chronic systemic vasculitis are associated with endothelial cell dysfunction (ECD)7, 8).
The histopathological features of BD are mainly characterized by vasculitis, with prominent neutrophil and monocyte infiltration in the perivascular regions with or without fibrin deposition in the vessel walls9). Although the definite pathogenic mechanism for the vascular lesions in BD patients remains unclear, endothelial dysfunction is thought to play an important role in the development of these lesions. Histologic evidence of vasculitis with endothelial cell activation or injury is a characteristics feature of BD10). ECD is widely regarded as the initial lesion in atherogenesis11) and recent studies have shown that an increased carotid intima-media thickness (IMT) was significantly correlated with ECD12).
In this study, we investigated the carotid IMT and plaque formation in BD by using high-resolution B-mode ultrasonography. We evaluated the relationship between such factors as vascular involvement, other severe manifestations, disease activity or the use of immunosuppressive agent and the carotid IMT in patients with BD.
MATERIALS AND METHODS
Subjects
We prospectively analyzed the patients who visited the Department of Rheumatology at Chonnam National University Hospital from July 2004 to October 2004 and from June 2005 to October 2005, and these were the patients who were eligible for participation in this study. Forty consecutive patients with BD (mean age: 39.1┬▒8.5 years, 60% males) who fulfilled the International Study Group Criteria13) were finally enrolled.
As the healthy control group, 20 age and gender-matched healthy controls (mean age: 40.2┬▒5.1 years, male 65%) were included. We excluded those subjects with hypertension, diabetes mellitus or a previous history of ischemic heart disease or cerebrovascular events. None of the subjects had a history of smoking or statin therapy.
Clinical assessments
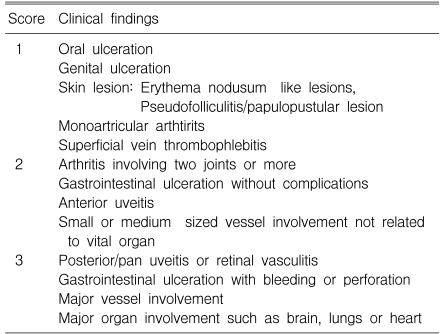
We collected the clinical data on the disease duration, the medication history, the clinical manifestation and the disease activity score for BD at the time of study and during the month before the study. The disease activity score was calculated by summing the points for all the disease manifestations as follows: 1 point for each mild symptom (oral or genital ulcer, skin lesions, monoarticular arthritis and superficial thrombophlebitis); 2 points for each moderate symptom (arthritis, small or medium-sized vessel involvement, anterior uveitis and gastrointestinal ulceration); and 3 points for each severe disease manifestation (posterior/pan uveitis or retinal vasculitis, gastrointestinal ulceration with bleeding or perforation, major vessel involvement and major organ involvement) (Table 1)14, 15).
Laboratory analysis
We measured the laboratory parameters at the time of the ultrasound scan. The erythrocyte sedimentation rate (ESR) was measured by using the modified Westergren method and the C-reactive protein (CRP) level was assessed by the turbidmetric immunoassay with using a Hitachi 7600 analyzer (Hitachi, Tokyo, Japan). The serum levels of total cholesterol, triglyceride, high-density lipoprotein cholesterol and low-density lipoprotein cholesterol were measured by standard enzymatic methods. The lipoprotein (a) level was measured by nephelometry and the serum homocysteine level was measured by chemiluminescence immunoassay.
Carotid artery evaluation by B-mode ultrasound
The carotid IMT and plaques were measured in the left and right common carotid arteries (CCA) by using high-resolution B-mode ultrasound (HP Sonos-5500, Philips, USA) with an electric linear transducer (midfrequency 11.0 MHz). The left and right CCAs were scanned with the beam focused on the near and far walls of the distal 2 cm of the common carotid artery proximal to its bifurcation. The IMT was measured, at 3 points on the far walls of both distal CCAs, as the distance from the leading edge of the first echogenic line to the leading edge of the second echogenic line, as defined by Pignoli16). The 3 IMT points were 1) the site of the greatest thickness, 2) a point 1 cm upstream and 3) a point 1 cm downstream from the site of the greatest thickness. The values from the 3 locations were averaged to produce a mean IMT for each side. Plaque was defined either as a distinct protrusion of > 1.5 mm into the vessel lumen or as definite echogenicity with a posterior echogenic shadow17).
Statistics
All the metric variables are described as means┬▒standard deviations. The differences of the metric variables between the groups were analyzed using t-tests and chi-square statistics. Correlations between the IMT and parameters were described with Pearson's correlation coefficients. All the statistical process was done using SPSS-PC 11.0 (Statistical package for the social sciences, SPSS-PC Inc. Chicago, USA). A p value <0.05 was considered to be significant.
RESULTS
Clinical and laboratory characteristics
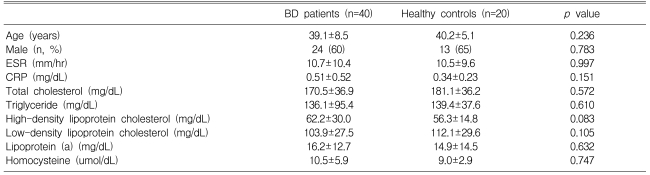
The clinical characteristics are listed in Table 2. No significant differences of age, gender and the lipid profiles were observed between the groups. The mean values of the ESR, CRP and lipid profiles were within the normal limits in both groups. There were no significant differences in the laboratory findings between the groups.
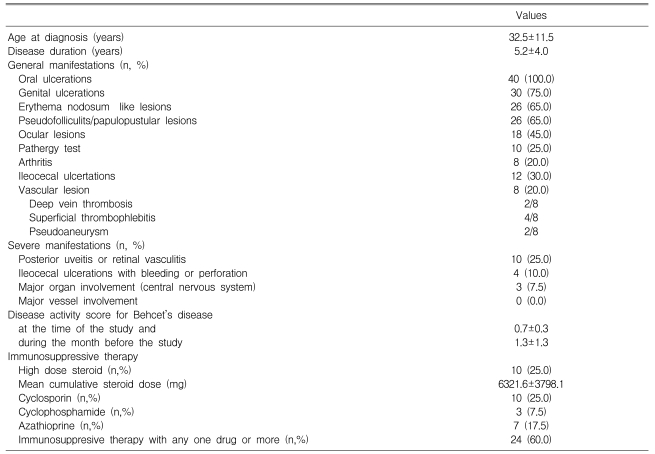
The clinical findings of BD are shown Table 3. The mean age at the time of diagnosis was 32.5┬▒11.5 years and the mean disease duration was 5.2┬▒4.0 years. Seventeen patients (42.5%) had severe manifestations; posterior uveitis or retinal vasculitis in 10 patients, ileocecal ulcerations with bleeding or perforation in 4 patients and neuro-Behcet disease in 3 patients. However, any cardiac involvement and major vessel involvement were not detected. Twenty-four patients (60%) received immunosuppressive therapy with one or more drugs. Ten patients took high-dose steroid, 10 took cyclosporine, 3 took cyclophophamide and 7 took azathioprine. The disease activity score at the time of the study and during the month before the study were 0.7┬▒0.3 and 1.3┬▒1.3, respectively.
IMT and the clinical and laboratory variables
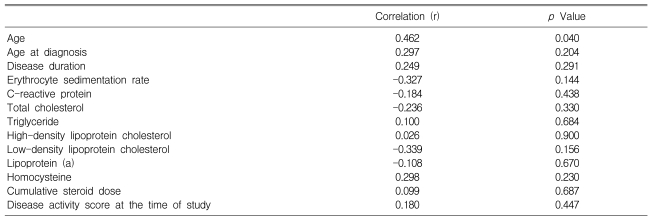
The carotid IMT in the BD patients was positively correlated with age (r=0.462, p=0.040), but it was not correlated with the disease duration, the cumulative steroid dose, the disease activity score, the ESR, the CRP level and the lipid profiles (Table 4).
IMT of the CCAs
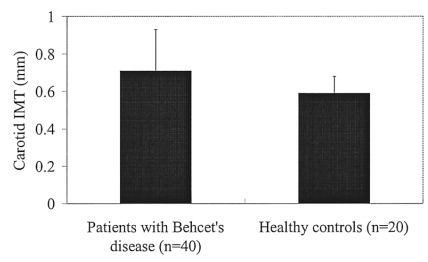
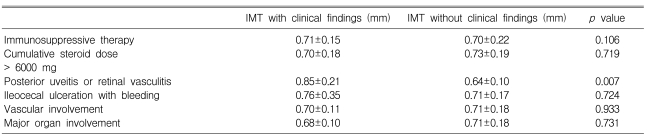
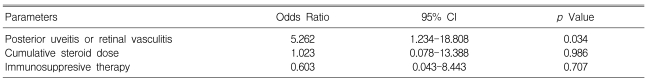
The carotid IMT in the BD patients was significantly higher than that in the healthy controls (0.71┬▒0.22 mm vs. 0.59┬▒0.11 mm, respectively, p=0.006) (Figure 1). Carotid plaque was present in 1 BD patient and in none of the controls. In the BD patient group, the BD patients with posterior uveitis or retinal vasculitis had a significantly higher carotid IMT than those without posterior uveitis or retinal vasculitis (0.85┬▒0.21 vs. 0.64┬▒0.10 mm, respectively, p=0.007) (Table 5). However, no difference of the carotid IMT in the patients with BD was observed according to the presence of vascular lesion, a history of immunosuppressive therapy or involvement of major organs (Table 5). The carotid IMT in the 20 patients who were treated with a cumulative steroid dose >6000 mg did not significantly differ from that in the 20 patients who had not received a cumulative steroid dose Ōēż6000 mg (0.70┬▒0.18 mm vs. 0.73┬▒0.19 mm, respectively, p=0.719; the mean cumulative steroid dose, 10357.8┬▒4818.1 mg vs. 2511.7┬▒930.1 mg, respectively, p<0.001). On the multivariate analysis, the presence of posterior uveitis or retinal vasculitis was associated with a high carotid IMT (Table 6).
DISCUSSION
Acute systemic inflammation and chronic systemic vasculitis are known to be associated with ECD7, 8). Systemic inflammatory rheumatic diseases such as RA and SLE are associated with a significantly increased risk of cardiovascular disease, which often occurs at a younger age for these patients as compared to the normal population5). Vasculitis also plays an important role in atherogenesis18), as has been shown for Takayasu arteritis19) and Wegener's granulomatosis20). BD is a chronic, systemic inflammatory disorder with a diverse spectrum of clinical manifestations, including mucocutaneous, ocular, vascular, gastrointestinal, musculoskeletal and central nervous system involvement1). Vascular involvement in BD is usually recognized as an unclassified vasculitis, and it involves both the veins and arteries of all sizes21, 22). Endothelial cell injury or their pathological activation is the characteristic features of BD. An endothelial product, von Willebrand factor, was elevated in BD patients and particularly in the patients with active BD23). Despite the uncertainty concerning the pathogenic mechanism of the vascular lesions in BD patients, vascular endothelial dysfunction has been recognized to occur in BD and it is thought to play an important role in the vascular lesions10).
In this study, we demonstrated that the carotid IMT was significantly higher in BD patients than that in healthy controls. The previous studies demonstrated that the carotid IMT was significantly higher in BD patients than that in the healthy controls17, 24, 25). However, the authors of those studies did not investigate the carotid IMT according to the presence of the clinical findings of BD, such as the medication history or severe disease manifestations. Alan et al.24) reported that the carotid IMT was significantly higher and the arterial distensibility was significantly lower in patients with BD as compared to the controls. The IMT values were even higher in BD patients with vascular involvement compared to those without vascular involvement24). However, BD patients with major vascular involvement were included in that study, and no information was given regarding the use of steroid, the smoking status and the conventional risk factors of atherosclerosis. Rhee et al. reported on the IMT and arterial stiffness of the carotid artery in Korean patients with BD26) and they demonstrated that BD patients had significantly increased arterial stiffness in all the regional arterial segments27). In that study26), there were significant differences in the carotid arterial stiffness parameters between the BD patients and the control subjects; however, the carotid IMT of the patients with BD was not different from that of the control group. They did not evaluate the influence of the serologic markers and the clinical manifestation of BD on the carotid IMT. In our study, the carotid IMT was significantly higher in the BD patients with posterior uveitis or retinal vasculitis than that in those BD patients without posterior uveitis or retinal vasculitis. There were no significant differences of the IMT in the subgroups of BD with regard to the presence of vascular involvement and other severe manifestations such as major organ involvement and a history of immunosuppressive therapy. Major vascular involvements were not detected in our study group. Posterior uveitis or retinal vasculitis was the most frequent severe manifestation of BD and this recurred frequently in our study group. Also, carotid plaque was detected in the patient with recurrent posterior uveitis.
CRP is a powerful predictor of cardiovascular disease and this was independent of the serum lipid levels. An elevated CRP level is a very accurate marker of ECD28). Our data failed to show a significant correlation between the IMT and the ESR and the CRP level. The absence of a significant association between the carotid IMT and the CRP level might be explained by our use a conventional CRP assay, rather than using a high-sensitivity CRP (hsCRP) assay. If we had used a hsCRP assay, we might have found a positive correlation. Alternatively, the serum CRP level as determined by conventional assay fluctuates in BD patients and the median of the serial measurements may be more reliable than a single measurement.
In this study, the carotid IMT in the BD patients was positively correlated with age, but it was not correlated with the disease duration and cumulative steroid dose. Additionally, we could not demonstrate any correlation between the disease activity score and the carotid IMT in the BD patient group. BD is characterized by remissions and exacerbations, and it is a multisystemic inflammatory disorder. Each of the BD patients had different and various clinical manifestations. Most patients were in the inactive period and their CRP and ESR values were within the normal limits at the time of the study. Severe disease manifestations appeared less frequently and so the disease activity score at the time of study and during the month before the study started was low in our study group. Further studies on patients with higher disease activity will clarify whether the disease severity increases the risk of subclinical atherosclerosis in patients with BD.
In conclusion, the BD patients without significant cardiovascular involvement demonstrated a higher carotid IMT compared with the healthy control subjects. The increased arterial wall thickness was independently associated with the disease duration and the disease activity score. Our data did not show a significant association between the carotid IMT and immunosuppressive therapy, including high dose steroid therapy for BD. These results suggest that BD itself may lead to an increased carotid IMT, and this is possibly due to an increase in the atherosclerotic changes.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





