 |
 |
| Korean J Intern Med > Volume 22(4); 2007 > Article |
|
Abstract
Leptomeningeal carcinomatosis occurs in approximately 5% of patients with cancer. The most common cancers involving the leptomeninges are breast, lung cancer and melanoma. However, gastric adenocarcinoma has been rarely reported with leptomeningeal carcinomatosis. The presenting manifestations are usually headache, visual disturbances and seizures. We report a case of leptomeningeal metastasis that presented as a gastric cancer. A 49-year-old woman was admitted to our hospital with the symptoms of headache and melena for 10 days. The endoscopy showed a thickening of the folds of the stomach compatible with the diagnosis of a Borrman type IV gastric cancer. The biopsy revealed a signet ring cell carcinoma. The MRI of brain showed no abnormal findings; however, the patient complained of an intractable persistent headache, nausea and vomiting on admission day 6. The cytology examination of the cerebrospinal fluid supported the diagnosis of metastatic signet ring cell carcinoma.
Leptomeningeal carcinomatosis (LC), also called carcinomatous leptomeningitis, neoplastic (lepto)meningitis, leptomeningeal metastasis, or more commonly leptomeningeal seeding, is the dissemination and growth of cancer cells within the leptomeningeal space; it is one of the most serious complications that can occur in cancer patients1). The incidence of LC is three to 8% of all solid cancers. LC is frequently found in patients with leukemia, breast cancer, lymphoma, and lung cancer2).
Gastric cancer (GC) is one of the most common causes of cancer related death in Asian countries including Korea. However, the development of LC from a GC is very rare. Kim, et al3) reported that the incidence of LC with GC was 0.06% of all cases of gastric cancer. LC is a more frequent manifestation of advanced or metastatic carcinoma rather than associated with early disease. It is usually discovered during the treatment period for the cancer. We report a rare case with LC as the presenting symptom and review the medical literature.
A 49-year-old woman was admitted to our hospital with headache, dizziness, easy fatigability, and melena for 10 days. She looked pale and acutely ill. There were no significant findings in the medical history. On physical examination, she was oriented to time, place and person. Her vital signs were as follows: a blood pressure of 130/75 mmHg, a pulse rate of 95 beats/min and a respiration rate of 21 breaths/min. There were no focal neurological signs or abnormal reflexes. The plantar reponses were flexor. There was no neck stiffness or nystagmus. The sensory examination was normal. The conjunctivae were pale, but not icteric. The breathing sounds were clear and the heart sounds were regular without a murmur. Hepatosplenomegaly was not noted. The symptoms were slightly improved with conventional analgesic agents. The MRI of brain showed no abnormalities, but the esophagogastroduodenoscopy showed thickening of the gastric folds on the greater curvature of the stomach and on the posterior wall of the stomach compatible with a Borrman type IV gastric cancer (Figure 1). The biopsy revealed a signet ring cell carcinoma (Figure 2, 3). The CT scan of abdomen and pelvis showed an advanced gastric cancer involving the posterior wall of the lower body of the stomach; in addition, the angle and the antrum had extensive metastatic lymphadenopathy. The patient complained of an intractable persistent headache as well as nausea and vomiting on admission day 6. A lumbar puncture and analysis of the cerebrospinal fluid (CSF) was performed. The results showed an elevated protein concentration with normal glucose content. There were many mucin-secreting cells and signet ring cell types, which were strongly positive for mucicarmin and alcian blue in the CSF (Figure 4). Symptomatic treatment was initiated with steroids and mannitol. A generalized tonic clonic seizure developed on day 11. The patient became stuporous and did not recover. Finally, the patient was discharged without further medical treatment.
Since the first report of leptomeningeal carcinomatosis (LC) in 1870, the incidence of LC has been increasing. The incidence of LC is three to 8% of all solid cancers. The most common causes of LC are breast and lung cancer and malignant melanoma. Gastric cancer is the most common cancer in Korea; however, LC associated with gastric cancer (GC) is relatively rare. In the Korean medical literature, the incidence of LC with GC has been reported to be 0.06% of all gastric cancers3). Among cases with LC, GC is reported as the most frequent cause of cancer in Korea4, 5). However, these results are based on the experience of one institution, so these findings may not be generalized to the entire Korean population.
The clinical manifestations of LC are very variable and depend on the site of involvement. Many patients complain of headache, nausea, vomiting, ataxia, and have a variety of neurological deficits. Some cases have presented with bilateral progressive blindness6) or bilateral hearing loss7). LC is usually a manifestation of advanced cancer discovered during follow-up after a cancer diagnosis; it is rarely the presenting symptom as it was in this case. The median time to the diagnosis of LC, from the initial cancer diagnosis, is very variable; reports have found a 76 day5) to 17 month4) time interval.. Diagnosis is based on cytology examination of the CSF and imaging studies (especially, magnetic resonance imaging with gadolinium enhancement)8). The CSF findings include an increased opening pressure, elevated protein, the presence of tumor markers (for example, CEA, CA19-9 in gastric cancer), decreased glucose, and pleiocytosis. However, the diagnosis is frequently missed on the first trial but the diagnosis rate increases by 30% on a second one. In our case, the definitive diagnosis was achieved by the first CSF cytologic examination. Chang, et al. reported on patients with breast cancer metastasized to the central nervous system over 20 years. There were 43 cases with 38 parenchymal metastases and 5 leptomeningeal metastases. Out of five LC patients, only one (20%) patient was confirmed cytologically9). Pathologically, poorly differentiated adenocarcinoma with signet ring cell features is the most frequent type of LC associated with GC in Japan10) and this is likely true for Korea3). LC was a more frequent finding in advanced disease with or without distant metastases; it is rarely reported with early gastric cancer11). Imaging studies (MRI with gadolinium enhancement) show abnormalities in 67% of LC patients12).
There are several treatment options for patients with LC including intrathecal (IT) chemotherapy with or without radiotherapy (RT). Most chemotherapeutic agents do not penetrate the blood-brain barrier (BBB), and are given by direct intrathecal administration. Agents that can be used for IT chemotherapy include methotrexate (MTX), cytarabine, thiotepa, and steroids. The benefit (i.e. the prolongation of survival) achieved by IT chemotherapy (single or multiple regimens) in LC patients continues to be debated. The median survival of LC is reported to be four to six months even with aggressive therapy. In one study, the objective response rate and median survival of IT MTX were 20% and 11 weeks; with sustainedrelease cytarabine they were 25% and 15 weeks. Therefore, the results did not confirm a definite benefit with the IT chemotherapy13). However, in a Korean study, there was a favorable outcome with combination IT chemotherapy, compared to the single IT chemotherapy14). MTX, one of most useful agents for IT chemotherapy, penetrates the blood brain barrier poorly in low doses, but can penetrate in the higher dose range. High dose intravenous MTX (3.5 g/m2) injection showed a 28% partial response and stable disease and 44% progressive disease. The median survival of patients with LC (with or without parenchymal disease) was 12.6 weeks; shorter than with isolated parenchymal disease (25.4 weeks)15).
The clinical risk factors that influence the survival of patients with LC are performance status, underlying malignant disease and previous responsiveness to anticancer therapy. The patients with disease that is more advanced, poor performance status (PS), chemoresistant tumors, pronounced neurological clinical findings, and a heavy pretreatment status have a poor prognosis and additional management should be with conservative treatment16). In our case, the patient had a poor PS, pronounced abnormal neurological clinical findings and a very rapidly deteriorating clinical course.
References
1. Larson DA, Rubenstein JL, Mcdermott MW. In: DeVita JVT, Hellman S, Rosenberg SA, eds. Treatment of metastatic cancer. Cancer: principles and practice of oncology 2005. 7th ed. USA: Lippincott Williams & Wilkins, 2333.
2. Lee JL, Kang YK, Kim TW, Chang HM, Lee GW, Ryu MH, Kim E, Oh SJ, Lee JH, Kim SB, Kim SW, Suh C, Lee KH, Lee JS, Kim WK, Kim SH. Leptomeningeal carcinomatosis in gastric cancer. J Neurooncol 2004. 66:167ŌĆō174PMID : 15015782.


3. Kim MH. Intracranial involvement by metastatic advanced gastric carcinoma. J Neurooncol 1999. 43:59ŌĆō62PMID : 10448872.


4. Park JO, Shin HJ, Kim HJ, Lee SW, Jeung HC, Kim SM, Yoo NC, Chung HC, Kim JH, Kim BS, Min JS, Roh JK. Leptomeningeal carcinomatosis in solid tumors: clinical manifestation and treatment. J Korean Cancer Assoc 2001. 33:34ŌĆō40.
5. Kim KW, Kim SM, Kim JS. Clinical features and prognosis of leptomeningeal carcinomatosis. J Korean Neurol Assoc 1989. 7:210ŌĆō217.
6. Kim SH, Koh SB, Lee KW. A case of leptomeningeal metastasis presented with bilateral loss of vision. J Korean Neurol Assoc 1999. 17:780ŌĆō782.
7. Wagemakers M, Verhagen W, Borne B, Venderink D, Wauters C, Strobbe L. Bilateral profound hearing loss due to meningeal carcinomatosis. J Clin Neurosci 2005. 12:315ŌĆō318PMID : 15851093.


8. Braeuninger S, Mawrin C, Malfertheiner P, Schildhaus HU, Seiler C, Dietzmann K, Lins H. Gastric adenocarcinoma with leptomeningeal carcinomatosis as the presenting manifestation: an autopsy case report. Eur J Gastroenterol Hepatol 2005. 17:577ŌĆō579PMID : 15827450.


9. Chang YS, Seo JH, Lee R, Ahn JB, Shim KY, Gong SJ, Lee HY, Rha SY, Yoo NC, Suh CO, Kim JH, Rho JK, Lee KS, Min JS, Kim BS, Chung HC. Brain metastasis and leptomeningeal carcinomatosis in breast cancer. J Korean Cancer Assoc 1998. 30:464ŌĆō474.
10. Lisenko Y, Kumar AJ, Yao J, Ajani J, Ho L. Leptomeningeal carcinomatosis originating from gastric cancer: report of eight cases and review of the literature. Am J Clin Oncol 2003. 26:165ŌĆō170PMID : 12714889.


11. Min KR, Lee SK, Ham JS, Rhee JC, Lee MH, Lee DH, Kee CS, Park KN, Hong EK. A case of leptomeningeal carcinomatosis on superficial spreading carcinoma of the stomach. Korean J Gastrointest Endosc 1986. 6:19ŌĆō23.
12. van Oostenbrugge RJ, Twijnstra A. Presenting features and value of diagnostic procedures in leptomeningeal metastases. Neurology 1999. 53:382ŌĆō385PMID : 10430430.


13. Glantz MJ, Jaeckle KA, Chamberlain MC, Phuphanich S, Recht L, Swinnen LJ, Maria B, LaFollette S, Schumann GB, Cole BF, Howell SB. A randomized controlled trial comparing intrathecal sustainedrelease cytarabine (DepoCyt) to intrathecal methotrexate in patients with neoplastic meningitis from solid tumors. Clin Cancer Res 1999. 5:3394ŌĆō3402PMID : 10589750.

14. Kim DY, Lee KW, Yun T, Park SR, Jung JY, Kim DW, Kim TY, Heo DS, Bang YJ, Kim NK. Comparison of intrathecal chemotherapy for leptomeningeal carcinomatosis of a solid tumor: methotrexate alone versus methotrexate in combination with cytosine arabinoside and hydrocortisone. Jpn J Clin Oncol 2003. 33:608ŌĆō612PMID : 14769837.


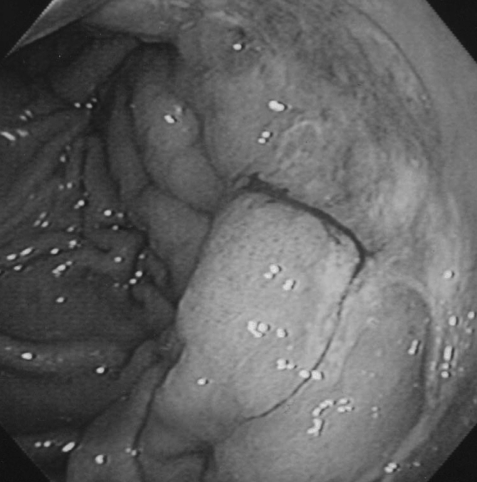
Figure┬Ā1
The gastroscopy showed a discrete ulcer with rugae like fold thickening on the high body and posterior wall of the stomach.
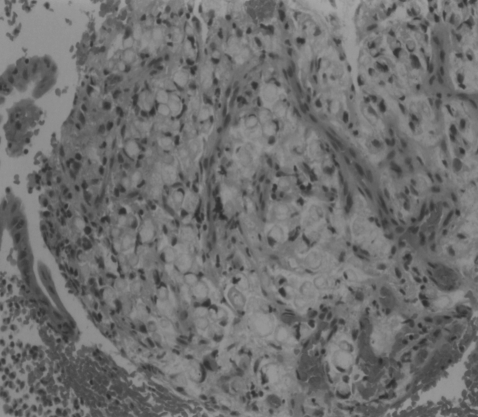
Figure┬Ā2
Diffuse infiltration of tumor cells shaped like a signet ring with intracytoplasmic mucin pooling, pushing the nuclei to the periphery in the gastric mucosa (H&E, ├Ś400).



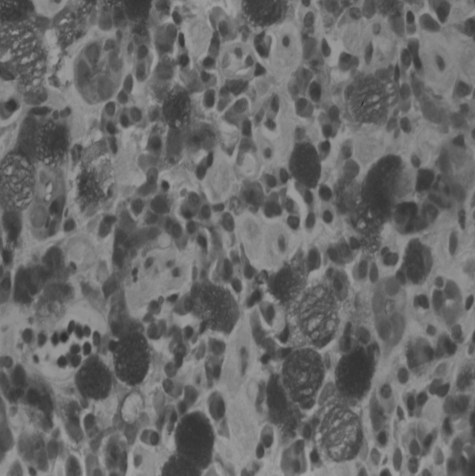
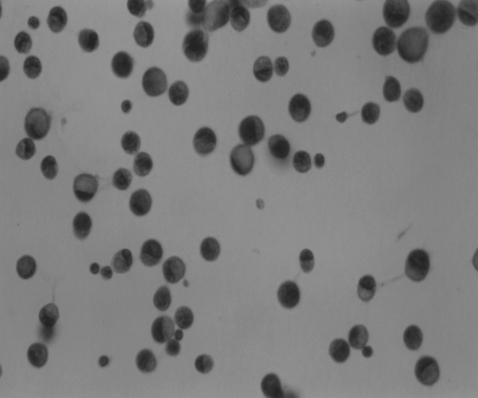
 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



