 |
 |
| Korean J Intern Med > Volume 24(1); 2009 > Article |
|
Abstract
Background/Aims
Risk factors for mortality resulting from anaerobic infection are incompletely defined. The clinical significance of a broad range of pathogenic obligate anaerobic organisms was examined, and factors independently associated with mortality were identified in patients with clinically significant anaerobic infections.
Methods
The medical records of 1,050 patients with anaerobic infections were retrospectively reviewed at Severance Hospital in Seoul, Korea.
Results
The mean age of the patients was 54.1┬▒16.8 years, and 57.7% were men. Overall, 320 (30.5%) patients with case-defined illness experienced pain at the affected site, and 230 (21.9%) experienced pus flow from lesions. Ten (1.4%) patients presented with shock, and 80.3% of the clinically significant cases were polymicrobial anaerobic infections. The mean number of pathogens, including aerobic and anaerobic bacteria, was 3.7┬▒1.0 (minimum 1, maximum 5), and the number of anaerobic organisms was 1.0┬▒0.3 in each specimen. The major pathogens by rank were the Bacteroides fragilis group, which accounted for 41.8% of anaerobic infections, followed by Clostridium spp. (11.8%), Prevotella spp. (9.4%), and Peptostreptococcus spp. (8.4%). Escherichia coli (17.5%), Staphylococcus aureus (7.5%), and Klebsiella pneumoniae (7.5%) were common concomitant aerobic organisms. The overall crude mortality rate resulting from anaerobic infection was 29.7%. Among the determining factors associated with mortality, liver disease (p=0.003) and old age (p=0.005) were significant in multivariate analysis.
Anaerobic bacteria can cause a variety of endogenous infections [1]. Because of their persistent nature, they are difficult to isolate from infection sites and are often overlooked [2]. Moreover, anaerobes play a key role in maintaining the balance between the host and its colonizing organisms; anaerobic infections occur when the harmonious relationship between the host and bacteria has been disrupted. Mixed infections may arise in the head and neck region (e.g., chronic sinusitis, chronic otitis media, Ludwig's angina, and periodontal abscesses) [3]. Brain abscesses and subdural empyema are the most common anaerobic infections of the central nervous system [4,5]. Anaerobes are also responsible for pleuropulmonary diseases, such as aspiration pneumonia, necrotizing pneumonia, lung abscess, and empyema [6]. In addition, these organisms play an important role in various intra-abdominal infections, such as peritonitis and intra-abdominal and liver abscesses. They are frequently isolated in female genital tract infections, such as salpingitis, pelvic peritonitis, tuboovarian abscess, vulvovaginal abscess, septic abortion, and endometritis. Anaerobic bacteria are also often found in bacteremia and infections of the skin, soft tissues, and bones [1,2]. Their isolation requires appropriate methods of collection, transportation, and cultivation of specimens [7]. Treatment is complicated by their slow growth, polymicrobial nature, and growing resistance to antimicrobial agents. Antimicrobial therapy is often the only form of treatment required, or may be an important adjunct to a surgical approach. Because anaerobes are generally recovered in association with aerobic organisms, the choice of antimicrobial agents should provide coverage for both types of pathogens [8].
Despite a relatively low incidence, anaerobic infection remains associated with significant mortality rates. Recent estimates of case fatality rates in patients with clinically significant anaerobic bacteremia range from 25% to 44% [9]. The aims of this study were to determine the clinical features of a broad range of obligate anaerobic organisms isolated from, for example, blood, cerebrospinal fluid, peritoneal fluid, and pleural fluid, and to define the factors independently associated with mortality.
The study was conducted at Severance Hospital at Yonsei University College of Medicine in Seoul, Korea, from January 1996 to December 2003. The facility is a 1,544-bed, tertiary care referral center with hematopoietic stem cell and solid organ transplant specialty services. This study examined 1,050 consecutive adult patients (>17 years old), with 3,169 specimens from sterile fluid and abscess cultures for anaerobic bacteria that were retrospectively identified from clinical microbiology records. Following the first positive anaerobic culture, outcome data were collected from the patients' medical and clinical microbiology laboratory records for 30 days, until discharge or death. The data were based on mortality within 30 days following the first positive blood culture.
The primary endpoint was death, recorded as the number of days after detection of the positive culture results for anaerobic bacteria. The following variables were assessed: personal information (i.e., age and sex), dates of admission and discharge, mode of acquisition (nosocomial or community-acquired), type of infection (polymicrobial or monomicrobial), source of infection, culture results, types of surgeries and procedures performed during hospitalization, diagnosis at discharge, and presence of concurrent underlying disease (e.g., heart, liver, lung, or kidney disease, hypertension, diabetes, malignancy, immunosuppression). The presence of an underlying disease was based on the description made by the physician. Each disease variable was similarly defined; a patient was only considered to have a disease if it was documented as ongoing at the time of the positive culture results. Immunosuppression was said to be present if the patient had a history of solid organ transplant, AIDS, pregnancy, or was undergoing high-dose steroid therapy.
Specimens for anaerobic cultures were primarily biopsy samples or needle aspirates; swabs made up only a minor part of the specimens. All specimens were transported and incubated in anaerobic systems. Colonies present on blood agar media were evaluated through aerotolerance tests, catalase tests, Gram staining, and susceptibility tests. Sources of infection were determined by radiological, surgical, or microbiological evidence of barrier compromise or infectious pathology, such as abscess or necrosis. The origin of bacteremia was also defined by radiological, surgical, or microbiological evidence. A case was designated bacteremia if no other proven focus was discovered.
In this study, no evidence of anaerobic infection was detected in brain abscess specimens. Based on the National Nosocomial Infection Surveillance System (NNISS) guidelines, the infection was deemed to be nosocomial if a positive culture was drawn more than 48 h following admission to the hospital [10]. Appropriate coverage was defined as culture-directed antibiotic coverage given at adequate doses. Inappropriate coverage was defined as non-culture-directed coverage at inadequate doses, but was deemed acceptable if it was non-culture-directed (especially when cultures were negative) but showed signs of clinical improvement.
The results were expressed as the mean┬▒standard deviation (SD), or as a proportion of the total number of patients or isolates. For continuous variables, mean values were compared using a two- sample t-test for independent samples. Differences in proportions were compared using a Žć2 test. All tests of significance were two-tailed and ╬▒ was set at 0.05. Mortality was evaluated using univariate and multivariate Cox proportional hazard models. All statistical analyses were performed using the SPSS software program (version 12.0; SPSS Inc., Chicago, IL, USA).
In total, 3,169 anaerobic organisms from 1,050 patients were reported. Patients with clinically significant anaerobic infections ranged in age from 18 to 88 years (median, 58.0 years; mean, 54.1┬▒16.8 years), and 57.7% of the patients were men. The most frequent underlying conditions (diagnosed at admission) were malignancy in 343 patients (32.7%), diabetes mellitus in 132 patients (12.6%), renal disease in 123 patients (11.7%), and liver disease in 88 patients (8.4%). Other underlying diseases included renal or ureter stones, skin fistula, Ebstein anomaly, deep vein thrombosis, and plegia. One hundred and fifty-seven (15.0%) patients who had undergone predisposing surgery were identified, and their mode of acquisition was regarded to be of nosocomial origin. In addition, more than half (56%) of those surgeries were performed on the abdomen.
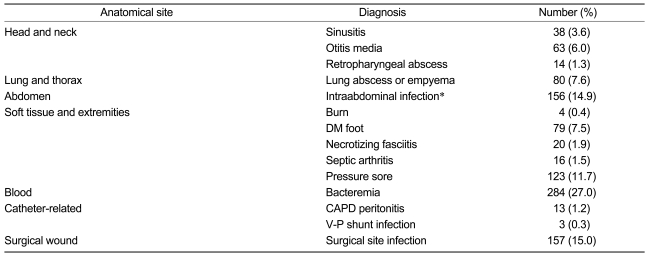
Information on the symptoms of anaerobic infection was available for 1,008 (96%) of 1,050 cases. Overall, 320 (30.5%) of those with case-defined illnesses experienced pain at the affected site, and 230 (21.9%) complained of discharged pus from lesions. Ten (1.4%) patients presented with shock. The mean duration of illness was 20.0┬▒23.8 days. Various diagnoses were derived from anaerobic infections (Table 1).
Each of the 38 and 63 specimens from patients with sinusitis and chronic suppurative otitis, respectively, were obtained by operative procedures; some were treated with antibiotics, having no anaerobic coverage. Consequently, a complete resection of the infected source was sufficient to cure, with no additional antimicrobials.
In addition, 80 consecutive adult cases of lung abscess and empyema were treated. Seventy-three percent of these patients had a history of prior antibiotic use. The most commonly administered antibiotic was cefoperazone/sulbactam at 40% (n=32), followed by vancomycin. Treatment of these infections included pleural space drainage (in the presence of empyema) and antimicrobial agents.
Fifty-eight patients with intra-abdominal infection (37.2%) died during treatment. Appropriate management of mixed intra-abdominal infections requires the administration of antimicrobials that are effective against both aerobic and anaerobic components of the infection, and as well as surgical correction and drainage of pus. Combination therapies including aminoglycoside, quinolone, or a third-generation cephalosporin plus an anti-anaerobic agent, such as clindamycin or metron-idazole, were used. Single-agent therapy included the use of a carbapenem or piperacillin-tazobactam.
Twenty cases of necrotizing fasciitis were treated. The paucity of cutaneous findings early in the course of this disease makes diagnosis difficult; four patients were diagnosed within a few hours of receiving acupuncture on the buttocks. Twelve of 20 patients died from necrotizing fasciitis, but only one of those who underwent debridement died.
Abscess of the buttocks and pressure sores comprised 123 cases; a few simple abscesses did not require antibiotics, and some cases resolved through simple aspiration or drainage. The cases of otitis media reviewed here did not require antibiotics, although some required surgical intervention, such as aspiration or drainage. Cultured anaerobes from intracerebral abscesses were not observed.
Polymicrobial anaerobic infections represented 80.3% of all clinically significant cases. The mean number of pathogens, including aerobes and anaerobes, was 3.7┬▒1.0 (minimum 1, maximum 5), and the mean number of anaerobic organisms was 1.0┬▒0.3 in each specimen. The most common pathogens by rank were Bacteroides fragilis, which accounted for 41.8% of anaerobic infections, Clostridium spp. (11.8%), Prevotella spp. (9.4%), and Peptostreptococcus spp. (8.4%). Escherichia coli (17.5%), Staphylococcus aureus (7.5%), and Klebsiella pneumoniae (7.5%) were common concomitant aerobic organisms (Table 2).
The most common anaerobic pathogen in 38 sinusitis patients was Propionibacterium, whereas the remaining sinusitis patients showed Peptostreptococcus and Bacteroides spp. Sixty-three cases involved chronic suppurative otitis media and the predominant anaerobes recovered were Peptostreptococcus and Bacteroides spp.
Lung abscess and empyema comprised 80 consecutive adult cases. The predominant anaerobes in these cases were Bacteroides, Prevotella, Fusobacterium, and Peptostreptococcus spp.
One hundred and fifty-six cases of intra-abdominal infections and nosocomial infections were suspected in 82 patients (51.3%). The predominant aerobic and facultative microbes were E. coli and Streptococcus spp. (including Enterococcus spp.); the most frequently encountered anaerobes were of the B. fragilis group and Peptostreptococcus and Clostridium spp.
Abscess of the buttocks and pressure sores were detected in 123 cases. The common pathogens were B. fragilis, Peptostreptococcus asaccharolyticus, and Bacteroides thetaiotaomicron, in order of the frequency of occurrence.
The only anaerobe in all four burn wounds was Peptostreptococcus. Two hundred and eighty-four cases involved bacteremia due to anaerobes, and the most frequent site of origin was the gastrointestinal tract.
All isolated strains of B. fragilis were sensitive to metronidazole, but the resistance rate was 43% to clindamycin, 33% to cefotaxime, and 25% to piperacillin.
Overall, the crude mortality rate was 29.7% (312 of 1,050 cases). The mean and median times to death were 10.7 and 3.5 days, respectively (range, 0.4-37 days). Twelve of 20 (60%) patients died from necrotizing fasciitis, but only one of the patients who underwent debridement subsequently died. Despite sufficient therapy, half of the patients (50%) suffering from pressure sores died. Fifty-eight patients (37.2%) died during the treatment of intra-abdominal infections. No recorded death was due to sinusitis or otitis media during the study period. In patients with anaerobic bacteremia, adequate surgical intervention resulted in a higher survival rate than in cases without appropriate intervention procedures (81.8% versus 57.1%, p<0.05). As expected, the incidence of death was higher among those who received inappropriate antibiotic therapy (32.5%) compared to those who received appropriate therapy (27.3%) as well as acceptable antibiotic coverage.
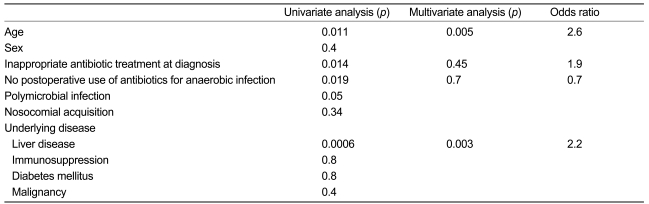
Each variable was evaluated using a Cox proportional hazard model to determine factors associated with mortality. The four discrete factors were increasing age per year over 18 years (p=0.011), inappropriate prescription of antibiotics at diagnosis (p=0.014), no use of postoperative antibiotics (p=0.019), and underlying liver disease (p=0.0006). In particular, liver disease (p=0.003) and old age (p=0.005) were significantly associated with mortality during multivariate Cox proportional hazard analysis (Table 3).
Neoplasms, hematologic disorders, organ transplant, recent surgery, diabetes mellitus, and the use of cytotoxic agents or corticosteroids are predisposing factors for anaerobic infection [2]. This study also showed that anaerobic infections often occurred in patients with cancer when the balance between the host and bacteria was disrupted.
In many patients, pain and pus discharge at the affected site were the chief complaints, whereas few patients presented with shock. Consistent with the findings of Cheon et al. [11], the most common presenting symptom in bacteremia patients was fever (70%).
The appropriate management of anaerobic infection depends on suspicion and appropriate documentation of the bacteria causing infection [12]. In this study, inappropriate anaerobic antibiotics prescriptions at the time of diagnosis (p=0.014) and the lack of postoperative antibiotics (p=0.019) were associated with a poor prognosis resulting in mortality. Regarding the use of antibiotics, Hecht noted that antibiotic-resistant anaerobic bacteria have become increasingly recognized as a confounding factor in the selection of therapeutic agents [13]. Although appropriate therapy for anaerobic infections has been associated with significant reductions in mortality, most clinical laboratories still do not perform routine anaerobic susceptibility tests [14,15]. In a previous 8-yr study, resistance to antimicrobial agents, such as piperacillin, cefotaxime, and clindamycin, was not uncommon among 913 clinical isolates of B. fragilis group organisms, and antimicrobial susceptibility testing was deemed necessary before using one of these drugs [16]. Thus, concentrating on an appropriate narrow-spectrum of antibiotics is important.
Procedures such as abscess drainage, necrotic tissue debridement, and closed-space infection decompression are important adjuncts to a medical approach [12]. However, this study did not find a correlation between surgical intervention and mortality.
Shin et al. [17] reported that B. fragilis and Peptostreptococcus magnus were most frequently isolated in anaerobic bacterial infections. However, in this study, the most common pathogens were B. fragilis (41.8%) followed by Clostridium spp. The most common concomitant aerobic isolate was E. coli, which is why a relationship between the development of intestinal tract cancer and intra-abdominal infection was postulated, although the results reported here were not statistically significant.
This study, which examined patients with infections due to a broad range of anaerobic pathogens, found that age and liver disease were strong, independent risk factors for mortality in multivariate analysis. Crude rather than attributable mortality was reported because the relative contribution of anaerobic infections to the overall mortality rate could not be reliably ascertained. The data demonstrate a 29.7% crude mortality rate for clinically significant anaerobic infections. The majority of previous studies on patients with anaerobic infections, which included a broad range of anaerobic pathogens, also reported mortality as crude mortality rates and published estimates ranging from 25% to 44% [9,18]. In several cases, mortality was assessed with reference to discharge from the hospital, or the interval of antibiotic treatment [18].
The present study provides a recent perspective on the clinical significance of infections due to a broad range of anaerobic species. When combined with careful clinical assessment, this information may be helpful in determining the clinical significance of cultures with anaerobic organisms. The strength of this study is the diversity of the study population and the relatively large sample size examined.
The present study, however, has certain limitations. First, as with any retrospective review, potential for selection bias and data omission exists. Second, not all analyzed variables were necessarily included in the present study; thus, both liver disease and the severity of illness should be included as independent variables in future studies to confirm their association with mortality. Despite these limitations, strong and independent associations were demonstrated among age, underlying liver disease, and mortality.
References
1. Henges DJ. The anaerobic microflora of the human body. Clin Infect Dis 1993. 16(Suppl 4):S175ŌĆōS180PMID : 8324114.


2. In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett's Principles and practice of infectious diseases 2005. 6th ed. Philadelphia: Elsevier Inc..
3. Brook I. Anaerobic bacteria in upper respiratory tract and other head and neck infections. Ann Otol Rhinol Laryngol 2002. 111:430ŌĆō440PMID : 12018328.


4. Le Moal G, Landron C, Grollier G, et al. Characteristics of brain abscess with isolation of anaerobic bacteria. Scand J Infect Dis 2003. 35:318ŌĆō321PMID : 12875518.


5. Kao PT, Tseng HK, Liu CP, Su SC, Lee CM. Brain abscess: clinical analysis of 53 cases. J Microbiol Immunol Infect 2003. 36:129ŌĆō136PMID : 12886965.

6. Levison ME. Anaerobic pleuropulmonary infections. Curr Opin Infect Dis 2001. 14:187ŌĆō191PMID : 11979131.


7. Holden J. Collection and transport of clinical specimens for anaerobic culture. Clinical microbiology procedures handbook 1996. Washington DC: American Society for Microbiology, 2.2.1-2.2.6.
9. Salonen JH, Eerola E, Meurman O. Clinical significance and outcome of anaerobic bacteremia. Clin Infect Dis 1998. 26:1413ŌĆō1417PMID : 9636872.


10. Horan TC, Gaynes RP, Martone WJ, Jarvis WR, Emori TG. CDC definitions of nosocomial surgical site infections, 1992: a modification of CDC definitions of surgical wound infections. Infect Control Hosp Epidemiol 1992. 13:606ŌĆō608PMID : 1334988.


11. Cheon SH, Kim JM, Kim E, Hong CS. Clinical analysis of bacteremia due to anaerobic organisms. Infection 1989. 21:87ŌĆō94.
12. Brook I. Management of anaerobic infection. Expert Rev Anti Infect Ther 2004. 2:153ŌĆō158PMID : 15482180.


13. Hecht D. Prevalence of antibiotic resistance in anaerobic bacteria: worrisome developments. Clin Infect Dis 2004. 39:92ŌĆō97PMID : 15206059.


14. Nguyen MH, Yu VL, Morris AJ, et al. Antimicrobial resistance and clinical outcome of Bacteroides bacteremia: findings of a multicenter prospective observational trial. Clin Infect Dis 2000. 30:870ŌĆō876PMID : 10852736.


15. Goldstein EJ. Anaerobes under assault: from cottage industry to industrialization of medicine and microbiology. Clin Infect Dis 1995. 20(Suppl 2):S112ŌĆōS116PMID : 7548530.


16. Lee K, Shin HB, Chong Y. Antimicrobial resistance patterns of Bacteroides fragilis group organisms in Korea. Yonsei Med J 1998. 39:578ŌĆō586PMID : 10097686.


17. Shin HJ, Kim MY, Lee K, Chong Y. Trends of anaerobic bacteria isolated from clinical specimens. Korean J Clin Pathol 1999. 19:70ŌĆō77.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



