INTRODUCTION
The prevalence of diabetes in Korea is reported to be approximately 10% and most patients have type 2 diabetes mellitus1). Maturity-onset diabetes of the young (MODY) is a heterogeneous form of diabetes characterized by onset at an early age, impaired insulin secretion, and inheritable as an autosomal dominant trait for more than three generations.
MODY has numerous subtypes, depending upon the gene that is involved: MODY1 is associated with chromosome 20; MODY2, with chromosome 7; MODY3, with chromosome 12; MODY4, with chromosome 13; MODY5, with chromosome 17; and MODY6, with chromosome 2. The MODY3 gene encodes hepatocyte nuclear factor (HNF)-1╬▒, which is a homeodomain-containing transcription factor required for tissue-specific expression of a variety of genes in the liver, kidney, pancreas (including the Islets of Langerhans), intestine, stomach, spleen, and thymus2, 3). Although mutations of the MODY2 gene (glucokinase) are the most common mutations in French MODY patients, MODY3 (HNF-1╬▒) mutations are the most common mutations in Western and Asian countries4-7). Nevertheless, only a few cases of MODY have been reported in Korea.
Diagnostic techniques for autosomal dominant MODY are currently being sought. In this study, we used a DNA chip to investigate the frequency of MODY3 mutations in Korean patients with early-onset type 2 diabetes.
MATERIALS AND METHODS
Study subjects
Thirty normal individuals [age (mean┬▒SD), 24.9┬▒8.6 years] who had no family history of diabetes and who showed a normal serum glucose level in a 75-g oral glucose tolerance test (OGTT) were enrolled in this study as controls. We also enrolled 25 patients (age, 27┬▒5.9 years) who had been diagnosed with early-onset diabetes before the age of 35 years7). The diagnostic criteria for type 2 diabetes followed those of the 57th ADA Conference held in Boston in June 19978). Glucose values of <110 mg/dL (6.1 mmol/L) and <140 mg/dL (7.75 mmol/L) were used as the normal fasting plasma glucose level and normal 2-h glucose level after a 75-g OGTT, respectively. The normal C-peptide level was >0.6 ng/mL. Insulin and glutamic acid decarboxylase auto-antibodies were noted at diagnosis. We measured the height, weight, body mass index (BMI), and blood pressure for each subject, and determined the fasting glucose level, glucose level 2-h after a 75-g OGTT, and insulin and C-peptide levels in both the normal and early-onset type 2 diabetes groups.
Nucleotide sequencing
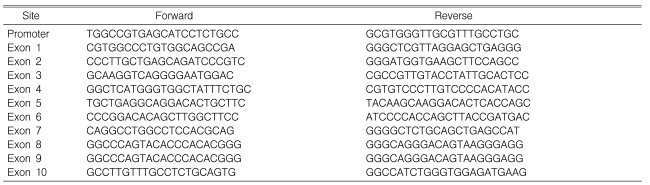
From each subject, a blood sample (4 mL) was collected into a tube containing EDTA. Genomic DNA was extracted from the whole blood using a genomic DNA extraction kit (Promega, Madison, WI, USA). Single or multiplex PCR amplification of the HNF-1╬▒ gene was carried out using 10 different primer sets (Table 2). Direct sequencing of the PCR products was performed using an ABI Dye Terminator Cycling Sequencing kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer's instructions, followed by analysis on an ABI3700 DNA sequencer (Applied Biosystems)12).
Site-directed mutagenesis
Site-directed mutations were introduced into MODY3 DNA by using a PCR method and primers modified for the specific mutations (Bioneer, Seoul, Korea). The PCR products were inserted into plasmids, which were used to transform bacteria. The plasmid DNA was purified using a commercial kit (Qiagen, Valencia, CA, USA), and the mutations were confirmed by direct sequencing13).
PCR and hybridization
For PCR, the reaction mixture contained 0.2 ┬Ąg of either wild-type or mutant DNA, 100 ┬ĄM of each dNTP (Amersham Biosciences, Uppsala, Sweden), and 100 nM each of five multiplex primer sets: one set for mutations in the promoter region and exons 2, 4, and 7, and four sets for mutations in exons 1, 3, 5, 6, 8, 9, and 10. The PCR conditions were 35 cycles of 95Ōäā for 30 s, 64Ōäā for 15 s, and 72Ōäā for 3 min. The PCR product was purified using a Qiagen kit, and the purified PCR product (A260/A550 = 1.0-3.5) was used for hybridization.
The PCR product was diluted to 150-187.5 nM, mixed with Cy3-labeled tag oligonucleotide, denatured at 94Ōäā for 5 min, cooled on ice, and then mixed with 30 ┬ĄL of hybridization buffer [6├ŚSSPE (0.15 M NaCl, 10 mM NaH2PO4, 1 mM EDTA, pH 8.0), 0.1% Triton X-100, 25% formamide]. A HybriWell (Molecular Probes, Eugene, OR, USA) was sealed to a Gen-Spector┬« MODY3 chip (2.54 ├Ś 7.62 cm; Samsung, Yongin, Korea), which has oligonucleotides of 15-25 bases, representing wild-type and mutant probes in both forward and reverse orientations, immobilized on its surface. Sixty microliters of PCR fragment-hybridization buffer mixture were added to the HybriWell through a hole in the sealer, the air bubbles were removed, and the hole was covered with a stopper. Hybridization was performed at 48Ōäā for 4 h, followed by washing with 3├ŚSSPE/0.005% Triton X-100 buffer and then 1├ŚSSPE/0.005% Triton X-100 buffer. The chip was dried for 15 min at room temperature.
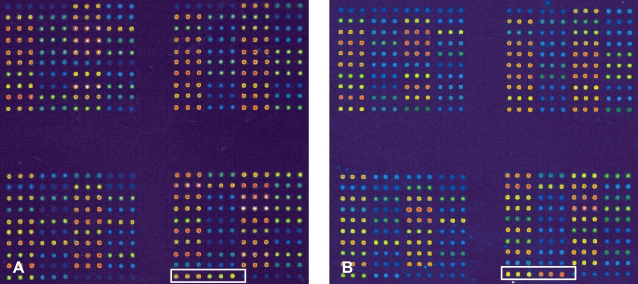
The hybridization signals were detected by scanning the array using a GenePix 4000B scanner (Axon Instruments, Union City, CA, USA), and digitized images were produced using GenePix Pro software (Axon) (Figure 2). The data were analyzed using a module of the GenSpector® TMC-1000 system (Samsung). The probability that a sample represented a specific mutant was calculated using an algorithm based on an MA-plot determined in preliminary experiments using wild-type and mutagenized DNA samples. A sample was considered to harbor a particular mutation if the probability that the sample belonged to the specific mutant was >99.9%. A sample was considered wild-type for the mutation if the probability that the sample belonged to the specific mutant was <0.001%. The call rate was 95%, and the remaining 5% of the spots were in the gray zone. The accuracy was 99.7%, and the reproducibility was 99.8%.
RESULTS
In the normal group (n = 30), both the fasting serum glucose level (93.8┬▒11.2 mg/dL) and the 2-h glucose level after a 75-g OGTT (95.6┬▒20.3 mg/dL) were normal. The insulin level was 7.7┬▒2.3 ┬ĄIU/mL when fasting and 33.4┬▒25.3 ┬ĄIU/mL at 2 h after a 75-g OGTT; the respective C-peptide levels were 2.3┬▒2.53 and 8.5┬▒4.6 ng/mL. These values are consistent with normal insulin secretion.
In the early-onset type 2 diabetes patients (n = 25) who were treated with oral anti-diabetic agents, the basal C-peptide level was 3.6┬▒4.55 ng/mL, and the C-peptide level at 2 h after a 75-g OGTT was 7.8┬▒8.1 ng/mL (Table 1).
We used DNA chips to detect mutations of the HNF-1╬▒ gene in patients and normal controls. No mutations were detected in any control subject. One patient in the diabetic group had a mutation (Figure 2). Direct sequencing of the HNF-1╬▒ mutation in this patient revealed a C substituted for a G in the promoter region of the gene (5'-ctaGGCTAGTGGGGTTTTGCGG GGGC AGTGGGTGCAAGG-3'). The patient with the mutation, a woman, had been diagnosed with diabetes at 25 years of age. Her plasma glucose levels were 151 and 273 mg/dL prior to and 2 h after the intake of 75 g of glucose, respectively. Her serum basal C-peptide level was 3.83 ng/mL, and her BMI was 22 kg/m2 at the time of diagnosis. She is now controlling her diabetes with diet, physical activity, and metformin. Her hemoglobin A1c level is being maintained at 6.5%. Her grandmother and father had diabetes mellitus, but their blood samples were not available for genotyping.
DISCUSSION
PCR techniques for genetic analysis were developed in the early 1990s and have enabled the identification of genes involved MODY. DNA microarray technology is a new automated method for obtaining large amounts of information in a few hours. The quick and accurate diagnosis of disorders caused by genetic mutations is an expected clinical application of DNA microarray technology. The development and application of DNA chips for specific genetic disorders would form the foundation of personal preventive medicine. A DNA chip could be used to rapidly, conveniently, and accurately predict the genetic predisposition of an individual. With classical methods of gene sequencing analysis, identifying MODY mutations takes more than 3 days; however, with the MODY3 chip (Figure 1), MODY can be diagnosed in only 0.5 days9-11). Using a MODY3 chip, more than 70 reported mutation sites in the HNF-1α gene can be screened with >99.7% accuracy, using our in-house-developed diagnostic software, GenSpector® Analyzer.
MODY accounts for up to 3% of diabetes cases in most Caucasian populations6, 14-19). The MODY gene on chromosome 12q has been shown to encode HNF-1╬▒, which had not previously been known to have a role in the functioning of pancreatic ╬▓ cells. Mutations in HNF-1╬▒ (MODY3) are the most common cause of MODY and are thought to be the result of a hotspot for spontaneous mutations in the gene, rather than the result of a single distantly related family mutation. Among families with a HNF-1╬▒ mutation, 15% have an insertion of an extra nucleotide (C) in the G tract in exon 4, which leads to a frameshift14, 19, 20).
The clinical symptoms associated with HNF-1╬▒ mutations are markedly different from those associated with glucokinase mutations21). With HNF-1╬▒ mutations, the onset of hyperglycemia occurs in adolescence or early adulthood and may be progressive and more severe, with more frequent microvascular complications. In contrast, with glucokinase mutations, hyperglycemia begins in early childhood (from birth) and is mild, with minor age-related deterioration; microvascular complications occur more rarely than with HNF-1╬▒ mutations.
Although the prevalence of MODY3 has been reported for many countries, the prevalence of MODY3 in type 2 diabetes has not been reported for Korea. If the prevalence and inheritance of MODY3 in Koreans were known, accurate diagnosis and treatment may be possible, and the risk for MODY3 could be predicted in normal individuals born into families with a history of diabetes. Knowledge of the frequency of MODY3 among Koreans would aid in the accurate and early diagnosis of diabetes, thus minimizing diabetic complications and early-onset diabetes.
In the present study with 25 early-onset type 2 diabetes patients, the prevalence of HNF-1╬▒ mutations was 4%. There have been three other reports regarding mutations of the HNF-1╬▒ gene in Koreans. Lee et al.7) identified a mutation in exon 4 (C900A), which occurred in only one of 69 Korean subjects with early-onset type 2 diabetes; this mutation was silent, although it did cause an amino acid substitution at Pro300. Kim et al.22) identified the HNF-1╬▒ R267L mutation in one of 16 unrelated patients who had been diagnosed with type 2 diabetes at <35 years of age and who had autosomal dominant inheritance of type 2 diabetes in at least two generations. Shin et al.23) reported that the overall frequencies of HNF-1╬▒ and glucokinase gene mutations were 5 and 2.5%, respectively, in 40 Korean adolescent patients with type 2 diabetes and a strong family history of diabetes.
In our opinion, the prevalence of MODY3 in early-onset type 2 diabetes in Korean patients is 1.4~6.4%. Thus, MODY 3 is not rare in early-onset type 2 diabetes, and MODY mutations in patients with early-onset type 2 diabetes need to be further evaluated.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





