 |
 |
| Korean J Intern Med > Volume 21(3); 2006 > Article |
|
Abstract
Multiple myeloma usually shows homogeneous enhancement on contrast-enhanced Magnetic Resonance imaging (MRI), and is accompanied by a monoclonal gammopathy in serum or urine. We report a case of nonsecretory myeloma, the diagnosis was difficult due to the absence of a monoclonal gammopathy and the presence of atypical imaging features.
Nonsecretory myeloma (NSM) is a rare variant of multiple myeloma that has no serum or urine monoclonal gammopathy on electrophoresis or immunoelectrophoresis. Although a monoclonal gammopathy is absent, NSM usually has the same clinical and radiologic findings as is found in classic multiple myeloma1-3). Classic multiple myeloma as well as NSM show homogeneous enhancement on MRI obtained after intravenous contrast injection4-9). An atypical enhancement pattern, of NSM on contrast-enhanced MRI, has not been reported previously. We here report a case of NSM, the diagnosis was made difficult due to atypical MR features and the absence of a monoclonal gammopathy.
A 45-year-old man was admitted to our hospital because of a one month history of fatigue and severe back pain for two days prior to presentation. There were no specific findings on physical examination. Results of routine laboratory tests showed a mild anemia (hemoglobin, 11.4 g/dL; normal range, 14~18 g/dL). A corrected reticulocyte count was slightly decreased (0.4%; normal range, 0.5~2.5%), suggesting impaired marrow synthesis of erythrocytes. The albumin to globulin ratio (A/G ratio) was normal (A/G ratio, 1.43; albumin, 3.0 g/dL; globulin, 2.1 g/dL). The serum C-reactive protein was markedly increased (151.0 mg/dL; normal range, 0.0~0.5 mg/dL). The serum creatine and total calcium levels were normal. The serum and urine protein electrophoreses, and immunofixation electrophoreses to detect protein more than 20 mg/dL, were repeatedly negative. The level of ╬▓2 microglobulin was also normal.
There was no evidence of lytic bone lesions on plain radiograph. Contrast enhanced chest CT obtained on admission showed multiple, well-defined, osteolytic lesions at the 8th and 11th thoracic vertebrae (Figure 1A). MRI showed the lesions to be hypointense on T1-weighted image (500/15) (Figure 2A) and heterogeneously hyperintense on T2-weighted image (1800/90) (Figure 2C). Although these were very small foci of enhancement, at the lesions of 8th and 11th thoracic vertebrae, most of the lesions were poorly enhanced on T1-weighted image (Figure 2B) after intravenous administration of gadopentetate dimeglumine (Magnevist; Schering, Seoul, Korea). The radiological differential diagnosis was multiple myeloma, metastasis and tuberculosis. Two days after the MRI, a CT-guided gun biopsy was performed on the poorly enhancing portion of the lesion at the 8th thoracic vertebra. The biopsy showed a coagulation necrosis within a population of uniform, monotonous cells (Figure 1B). However, the definite diagnosis was not made at that time because the pathologist considered this finding to be nonspecific. Polymerase chain reaction (PCR) was performed with part of biopsy specimen obtained from the 8th thoracic vertebra using an M tuberculosis kit (Amplicor; Roche Diagnostic Systems, Somerville, NJ), which amplified part of the 16S rRNA gene. The result of the PCR was positive for M tuberculosis. Although pathologic confirmation of tuberculous granuloma with caseation necrosis was not proven, a presumptive diagnosis of tuberculous spondylitis was made. Combination chemotherapy with isoniazid (Yuhan-zid; Yuhan, Seoul, Korea), rifampin (Yuhan), etambutol (myambutol; Yuhan), and pyrazinamide (Yuhan) was started. Twenty-five days after the biopsy, the patient complained of more severe and constant back pain. A thoracolumbar roentgenogram showed compression fractures of the 8th and 11th thoracic vertebral bodies. Follow-up CT examination showed a new osteolytic lesion (Figure 3B), with a well-defined outer margin and cortical disruption, at the junction of the spinous process and the lamina of the 12th thoracic vertebra; this lesion showed strong enhancement after intravenous administration of contrast media. On a retrospective review of the previous CT studies, this lesion (Figure 3A) had been overlooked. The next day, a CT guided gun biopsy of the lesion at the 12th thoracic vertebra was performed. Histological examination of the biopsy tissue showed a proliferation of plasma cells (Figure 3C). There was a strong positive reaction for the lambda light chain (Figure 3D). On the basis of these findings, we considered that the lesion at the 8th thoracic vertebra was also a plasma cell lesion with coagulation necrosis. The tissue from a bone marrow biopsy of the right posterior iliac bone showed 40% plasmacytosis. A diagnosis of a stage I nonsecretory myeloma was made. Combination chemotherapy with intravenous adriamycin (Il-dong Pharmaceuticals, Seoul, Korea), vincristine (Boryung Pharmaceuticals, Seoul, Korea), and dexamethasone (Il-sung Pharmaceuticals, Seoul, Korea) was started in a dose of 13.5 mg, 0.4 mg and 400 mg per day for four days, respectively.
Minimal criteria for the diagnosis of multiple myeloma include the presence of at least 10% abnormal plasma cells in the bone marrow or histological proof of a plasmacytoma, the standard clinical features of multiple myeloma, and at least one of following abnormalities: monoclonal serum protein (usually greater than 3 g/dL), monoclonal protein in the urine, or osteolytic lesions10).
NSM is a rare variant of multiple myeloma that has no serum or urine monoclonal gammopathy on electrophoresis or immunoelectrophoresis. Since protein electrophoresis is usually the initial diagnostic tool for multiple myeloma, its diagnosis is difficult to achieve with the nonsecretory variant. Therefore, a thorough understanding of the full spectrum of multiple myeloma should be helpful to make a correct diagnosis of NSM. NSM has a tendency for a longer survival1). There are two types of NSM. In the first form, there is the production, but no secretion of immunoglobulin by plasma cells, while in the second form there is the absence of production of immunoglobulins3). Our case had findings consistent with the first type of NSM because of the lambda chain production as demonstrated by immunohistochemical staining.
The false positive result of the PCR for M tuberculosis in the biopsy specimen led to the incorrect initial diagnosis. Therefore, direct anti-tuberculous chemotherapy, based only on a PCR result without a definite histological or bacteriologic confirmation for tuberculosis, is inappropriate.
Although NSM lacks a monoclonal gammopathy, its clinical, laboratory, and radiologic findings are similar to the classic form of multiple myeloma. Multiple osteolytic lesions in the spine, showing hypointensity on T1-weighted images, hyperintensity on T2-weighted images, and homogeneous enhancement on contrast-enhanced T1-weighted images are the typical presentation in patients with multiple myeloma4). There have been no prior large series of MR studies in patients with NSM. However, several studies on small numbers of patients with NSM have suggested that MR findings of NSM are similar to that of classic multiple myeloma6-8). In our case, the lesions, except for very small foci, were poorly enhanced on MRI after intravenous administration of contrast media; this finding was atypical relative to the common MRI findings for multiple myeloma. We assume that the reason for this atypical MRI finding may be related to the coagulation necrosis present in the myeloma.
Multiple thickened trabeculae were found in the involved vertebral lesions on CT scans of the present case. Major NM, et al reported that the appearance of a thickened cortical strut in myeloma is probably caused by a stress phenomenon from the lytic process of the myeloma making the remaining bone increase in thickness as a compensatory mechanism11). However, there are some differences between their cases and the present case. Their cases showed a thickened cortical strut with or without a thickened intralesional trabeculae, and demonstrated an expansile nature of the involved spinal lesion. By contrast, the present case revealed only thickening of the intralesional trabeculae without a thickened cortical strut or an expansile nature of the involved spinal lesion. This feature might make the diagnosis of myeloma more difficult. Although thickened trabeculae can also be seen on CT images of hemangioma, hemangioma usually show a high signal intensity on T1-wighted MR images due to the fat components contained in the hamangioma, and high signal intensity in T2-weighted MR images due to the slow flow velocity of the blood vessels contained in hemangioma. Therefore, exclusion of hemangioma in the differential diagnosis might be possible in the present case. We suggest that the diagnosis of NSM should be suspected in cases where CT images show thickening of the intralesional trabeculae of the involved vertebral bodies in a patient with no monoclonal gammopathy.
References
2. Bosman C, Fusilli S, Bisceglia M, Musto P, Corsi A. Oncocytic nonsecretory multiple myeloma: a clinicopathologic study of a case and review of the literature. Acta Haematol 1996;96:50ŌĆō56PMID : 8677762.


3. Cavo M, Galieni P, Gobbi M, Baldrati L, Leardini L, Baccarani M, Tura S. Nonsecretory multiple myeloma: presenting findings, clinical course and prognosis. Acta Haematol 1985;74:27ŌĆō30PMID : 3934904.


4. Angtuaco EJ, Fassas AB, Walker R, Sethi R, Barlogie B. Multiple myeloma: clinical review and diagnostic imaging. Radiology 2004;231:11ŌĆō23PMID : 14990813.


5. Mahnken AH, Wildberger JE, Gehbauer G, Schmitz-Rode T, Blaum M, Fabry U, Gunther RW. Multidetector CT of the spine in multiple myeloma: comparison with MR imaging and radiography. AJR Am J Roentgenol 2002;178:1429ŌĆō1436PMID : 12034612.


6. Rahmouni A, Divine M, Mathieu D, Golli M, Dao TH, Jazaerli N, Anglade MC, Reyes F, Vasile N. Detection of multiple myeloma involving the spine: efficacy of fat-suppression and contrast-enhanced MR imaging. AJR Am J Roentgenol 1993;160:1049ŌĆō1052PMID : 8470574.


7. Lecouvet FE, Vande Berg BC, Michaux L, Malghem J, Maldague BE, Jamart J, Ferrant A, Michaux JL. Stage III multiple myeloma: clinical and prognostic value of spinal bone marrow MR imaging. Radiology 1998;209:653ŌĆō660PMID : 9844655.


8. Stabler A, Baur A, Bartl R, Munker R, Lamerz R, Reiser MF. Contrast enhancement and quantitative signal analysis in MR imaging of multiple myeloma: assessment of focal and diffuse growth patterns in marrow correlated with biopsies and survival rates. AJR Am J Roentgenol 1996;167:1029ŌĆō1036PMID : 8819407.


9. Libshitz HI, Malthouse SR, Cunningham D, MacVicar AD, Husband JE. Multiple myeloma: appearance at MR imaging. Radiology 1992;182:833ŌĆō837PMID : 1535904.


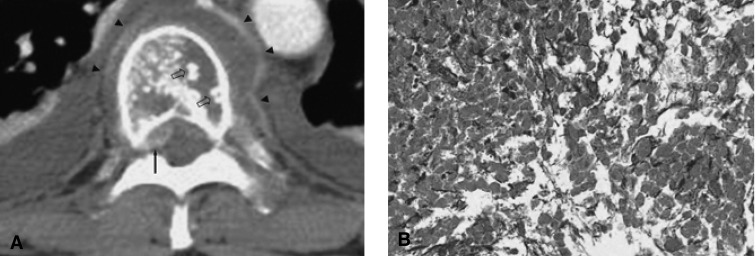
Figure┬Ā1
(A) Contrast-enhanced CT scan of the 8th thoracic vertebra shows multiple osteolytic lesions, thickened trabeculae (open arrows) and a perivertebral mass displacing the anterior thecal sac (arrow) and intercostal vessels (arrowheads). (B) Photomicrograph (original magnification, x200; hematoxylin-eosin stain) shows coagulation necrosis within the population of uniform monotonous cells.
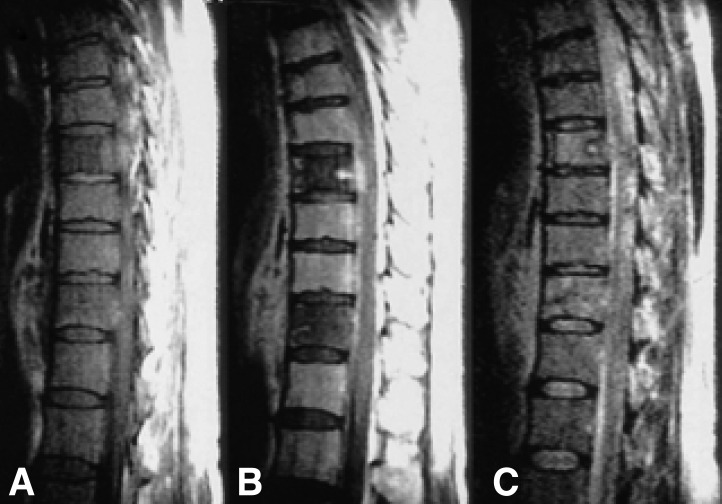
Figure┬Ā2
(A) T1-weighted sagittal image (500/15) shows ill-defined hypointense lesions in the 8th and 11th thoracic vertebrae. (B) Although there are very small foci of enhancement in the lesions at the 8th and 11th thoracic vertebra, most of the lesions are poorly enhanced on T1-weighted image after intravenous administration of contrast media. (C) T2-weighted sagittal image (1800/90) shows heterogeneously hyperintense lesions at the 8th and 11th thoracic vertebrae.
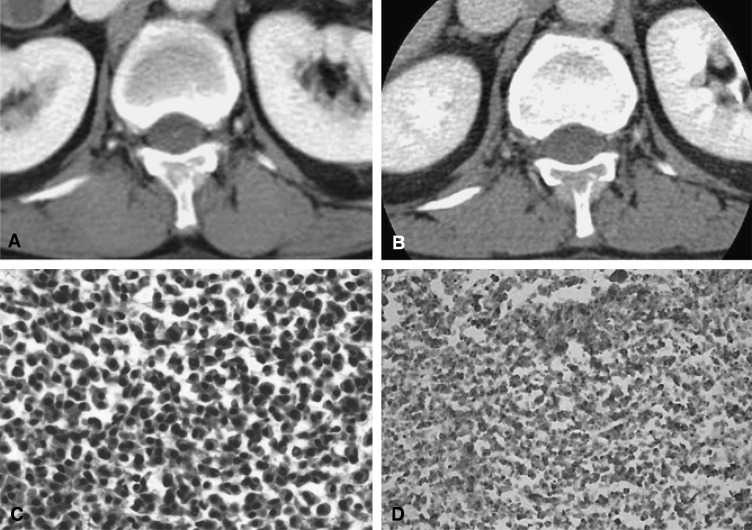
Figure┬Ā3
(A) Initial contrast-enhanced CT scan obtained at the level of the 12th thoracic vertebra shows an enhancing mass with cortical destruction of the posterior element of 12th thoracic vertebra. (B) The size of the mass was increased on contrast-enhanced CT obtained 30 days after the initial CT. (C) A photomicrograpth (original magnification, ├Ś400; hematoxylin-eosin stain) shows the proliferation of plasma cells. (D) A photomicrograph (original magnification, ├Ś200; immunohistochemical stain) shows strong positive reaction for lambda light chain.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



