INTRODUCTION
Hepatic subcapsular steatosis is a rare and specific form of fatty change in the liver. It is unique to diabetic patients on continuous ambulatory peritoneal dialysis (CAPD) who receive intraperitoneal insulin treatment1, 2). Intraperitoneal administration of insulin causes a unique pattern of fatty infiltration in the subcapsular location of the liver1-5). The incidence of this side effect is very low and only a small number of cases have been reported worldwide1-5). To date, there have been no cases reported in Korea. Here we report a case of hepatic subcapsular steatosis in a diabetic CAPD patient who received intraperitoneal insulin, and who improved after changing from CAPD to HD.
CASE REPORT
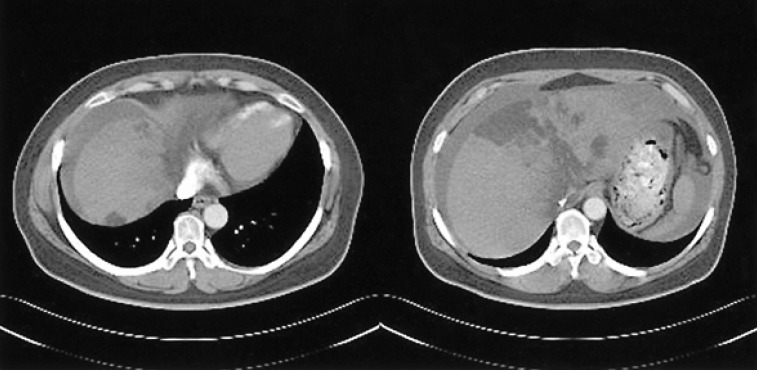
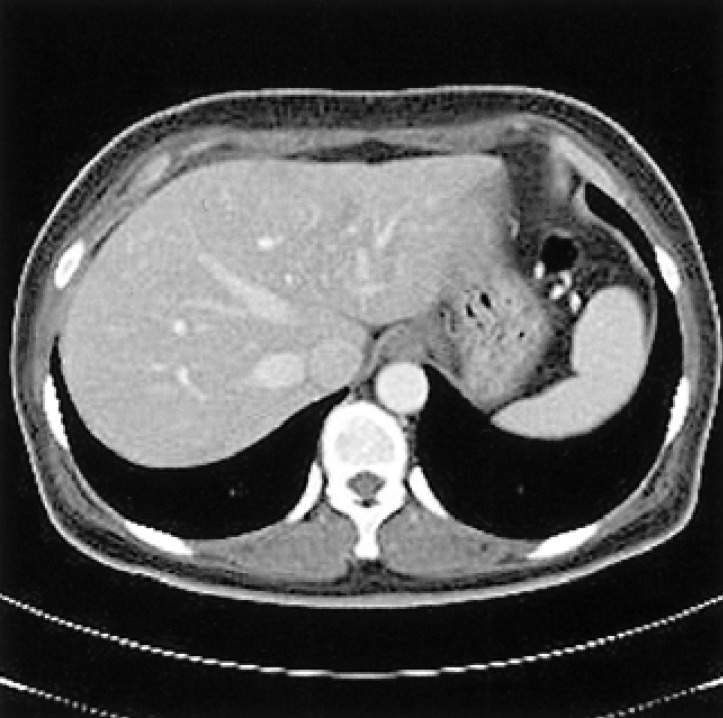
A 46-year-old woman presented with general weakness that had lasted for several weeks. She was diagnosed with type 2 diabetes mellitus (DM) 15 years ago and began continuous ambulatory peritoneal dialysis (CAPD) three years ago. An abdominal sonography obtained before CAPD began revealed a normal liver. The patient received CAPD with four daily exchanges of 2 L each time and switched to three exchanges with 2.5% glucose and one with 4.25% glucose solution; this was required because the ultrafiltration capacity was not sufficient and she became volume overloaded. A peritoneal equilibrium test was not performed. The patient had insulin administered subcutaneously when she started the CAPD but changed to intraperitoneal insulin treatment combined with subcutaneous treatment two years later. Hyperlipidemia treated with simvastatin and fenofibrate were started one year previously but stopped three months later because the drugs caused rhabdomyolysis. Lipid profiles began to worsen and she had to resume with atovastatin but did not develop rhabdomyolysis. Plasma glucose was poorly controlled despite the combination therapy of subcutaneous and intraperitoneal insulin injections. Insulin doses increased from 102 units daily up to 110 units daily during the previous month. The patient was 158 cm tall and weighed 58 kg. When she visited our clinic, her body mass index was 23 kg/m2. Vital signs were a blood pressure of 130/80 mmHg, pulse rate 68/min, respiratory rate 20/min, and body temperature 36.6℃. Her abdomen was soft and mildly distended. The initial complete blood count was white blood cells 8,000/mm3 (neutrophils 70.7%, lymphocytes 18.5%, eosinophils 3.2%), hemoglobin 11.5 g/dL, hematocrit 33.3%, and platelet 288,000/mm3. Her blood chemistry values were, fasting glucose 293 mg/dL, blood urea nitrogen 46.6 mg/dL, creatinine 10.3 mg/dL, sodium 134 mEq/L, potassium 4.4 mEq/L, chloride 90 mEq/L, AST 119 IU/L, ALT 192 IU/L, alkaline phosphatase 850 IU/L, amylase 110 U/L, total protein 6.8 g/dL, albumin 4.0 g/dL, calcium 11.1 mg/dL, phosphorus 6.2 mg/dL, CPK 43 IU/L, LDH 468 IU/L, total cholesterol 450 mg/dL, and triglyceride 1,096 mg/dL. Hepatitis B antigen and hepatitis C antibody were negative. ANCA, anti-liver kidney microsome antibody, and anti-smooth muscle antibody were all negative. Sonography of the abdomen revealed an increased echogenicity of the liver. A CT scan of the abdomen showed extensive low attenuated lesions in the liver capsule, the parenchyma of the left hepatic lobe and a small nodular lesion in the capsular area of the right lobe of the liver (Figure 1). The MRI findings were consistent with the CT findings. The hepatic subcapsular area showed bright signals in the on-phase T1weighted images, but a marked signal decrease in the off-phase T1-weighed sequences; these findings suggested that the subcapsular area was composed of fat tissue. Hepatic subcapsular steatosis due to high dose intraperitoneal insulin was suspected; the patient was changed from CAPD to HD. Insulin was given only subcutaneously after changing to HD. A CT scan performed two months after starting HD showed complete resolution of the hepatic subcapsular steatosis (Figure 2). Laboratory values were also improved: AST 34 IU/L, ALT 81 IU/L, alkaline phosphatase 733 IU/L, total cholesterol 259 mg/dL, and triglyceride 177 mg/dL. The blood sugar has been well controlled insulin therapy could be discontinued. Six months after changing from CAPD to HD, oral hypoglycemic agents have been sufficient to maintain adequate plasma glucose levels. Furthermore, liver enzyme levels are within normal range; the total cholesterol level was 206 mg/dL, and triglyceride level 157 mg/dL without requirement for lipid-lowering agents.
DISCUSSION
This is a case of hepatic subcapsular steatosis that occurred in a diabetic CAPD patient who had intraperitoneal insulin injections for glycemic control. Hepatic subcapsular steatosis is a unique form of nonalcoholic fatty liver that occurs only in diabetic CAPD patients who receive intraperitoneal insulin therapy1-5). It has been reported that if diabetic CAPD patients receive insulin by the subcutaneous route only, hepatic subcapsular steatosis does not occur1, 2).
Hepatic subcapsular steatosis is a rare disease worldwide1-5). To date, there have been no cases reported in Korea. Wanless et al.3) was the first to describe hepatic subcapsular steatosis in diabetic CAPD patients in 1989; This report examined the hepatic histology, at autopsy, in 11 individuals with renal failure who had received intraperitoneal insulin in conjunction with CAPD; they found hepatic subcapsular steatosis in 10 out of 11 patients. Kallio et al.2) was the first to demonstrate hepatic subcapsular steatosis in an in vivo study. Ultrasonography and/or MRI was performed in 16 end-stage renal disease patients with type 1 DM. Subcapsular steatosis occurred in seven out of eight patients treated with intraperitoneal insulin but did not occur in any of the patients receiving subcutaneous insulin.
The pathophysiology of hepatic subcapsular steatosis resulting from intraperitoneal insulin therapy is due to the high glucose and insulin levels in the peritoneal fluid1, 3). The subcapsular hepatocytes in CAPD patients, receiving intraperitoneal insulin therapy, are exposed to high concentrations of insulin and glucose from the hyperosmolar dialysate bathing the hepatic surface. This leads to hepatic steatosis limited to the subcapsular area in diabetic CAPD patients receiving intraperitoneal insulin treatment. Increased concentration of glucose and insulin delivered by the portal circulation potentially can lead to generalized steatosis and lipoprotein production6).
Hepatic subcapsular steatosis in diabetic CAPD patients is diagnosed by radiological imaging studies such as ultrasonography, CT scan and MRI of the abdomen2, 7, 8). Clinical symptoms are vague and can therefore be overlooked, but liver enzymes and lipid profiles are elevated. Hepatic subcapsular steatosis appears as bright echogenic rims on ultrasonography, and as low-attenuated lesions at subcapsular liver locations on CT scan images7). MRI is an excellent noninvasive method for identifying subcapsular fatty infiltration. Hepatic subcapsular steatosis appears as a hyperintense rim around the liver on MR T1 images, and shows very low signal intensity in fat suppression sequences. MRI can be a useful tool for the differential diagnosis of localized lesions or hepatic steatosis including: liver tumor, metastatic tumor and liver abscess8, 9). In our case, the CT scan and MRI findings were compatible with
the diagnosis of hepatic subcapsular steatosis.
In this case, the patient did not have detectable fatty changes of the liver on ultrasonography and her lipid profile was within normal range before she began CAPD. The blood sugar level was controlled with subcutaneous insulin therapy when she began CAPD; however, glycemic control was poor and intraperitoneal insulin therapy was started a few months later. Hepatic subcapsular steatosis resolved completely and the lipid profile and liver enzymes were normalized after the patient was changed to HD and subcutaneous insulin therapy. Therefore, the hepatic subcapsular steatosis, in this case, appears to have been associated with CAPD and intraperitoneal insulin therapy.
Hepatic subcapsular steatosis occurs frequently when the triglyceride levels and the intraperitoneal insulin doses are high6). A high transport type in the peritoneal equilibrium test has a higher propensity to develop hepatic subcapsular steatosis1). The triglyceride level in our case was markedly elevated, up to 1,096 mg/dL, despite adequate therapy with lipid lowering agents; a considerable dose of insulin up to 110 units was administrated. However, we did not perform peritoneal equilibrium test.
Eliminating the underlying cause is most important part of treatment for hepatic subcapsular steatosis7). Intraperitoneal insulin therapy should be substituted by subcutaneous therapy when maintaining CAPD if necessary, diet modification and exercise should be started, and hyperlipidemia corrected. CAPD should be stopped when response to treatment is poor, or if the degree of hepatic subcapsular steatosis is severe and the blood lipid and sugar control is poor7). The patient in our case was changed to HD and the insulin was given subcutaneously. CAPD was stopped for several reasons: first, the hepatic subcapsular steatosis was severe; second, the ultrafiltration capacity had decreased; third, the blood sugar level could not be controlled by subcutaneous insulin therapy alone; fourth, hyperlipidemia did not improve despite medical therapy. Hepatic subcapsular steatosis was completely resolved and liver enzymes and lipid profiles were normalized after changing from CAPD to HD.
In conclusion, though quite rare, hepatic subcapsular steatosis can occur in diabetic CAPD patients on intraperitoneal insulin treatment. Therefore, abdominal CT or MRI should be performed when liver enzymes or lipid profiles are elevated in such patients. Once hepatic subcapsular steatosis is diagnosed, intraperitoneal insulin therapy should be converted to subcutaneous therapy and hemodialysis should be considered in severe cases.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





