INTRODUCTION
Hemobilia, hemorrhage into the biliary tract, can result from a variety of iatrogenic and spontaneous causes. More than two-thirds of cases with hemobilia are iatrogenic. Non iatrogenic causes include blunt abdominal trauma, inflammation, gallstone diseases, tumors and vascular disorders1). Vascular disorders accounts for 10.7% of all cases1). Polyarteritis nodosa (PAN) is a multisystem necrotizing inflammatory vasculitis of small and medium sized muscular arteries, and is associated with immune complex deposition in the vessel walls2). Hemobilia, secondary to the rupture of a hepatic aneurysm in PAN, is a rare entity. A thorough search on medline revealed only four such cases reported in the current medical literature3-6). We report a case of an angiographically documented arteriobiliary fistula in massive hemobilia due to rupture of a hepatic artery aneurysm as a manifestation of polyarteritis nodosa. Hemobilia was successfully controlled by coil and gelfoam embolization.
CASE REPORT
A 39-year-old man was admitted with severe right upper quadrant abdominal pain. The history was significant for partial small bowel resection for intestinal infarction about 5 years prior to this presentation. The patient was otherwise stable without any further investigations or management, and had been asymptomatic until the current episode.
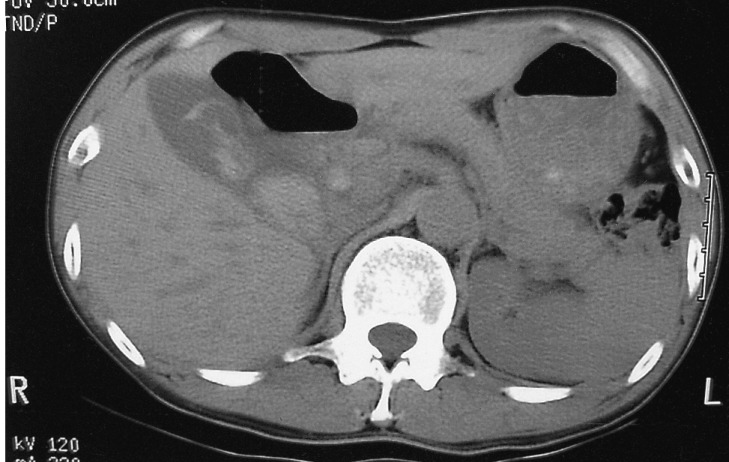
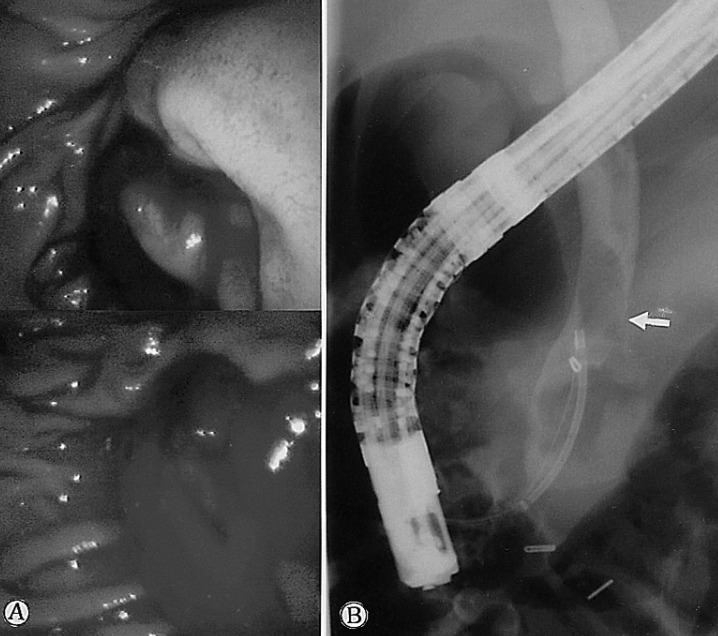
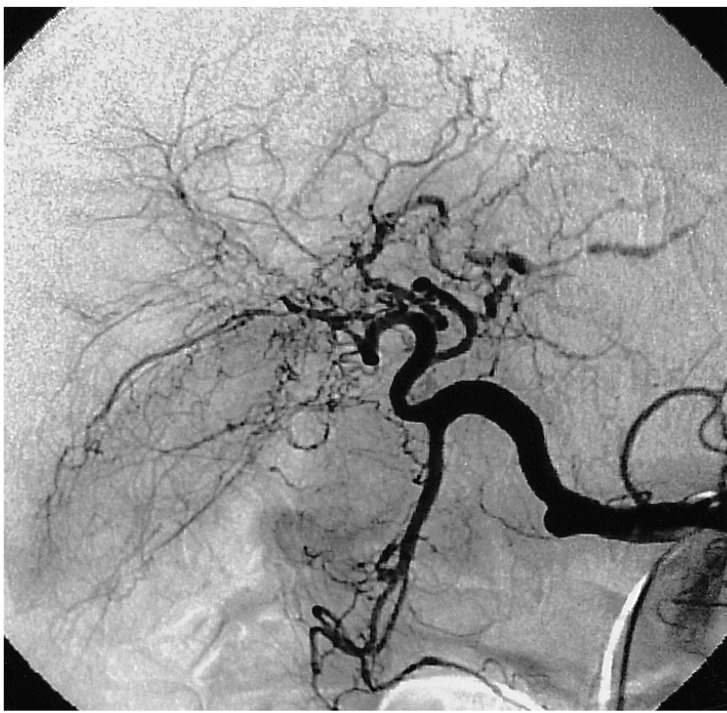
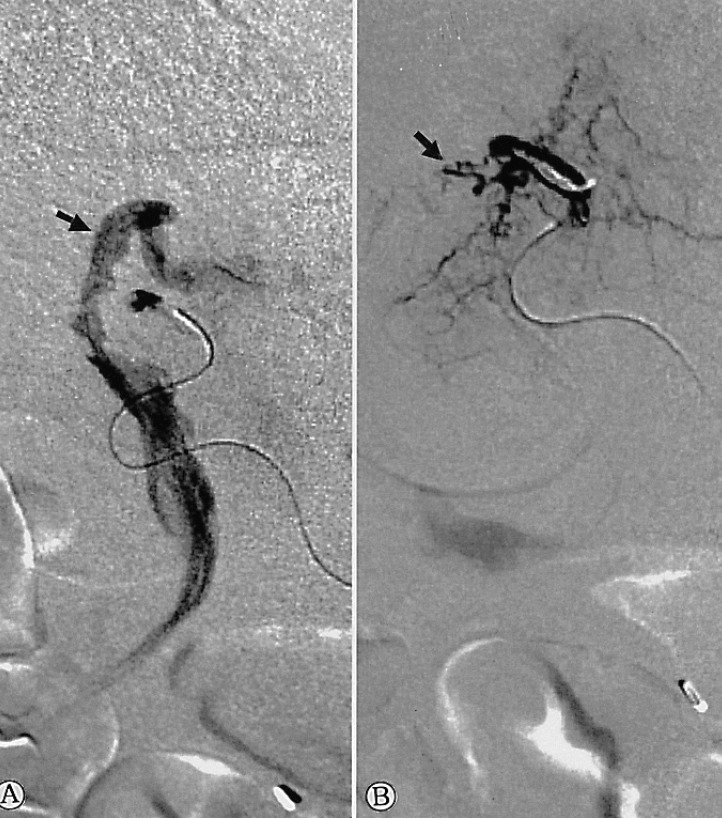
On admission, his vital signs were as follows: blood pressure 160/100 mmHg, pulse rate 72/min, respiratory rate 18/min, temperature 36.3Ōäā. He had minimal conjunctival injection and icteric sclerae. Abdominal examination revealed normal bowel sounds with no palpable abdominal mass or organomegaly. There was tenderness to palpation over the right upper quadrant and epigastrium. He did not have any rebound tenderness and Murphy's sign was negative. Laboratory studies showed WBC 5700/mm3, Hb 12.3 g/dL, hematocrit 35.0%, and platelets 183,000/mm3. Liver function tests revealed a total bilirubin of 5.2 mg/dL, AST 175 IU/L, ALT 185 IU/L, alkaline phosphatase 899 IU/L, and GTP 615 IU/L. Abdominal CT demonstrated multiple high attenuation areas in the biliary duct and gallbladder (Figure 1). Endoscopic retrograde cholangiopancreatography (ERCP) showed a mild but active blood leak and adherent clots at the ampulla of Vater (Figure 2A). Cholangiography revealed multiple tubular filling defects in the common bile duct as well as the intrahepatic ducts (Figure 2B). Endoscopic sphincterotomy and nasobiliary drainage were performed. During the procedure, hemobilia spontaneously stopped. He reported an improvement in his abdominal pain after the procedure. However, the following day, the patient developed a massive hematemesis and hematochezia, with a drop in his blood pressure to 70/50 mmHg and Hb to 7.5 g/dL. Emergency selective hepatic angiogram demonstrated multiple microaneurysms in the branches of the hepatic arteries (Figure 3) and a slow leak of the contrast medium into the bile duct from the arteriobiliary fistula (Figure 4A). This fistulous tract was blocked with coil and gelfoam (Figure 4B). Following this procedure, his blood pressure stabilized rapidly. There was no evidence of rebleeding. Superior mesenteric, inferior mesenteric and renal artery angiography revealed multiple stenoses, smooth occlusions and microaneurysmal changes in small- to mid-sized vessels. Antinuclear antibody, antineutrophil cytoplasmic antibodies, rheumatoid factor, antiphospholipid antibody and lupus anticoagulant were all negative. A clinical diagnosis of polyarteritis nodosa was made and he was managed with prednisolone 25 mg per day, which was tapered to 5 mg per day, in combination with cyclophosphamide 50 mg per day. He is entirely asymptomatic at six month follow up.
DISCUSSION
Hemobilia, is a condition where there is an abnormal connection between the blood vessels and bile ducts; this is usually caused by trauma or iatrogenic injury to the liver and biliary tree. Hemobilia originates in the liver in one half of reported cases7). Non traumatic vascular causes include congenital and developmental disorders, atherosclerosis and inflammation. Vascular lesions, usually hepatic artery aneurysms secondary to atherosclerosis, trauma, gallstone diseases, or systemic infections, account for about 10% of identified lesions8, 9). Aneurysms are usually isolated, although multiple aneurysms may be seen in patients with vasculitis9). Abrupt episodes of bleeding with disproportionately severe hemodynamic collapse strongly suggest a vascular cause for hemobilia10).
The classic triad of gastrointestinal bleeding, biliary pain, and jaundice should alert the clinician to the possibility of hemobilia9). The typical clinical symptoms associated with hemobilia are seen in only 40% of patients9). The diagnosis of hemobilia can be made on ERCP, as in the present case, where active bleeding at the papilla and filling defects consistent with blood clots in the biliary system were identified.
Confirmation of this diagnosis usually requires angiography with visualization of the hepatic artery and its branches. Cholangiography helps in the definitive pre-operative identification of the bleeding site and also serves as the treatment of choice for patients with hemobilia11). Although there have been some cases of spontaneous hemostasis reported, hemobilia usually requires intervention. Traditional therapies have involved hepatic resection or hepatic artery ligation. Recently, angiographic therapy has supplanted surgical repair as the treatment of choice for hemobilia. Transcatheter embolization with gelfoam, steel coil, or a combination of both has been shown to be effective11). Surgical intervention becomes necessary when non-operative attempts to stop the bleeding fail, and is required for tumors and parasitic diseases11). In the present case, hemobilia stopped spontaneously at the time of admission, but massive gastrointestinal bleeding with hypotension followed. Selective embolization of the right hepatic artery was successful in controlling the hemorrhage.
A sphincterotomy at the time of ERCP helped to relieve the obstructive jaundice and abdominal pain. The passing of large quantities of blood clots probably relieved the abdominal pain at the time of admission. However, the release of a tamponade effect from the blood clots in the biliary system probably resulted in massive hemorrhage several hours later. This emphasizes the importance of hemostasis at the primary bleeding site. After obtaining hemostasis through arterial embolization, ERCP with sphincterotomy may be valuable in selected cases to manage the complications of progressive obstructive jaundice and abdominal pain resulting from retained intrabiliary clots12).
PAN is associated with thrombosis and aneurysm formation in any organ of the body. The gravest complications involve the gastrointestinal tract and occur in more than half of patients with the diagnosis of PAN13). The hepatic arteries are a frequently involved; although the rupture of a hepatic aneurysm is a rare event14).
Biopsy of skin, muscle or testicle, or a percutaneous biopsy of the liver or kidney establishes the diagnosis of PAN. Abdominal angiography is the diagnostic procedure of choice. Of all known patients with PAN, 70~80% have positive abdominal arteriograms consistent with arteritis. Of those with positive arteriograms, 70% of cases have multiple microaneurysms. Arteriographic findings consistent with PAN usually establish the diagnosis14). In the present case, angiographic evidence pointed towards intestinal infarction and aneurysmal rupture; two of the most dreaded complications of mesenteric vasculitis14). Mesenteric angiography demonstrated multiple stenoses, collateral vessels, and multiple microaneurysms of hepatic, superior and inferior mesenteric, as well as renal arteries. The diagnosis of PAN was made clinically based on the above findings. Active bleeding from the aneurysms can be controlled by selective embolization of the bleeding sites, as was done in our case.
In conclusion, this case report describes an unusual manifestation of PAN, hemobilia that resulted from hepatic artery aneurysmal rupture. The arteriobiliary fistula was demonstrated on angiography, and was successfully treated with arterial embolization.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





