INTRODUCTION
BehcetŌĆÖs disease (BD) is known to be a systemic disease of unknown process affecting multi-organs with a variety of clinical manifestations, which are a result of systemic vasculitis caused by the deposition of immune complex onto the eye, skin, joint and central nervous system1). But the pulmonary involvements, which include pulmonary artery aneurysm (PAA), pulmonary infiltrates and pulmonary infarction, in BD are uncommon2). Furthermore, in the female, involvement of pulmonary artery is quite rare3). There were a few cases of female BD patients with PAA reported, but there was no report in Korea.
In this article, we report a rare case of BD, combined with PAA, confirmed by pulmonary angiogram in a Korean female patient.
CASE REPORT
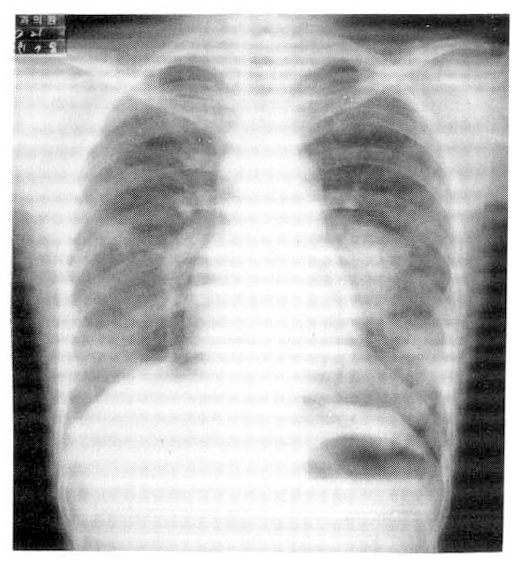
A 21-year-old Korean woman had suffered from dyspnea and cough for the past 3 months. She was healthy until 2 years ago, when she first noticed both knee joints to swell and subside spontaneously. She also experienced intermittent both lower leg pain. Two months ago, chest P-A showed heterogenous, hazy, streaky densities on both lower lung fields and a well-marginated homogenous squared shape of mass-like density on the right middle lung (Fig. 1). She was treated with isoniazid, rifampin and pyrazinamide under the diagnosis of pulmonary tuberculosis, which was not confirmed by microbiologic study, at a private clinic. After 2 months of anti-tuberculous drugs, the chest X-ray revealed much improved infiltrations on both lower lung fields and right middle lung, but with newly developed lobulated, round homogenous density, with 4├Ś4 cm in diameter, at the left hilar region (Fig. 2). She was transferred to our hospital for the evaluation of the left hilar lung mass.
The patient was single, non-smoker and non-drinker. There was no history of pulmonary tuberculosis. She denied fever, sweats, hemoptysis, headache, otalgia and otorrhea, and bladder or bowel dysfunction.
The temperature was 36.8 ┬░C, the pulse was 68 and the respiration was 18. The blood pressure was 130/80 mmHg.
On physical examination, she appeared pale and chronically ill. She had multiple ulcerations on lower lip and oral cavity near right premolar teeth, but not on genital area. The ophthalmologic study showed no abnormal findings. The lung, heart and abdomen were normal. The skin examination revealed erythematous, tender nodules on wrist and both pretibial areas, which were pathologically proven as erythema nodosum. A gynecologist notified the aphthous ulceration of labia minora. The remaining physical examination was normal.
The urine was normal. The hematocrit was 27 percent; the white-cell count was 9,000/mm3, with 68 percent neutrophils, 29 percent lymphocytes, 2 percent monocytes and 1 percent eosinophils. The ESR was 28 mm/hour. The shape of RBC was microcytic and hypochromic. The urea nitrogen was 18 mg%, the glucose 80 mg%, the serum alanine aminotransferase 20 IU, the alkaline phosphatase 56 U. The VDRL was negative. The arterial blood gas analysis showed PH; 7.40, PaCO2; 41 mmHg, PaO2; 96 mmHg. An electrocardiogram was normal.
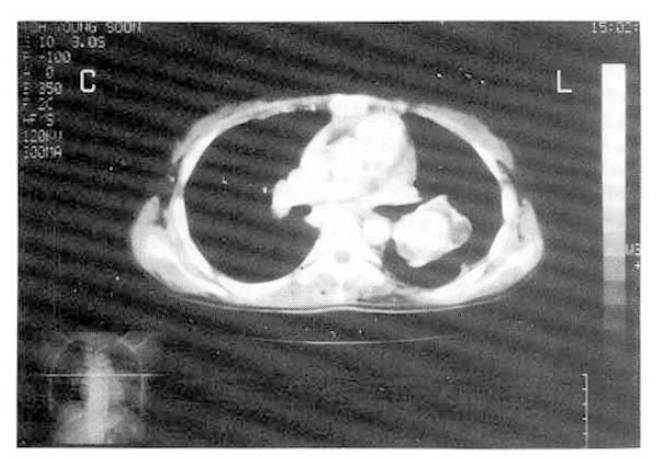
The perfusion scan showed lobar perfusion defect of left lower lobe and focal segmental perfusion defect of right lower lobe. The ventilation scan showed round single defect in left hilar area. The computerized tomogram of thorax revealed 5├Ś5cm-sized, round, mass with surrounding low density, suggesting PAA with thrombosis in left lung (Fig. 3). Ascending venogram of lower leg showed diffuse obstruction of right femoral vein. The digital subtraction pulmonary angiogram revealed huge dilatation of left main pulmonary artery, proximal to the obstructed pulmonary artery (Fig. 4).
Ten days after admission, blood-tinged sputum occurred abruptly at early morning. The patient was treated with steroids for 14 days without improvement of hemoptysis. During the next 6 weeks, although she was given steroids persistently, the left hilar lesions grew to large mass with diameter of 5 cm. We decided to excise the aneurysmal mass. Macroscopic examination of the excised lobe showed saccular dilation of pulmonary artery surrounded by the underventilated pulmonary parenchyma. On section, the aneurysmal dilatation was 5 cm in diameter and lined with laminated old blood clots. The main histopathological findings were located in the arterial walls. The arterial wall was markedly destructed with minimal fibrous thickening, medial destruction, marked infiltration of inflammatory cells, predominantly neutrophils. A large area of aneurysmal wall was necrotic, ulcerated and attached by thrombus. On special stain, elastic lamina was marked destructed, fragmented and frequently lost. There were multifocal infarctions, both in parenchymal and subpleural portion.
She was treated with maintenance prednisolone during 1 year after lobectomy, but she noted sudden massive hemoptysis and eventually died.
DISCUSSION
The underlying causes of PAA without arteriovenous communication are infection (mycotic), structural cardiac abnormalities, structural vascular abnormalities including MarfanŌĆÖs syndrome and BD, pulmonary hypertension and trauma4). In this case, BD is the cause of PAA because of the presence of recurrent oral and genital ulceration and skin lesion (erythema nodosum) which fulfill the diagnostic criteria of BD5).
Of 137 and 316 patients with BD, 38 (21%) and 69 (28%) patients had vascular involvement, respectively6,7). Both studies showed marked male and vein predominance. Therefore, there were a few cases of female patients with BD combined with PAA confirmed by pulmonary angiogram3,8,9). So, it is valuable to report this rare case of female BD with PAA confirmed by angiogram.
The pulmonary manifestations, including recurrent episodes of dyspnea, cough, chest pain and hemoptysis, occur in 5% to 10% of patients with BD10). The major symptom is hemoptysis, which is the result of pulmonary embolism evoked by deep thrombophlebitis or pulmonary thrombophlebitis11) or communication between PAA and its related bronchus3). In this case, the first manifestation of blood-tinged sputum may be related with pulmonary embolism secondary to deep vein thrombosis which were proven by lung scan and RI venogram. The massive hemoptysis resulting in death may be rupture of the main aneurysm to related bronchus.
Although the CT findings of thorax have not been well documented, aneurysm with or without thrombus, irregular configuration of peripheral vessels, peripheral opacities in the lower zones and enlarged peripheral arteries were reported3,8,11,12). In this case, CT scan with contrast media showed well-opacified round arterial lumen with eccentric soft tissue mass which suggested thrombus within aneurysm. We think that the thrombus within aneury sm may be related with stasis of blood flow due to obstructed distal arterial lumen by vasculitis.
Pulmonary angiogram is gold standard in the diagnosis of PAA4). The angiogram might deteriorate pulmonary disease resulting in death within one year10,11). This deterioration was the result of a puncture of the inflamed arterial wall12), severe ill state and pulmonary catheterization itself11). So, it is doubtful to perform pulmonary angiogram for demonstration of aneurysm and vascular change. Fortunately, we did not find any deterioration of pulmonary symptoms after angiogram in this case. In future, high resolution computerized tomogram and dynamic CT may replace angiogram in confirmative or routine diagnostic modality of aneurysm without any complication.
The steroid, with or without immunosuppressive agents, is the first line therapy in BD with lung involvements. But the long-term effect of steroid is not defined, in spite of the short-term effect of steroid being reported to be very good12). Furthermore, patients with PAA died due to massive hemoptysis by rupture of aneurysm during treatment with massive steroids3,13). If the aneurysm dilates, the dilating force increases and in turn, results in the rupture of aneurysm4). Thus, we used steroid in management of PAA without improvement. Eventually, we performed lobectomy due to the fear of rupture of the aneurysm.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





