 |
 |
| Korean J Intern Med > Volume 2(2); 1987 > Article |
|
Abstract
The effects of interferons (IFN) on in vitro differentiation of B-lymphocytes were studied. Peripheral lymphocytes from normal subjects were cultivated under polyclonal activator pokeweed mitogen (PWN) or Epstein-Barr virus (EBV) stimulation. The secreted Ig in the culture supernatants were measured for IgM by ELISA method.
To determine the cellular level of IFN action T-cell enriched fraction (Te) or B-cell enriched fraction (Be) were preincubated with IFN prior to recombination culture.
IFN had modulatory activities on Ig production; at low to moderately high doses (10–1000 U/ml of IFN-alpha or 12–120 U/ml of IFN-gamma) stimulating when IFN was added until 48 hr after the start of the culture, while after 72 hr from culture start IFN suppressed Ig production.
Preincubation of Be-cells with moderately high doses of IFN (120 U/ml of IFN-gamma or 1000 U/ml of IFN-alpha) prior to PWM-stimulation suppressed Ig production. Likewise, in EBV-stimulated culture, high dose IFN suppressed Ig production. But low dose of IFN enhanced ig production in EBV-stimulated culture.
Preincubation of Te-cells with IFN prior to PWM-stimulation with Be-cells enhanced the Ig production. The T-cell subset analysis at the end of these culture showed enhanced ratio of T-helper cell relative to T-suppressor cells, suggesting increased T-helper cell proliferation after incubation with IFN.
Thus, it is concluded that IFNs have modulatory activities on B-cell differentiation. The mechanism seems to be direct effects on B-cells (in PWM and EBV system) as well as through T-helper cell mediation (PWM system). The IFN-gamma showed more potent (2-to 6-fold) stimulatory activities than IFN-alpha.
The first identifiable marker in the development of an antibody-producing plasma cell from the bone marrow precursor cell is the rearrangement of the immunoglobulin heavy-chain genes which is followed by the rearrangement of light chain genes1).
At this point the stem cell becomes the pre-B cell which contains heavy chains in its cytoplasm. The pre-B cell gives rise to B-cells bearing their surface immunoglobulin and other markers1,2). Antigens binding to immunoglobulin on B-cell surface initiate the B-cell proliferation and terminal differentiation into antibody-producing plasma cells.
This process of B-cell maturation is also brought about by Epstein-Barr virus3,4) and other plant and bacterial products known as poly-clononal B-cell activators.
Many of them require T-helper cells and macrophages for B-cells to induce maximum immunoglobulin response.
In the studies of human immunoglobulin synthesis, polyclonal activiators pokeweed mitogen or Epstein-Barr virus have been extensively utilized5).
These studies could define stages of B-lymphocyte differentiation and the roles played by networks of immunoregulatory T-cells and macrophages in the B-cell maturation.
These studies clarified the pathogenic mechanisms of primary immunodeficiency disease6) autoimmune disorders7,8) and acquired immunodeficiency disorders9,10).
Recently the effects of interferons have been extensively studied and interferon is being introduced as a theurapeutic agent for various disorders in the clinic11,12).
In addition to their antiviral effects13,11) interferons are reported to have a variety of immunologic functions, such as inhibition of cellular proliferation13–16) activiation of the natural killer cells17–19) or the enhancement of the cytotoxicity of the immune T-cells20).
Interferon is also known to have modulatory activities on B-lymphocyte activiation21–23). Inteferon suppressed the antibody response to antigen when interferon was given to cells 6 hour prior to or up to 48 hours after antigen challange21). However, pretreatment of B-cells with low amounts of Interferon or late addition of interferon to antigen stimulated culture led to enhanced antibody synthesis.
Gisler et al21) found inteferon did not affect T-helper function or macrophage function in their system and concluded Interferon acted directly on B-cells. Choi, et al24) studied the effects of Interferons on human B-cell differentiation with the polyclonal activator pokeweed mitogen.
They found that the effect of interferon was dose-dependent; enhancing B-cell differentiation at low dose, while suppressing it at high dose.
Their experiments suggested the increased production of immunoglobulin was due to macrophage activation by interferon and the suppression of immunogobulin production was the result of impaired T-helper cell proliferation.
At a high dose of interferon, B-cell proliferation was directly suppressed.
Harfast et al25) also found that depending on the time of exposure, interferon either enhanced or suppressed the pokeweed mitogen-induced immunoglobulin synthesis by human peripheral blood lymphocytes. They and Pelton and Denman26) found that interferon acted exclusively on B-cells.
The aims of the present study were as follows.
First, to further investigate the effects of interferons on B-cell differentiation induced by polyclonal activators.
Using T-dependent (pokeweed mitogen) and T-independent mitogen (Epstein-Barr virus) and other techniques, the mechanism of interferon action on the B-cell differentiation in vitro was studied.
Second, to verify the effect of the conventional virus-induced interferon preparations as the real actions of inteferon.
By comparing the effect of conventional inteferon preparations with the effect of recombinant forms of interferon this study could exclude the effects of possible impurities in the former preparations.
Additionaly, the relative potency of different types of inteferons (IFN-α and IFN-γ) was studied.
Peripheral blood was obtained from normal adult volunteers.
Mononuclear cells were Isolated by Ficoll-Hypaque density gradient centrifugation. T-and B-enriched cell fractions were obtained by rosetting with neuraminidase-treated sheep erythrocytes27).
Mononuclear cells and sheep erythrocytes were mixed in a ratio of 1:50, centrifuged on a Ficoll-Hypaque gradient in siliconized tubes. Cells at the interface were subjected to a second cycle of sheep erythrocytes rosetting to further separate the rosette forming cells. The interface cells obtained after 2 cycles of rosetting were obtained as B-enriched cells which contained less than 5% T-cells. The rosette forming cells obtained after hypotonic shock in 0.9% ammonium chloride solution were used as T-cells which contained less than 5% non-T. The T-and Be-cells were were washed twice and were suspended in RPMI-1640 medium plus 10% fetal calf serum containing glutamine(1:1000 v/v) and antibiotics (1: 1000 v/v) (Grand Island Biological Co. NY, USA).
Virus-induced human leukocyte IFN-α (Polyferon, Bioferon, West Germany) and recombinant IFN-α (rIFN-α) and recombinant IFN-γ (rIFN-γ) (Dr. karl Thomae Ltd., west Germany) were used.
For mononuclear cell culture, 1 ×106/ml cells in 12×75 mm plastic tubes (Falcon, MD, USA) were stimulated with 10 μg/ml of pokeweed mitogen (Grand Island Biological Co., NY, USA).
For coculture of T-and Be-cells, 2×106/ml T-cells and the same number of Be-cells were mixed together into round bottomed 96-well plates (Nunc, denmark) in a total volume of 0.2 ml. Culture were incubated for 12 days at 37°C in humidified air containing 5% CO2.
In experiments using Epstein-Barr virus as the polyclonal activiator of B-cells, the mononuclear cells at a concentration of 3×106/ml were incubated with culture supernatants of B-95-8 cell line for 2 hrs, washed twice and plated in round-bottom culture plates at a concentration of 2×106/ml in volumes of 0.2 ml. Desired amounts of rIFN-γ were added to cultures.
An enzyme-linked immunosorbent assay (ELISA) modified from Kelly et al.28) was used. Microtiter plates (Dynatech, Va, USA) were coated with 0.2 ml of affinity column-purified rabbit anti-human IgM (Atlantic Antibodies, Me, USA). The anti-Ig had been diluted in 0.1 M sodium bicarbonate buffer, PH 9.6, to a concentration of 1 μg/ml.
The plates were incubated at 4°C overnight and stored at −20°C and used in less than 4 weeks. The coated plates were extensively washed with phosphate buffered saline (PBS) containing Tween 20 (0.05% v/v) (Tween-PBS).
Each well received 0.2 ml of Tween-PBS containing 5% of culture supernatant to be tested or serial dilutions of known amounts of IgM.
After 4 hrs incubation at room temperature or overnight incubation at 4°C, the plates were extensively washed with Tween-PBS and 0.2 ml of alkaline-phosphatase-conjugated polyvalent anti-lg in Tween-PBS (1 μg/ml) (TAGO Inc., CA, USA) were added.
After 2 hrs incubation at room temperature, the plates were again washed extensively with Tween-PBS and 0.2 ml of P-nitrophenyl phosphate disodium (1 mg/ml, Sigma, St. Loius, USA) in 0.1 M sodium bicarbonate buffer, pH 8.6, containing 0.001 M MgCl2 were added to each well.
After 1hr incubation at room temperature, the absorbance at 492 nm was measured with a Dynatech automatic reader.
In each experiment a standard curve was constructed from samples containing known amounts of IgM (Nor-Partigen, Behringwerke, West Germany).
All cultures were run in triple and each sample was measured duplicate by ELISA.
The effect of pretreating T-cells or B-cells with IFN on PWM-induced Ig production was studied by incubating 2×108/ml cells with different IFN concentrations for 16 hrs at 4°C.
The cells were then washed twice, resuspended in culture medium at desired cell concentration.
The IFN-treated T-cells were recombined with untreated Be-cells and the IFN-treated Be-cells were recombined with untreated T-cells.
The effect of preincubation of T-cells with IFN on T-cell subset composition was studied by testing the T-cell marker profile of the lymphocytes at the termination of the culture.
T-cell subsets were determined with the immunoenzyme technique.
Leu-4, Leu-3 and Leu-2 (Beckton-Dickinson, CA, USA) monoclonal antibodies were utilized.
Virus-induced human leukocyte IFN-α added to the lymphocyte culture in a dose of 300 U/ml enhanced Ig production, most prominently when it was given 24 hrs prior to PWM stimulation (Table 1).
The effects of IFN-α at doses 10,100,1000, and 10,000 U/ml at the time of culture start, 48 hrs, 72 hrs and 96 hrs after the culture start are shown in Fig. 1. At all the doses employed, IFN-α stimulated Ig production when it was added until 48 hrs after culture start (+ 18%, +76% of control). The addition of IFN-α after 72 hrs from culture start had a suppressive effect, while at the dose of 10,000 U/ml it retained stimulatory activity.
In a preliminary study, the addition of 100 U/ml of rIFN-γ to lymphocyte culture enhanced the Ig production three-to five-fold (Table 2).
This effect was noted when rIFN-γ was added to culture 24 hrs prior to, at the time of culture or 72 hrs after the culture start.
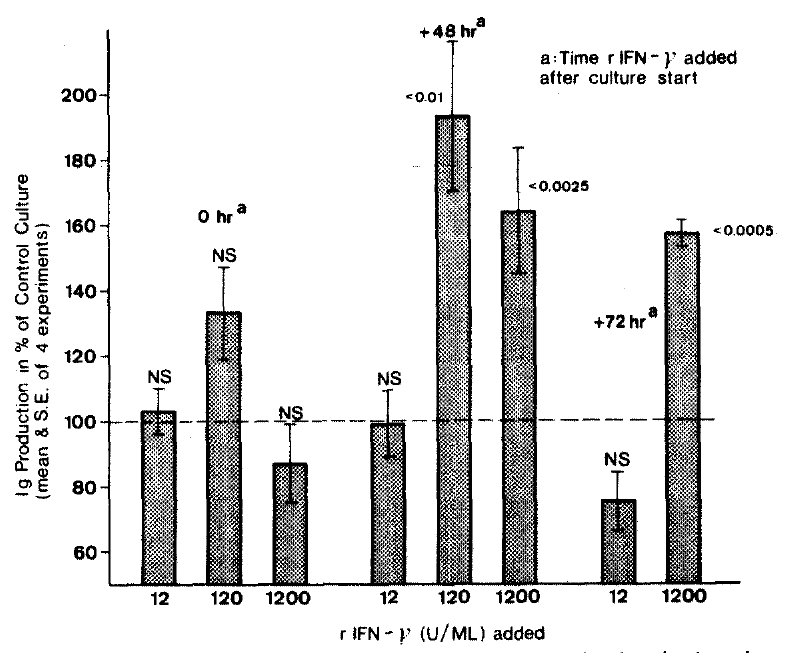
In further experiment employing doses of r-IFN-γ 12,120 and 1200 U/ml (Fig. 2), the dose of 120 U/ml rIFN-γ given 48 hrs after culture start was most stimulating. It enhanced the Ig production by 94%.
When T-cells preincubated with IFN were recombined with untreated autologous Be-cells, Ig production was enhanced (+ 85% and +487% of control) (Table 3). Conversely, when B-enriched cells were preincubated with IFNs and then recombined with untreated T-cells, there was marked suppression of subsequent Ig production (<20 ng/ml) (p<0.05).
To study the effects of IFNs on the proliferation of T-cell subsets, the T-cell subset profile was determined in the cultures at the termination of culture, on day 12 (Table 4). The cultures which contained T-cells preincubated with rlFN-α or with rIFN-γ had increased helper to suppressor T-cell ratio in comparison to control culture (p<0,025).
At a dose of 10 U/ml, rIFN-γ stimulated Ig production induced by EBV (+ 112%) (Table 5). At higher doses (100 U/ml and 500 U/ml), rIFN-γ had significant suppressive activity on EBV-induced B-cell differentiation.
In studying the regulation of Ig production in the human system, the polyclonal activator PWM has been widely used.
The merit of this system is that analysis of the respective functions of T-helper, T-suppressor, macrophage or B-cells is possible.
After the polyclonal activation, the secreted Igs can be assessed by hemolytic plaque assay or quantitiated by double antibody radioimmunoassay or Enzyme-liked immnuo-sorbent assay28).
The present study employed polyclonal activators PWM and EBV and quantitated the secreted Ig by ELISA method to study the effects of inteferons on human B-cell maturation.
By employing T-dependent system using PWM as well as the pure B-cell mitogen EBV, the cell types on which IFN acted could be dissected.
The ELISA techique is an objective and sensitive method for measuring Ig levels.
The results of the present study show that IFNs have modulatory activity on polyclonal activator (PWM or EBV)-induced Ig production by human peripheral blood lymphocytes. In accordance with previous results24,25,29), IFN enhanced or suppressed the Ig production of lymphocytes depending on time and dose of addition.
When low to moderately high dose of IFN-α (10 to 1000 U/ml) were added to culture until 48 hrs after PWM stimulation, it had stimulatory activity.
With a very high dose of IFN-α (10,000 U/ml) or with r-IFN-γ the Ig production was enhanced until 72 hrs after the PWM-stimulation of the culture.
The effects of virus-induced interferon and the recombinant types of interferons were similar in their actions, suggesting that the action of the former preparations were not that of possible contaminating lymphokines.
The effects of recombinant inteferon-γ were most striking and enhanced the Ig procuction 2-to 6-fold of control cultures. In EBV-induced Ig production by peripheral blood lymphocytes, r-IFN-γ displayed regulatory activities: at low dose (10 U/ml) enhancing and at higher doses (100 U/ml and 500 U/ml) suppressing Ig production. Since EBV is known to stimulate B-cells independent of accessory cells3,4) this regulatory effect of IFN on B-cell differentiation may be its direct effect on B-cells, as was reported by other group30). This modulatory effect of IFN on EBV-driven Ig production by human lymphocyte culture is first reported in this study. A novel interpretation of the regulatory effects of IFN on antibody production is that of Gresser13).
According to him, low blood level of IFN at initiation of viral infection could amplify the antibody response while high blood level of IFN at termination of viral infection could suppress the further B-cell activiation.
In co-culture studies, T-cells preincubated with IFN were recombined with untreated B-cells under PWM stimulation. This culture produced more Ig than control culture and contained more T-helper cells than in control culture, suggesting IFN preincubation of T-cells favors T-helper cell proliferation, which then stimulates B-cells to produce more Ig.
Thus the mechanism of action of IFN on in vitro Ig production by human lymphocytes seems two-fold; direct regulatory action on B-cell activation as reported by Gisler et al21) Harfast et al25) and Pelton and Denman26) as well as through T-helper cell mediation as was reported by Sonnenfeld et al29).
To sum up above results; 1) A low dose of IFN stimulates B-cell differentiation in vitro.
2) A moderately high dose of IFN stimulates T-helper cell proliferation which then sitimulates B-cell differentiation.
3) B-lymphocytes exposed to moderately high to high dose of IFN were directly suppressed in differentiation.
Recently, using anti-immunoglobulin (anti-μ) or Staphylococcus aureus Cowan strain (SAC) for B-cell activation followed by addition of factors for maturation of B-lymphocytes, more direct analysis of B-cell differentiation events became possible31,32). SAC activted B-cells were induced to proliferate by interleukin-2 but secreted no Ig. The subsequent addition of interferon-γ brought Ig secretion30). Sidman et al23) also found that IFN is one of B-cell differentiation factors. As clinical application of IFN is being carried out, it may be interesting to study the effects of in vivo application of IFN on the regulation of Ig production.
Another subject for further study would be the effects of various lymphokines on normal and malignant B-cell proliferation and differentiation after anti-μ or SAC induced activation.
In accordance with the previous results24,25) the present study showed that IFN has a modulatory activity on polyclonal activator-induced Ig production by human peripheral blood lymphocytes: depending on the time and dose of addition, IFN either enhanced or suppressed the Ig production by MNCs.
This modulatory effect of IFN has also been demonstrated in the EBV-driven Ig production by human MNCs, which is first reported in this study.
In coculture studies, T- or B-cells were preincubated with IFNs and recombined with untreated autologous B- or T-cells for culture under PWM stimulation, preincubation of B-cells with moderately high dose of IFN prior to PWM-stmulation with T-cells suppressed the subsequent Ig production.
On the other hand, T-cells preincubated with IFN prior to PWM-stimulation with B-cells produced increased amount of Ig. The T-cell subset analysis at the termination of these culture showed enhanced proliferation of T-helper cells relative to T-suppressor cells.
Thus the mechanism of action of IFN on in vitro Ig production by lymphocytes seems twofold; direct regulatory action on B-cell activation as well as through T-helper cell mediation.
Compared to IFN-α, IFN-γ showed 2- to 6-fold stimulating activity on Ig production by MNCs. The present study employed the recombinant forms of interferon as well as virus-induced leukocyte interferon to compare their effects on in vitro B-cell differentiation. The effects of both preparations of interferons were similar on B-cell differentiation.
This result confirms the previous experiments which used the conventional leukocyte-IFN preparations.
Further studies on the effects of interferons on the diffferentiation of malignant B-cell disorders would be meaningful.
Acknowledgments
The present study was pursued at the Department of Internal Medicine, University of Ulm in Ulm, West Germany during the period from March 1, 1984 till December 31, 1985, under the financial support from the Alexander-von-Humboldt Foundation.
I deeply appreciate the encoragement and support provided by professor Munho Lee and professor Herrman Heimpel during the investigation of the present study.
Table 1.
Effects of Interferon-α on Immunoglobulin Production by Lymphocytes
| Time IFN added in relation to culture start | IgM production (ng/ml)±S.E. | % change | P value |
|---|---|---|---|
| −24 hrs | 740 ± 16 | + 106 | < 0.005 |
| 0 hrs | 420 ± 12 | + 17 | < 0.05 |
| + 48 hrs | 470 ± 1 3 | + 31 | NS |
| Control culture | 360 ± 1 |
Table 2.
Effects of Recombinant Interferon-γ on Immunoglobulin Production by Lymphocytes
Table 3.
Effects of Preincubation of Cell Fractions with Inteferon on Ig Procuction
| Culture combinations | IgM production (ng/ml)±S.E. | % change | P value |
|---|---|---|---|
| T+B | 286 ± 17 | ||
| Ta + B | 530 ± 23 | + 85 | 0.25 |
| Tb + B | 1680 ± 41 | + 487 | 0.01 |
| T + Ba | <20 | − 94 | 0.05 |
| T + Bb | <20 | − 94 | 0.05 |
Table 4.
Effects of Preincubation of T-cells with Interferon on T-cell Subset after Culture
| Culture combination T |
T cell marker
|
Leu-3/Leu-2 ratio | |
|---|---|---|---|
| Leu-3 (%) | Leu-2 (%) | ||
| T + B (control) | 64 | 21 | 3.1 |
| Ta + B | 75 | 16 | 4.7 |
| Tb + B | 58 | 11 | 5.3 |
| Tc + B | 72 | 15 | 4.8 |
| Td + B | 79 | 14 | 5.6 |
REFERENCES
1. Calvert JE, Maruyama S, Tedder TF, Webb CF, Cooper MD. Cellular events In the differentiation of antibody-secreting cells. Sem Hematol 21:226–2431984.
2. Gainings WE, Lawton AR, Cooper MD. Immunofluorescent studies of the development of pre-B cells, B lymphocytes and immunoglobulin isotype diversity in humans. Eur J Immunol 7:804–8101977.


3. Kirchner H, Tosato G, Blaese RM, Broder S, Magrath IT. Polyclonal immunogobulin secretion by human B-lymphocytes exposed to Epstein-Barr virus in vitro. J Immunol 122:1310–13131979.

4. Bird AG, Britton S, Ernberg I, Nilsson K. Characterristics of Epstein Barr virus activation of human B-lymphocytes. J Exp Med 145:832–8391981.



5. Waldman TA, Broder S. Polyclonal B-cell activators in the study of the regulation of immunoglobulin synthesis in the human system. Advances in Immunology. In: Kunkel H, Dixon FJ, eds. Academic Press, Inc, New York: 32:1–631982.


6. Waldman TA, Broder S, Goldman CK, Frost K, Korsmeyer SJ, Medici MA. Disorders of B cells and helper T cells in the pathogenesis of the immunoglobulin defeiciency of patients with ataxia telangiectasia. J Clin Invest 71:282–2951983.



7. Wangel Ag, Milton A, Egan JB. Spontaneous plaque forming cells in the peripheral blood of patients with systemic lupus erythematosus. Clin Exp Immunol 49:41–491982.


8. Weetman AP, Volkrnan DJ, Burman KD, Fauci AC. Activation, proliferation, differentiation of circulating B cells in autoimmune thyroid disease. J Immunol 135:3138–31431985.

9. Gorski A, Pazek L, Rancewicz Z, Morzycka-Michalik M, Korczak-Kowalska G, Dzierznowska D. Factors affecting immunoglobulin production of lymphocytes from renal allograft recipients. Trans Proceed 15:1777–17801983.
10. Pahwa SG, Theresa M, Quilop J, Lange M, Pahwa R, Grieco MH. Defective B-lymphocyte function in homosexual men in relation to the acquired immunodeficiency syndrome. Ann Int Med 101:757–7631984.


11. Stewart WE. The interferon System. Springer-Verlag, New York: 1981.
12. Gutterman JU, Blumenschein R, Alexanian R, Yap YH, Buzdar AU, Cabanillas F, Hortobagyi GN, Hersh EM, Rasmussen SI, Kramer M, Pestka S. Leukocyte interferon induced tumor regression in human breast cancer, multiple myeloma, and malignant lymphoma. Ann Intern Med 93:399. 1980.


15. Taylor-Papadimitiou J. The effects of inteferons on the growth and function of normal and malignant cells. Interferons. In: Burke DC, Morris AG, eds. Cambridge University Press, 1983;109–147.
16. Balkwill FR, Review Inteferons. as Cell-regulatory molecules. Cancer Immunol Immunother 7:7–141979.

17. Moore M, Potter MR. Enhancement of human natural cell-mediated cytotoxity by inteferon. Br J Cancer 41:378–3891980.



18. Einhorn S, Bloomgren H, Strander H. Interferon and spontaneous cytotoxity in man. Int J Cancer 22:405–4121978.


19. Trinchieri G, Santoli D. Antiviral activity induced by culturing lymphocytes with tumor-derived or virus-transformed cells. J Exp Med 147:1315–1331978.



20. Zarling JM, Sosman J, Eskra L. Enhancement of T-cell cytotoxic responses with purified human fibroblast interferon. J Immunol 121:2002–20041978.

21. Gisler RH, Lindahl P, Gresser I. Effects of interferon on antibody synthesis in vitro. J Immunol 113:438–4441974.



22. Johnson HM, Smith BG, Baron S. Inhibition of the primary in vitro antibody response by inteferon preparations. J Immunol 114:403–4061975.

23. Sidman CL, Marshall JD, Gray PW, Johnson NM. Gamma-interferon is one of several direct B-cell maturing lymphokines. Nature 309:28–301984.

24. Choi YS, Lim KH, Sanders K. Effects of interferon α on pokeweed mitogen induced differentiation of periperal blood B-lymphocytes. Cell Immunol 64:20–281981.


26. Pelton BK, Denman AM. Immunoregulatory effects of interferon-α Clin. Exp Immunol 59:398–4041985.
27. Gmelig-Meyling F, Ballieux RE. A simplified procedure for the separation of human T- and non-T cells. Vox Sang 33:5–81977.


28. Kelly BS, Levy IG, Sikora L. The use of enzyme-linked immunosorbent assay (EKLISA) for the detection and quantification of specific antibody from cell cultures. Immunol 37:45–521979.
29. Sonnenfeld G, Mandel AD, Merigan TC. Time and dosage dependence of immunoenhancement by murine Type II interferon preparations. Cell Immunol 40:285–2931978.


30. Nakagwa T, Hirano T, Nakagawa N, Yoshizaki K, Kishimoto T. Effect of recombinant IL-2 and γ-IFN on proliferation and differentiation of human B-cells. J Immunol 134:959–9661985.

-
METRICS

-
- 1 Crossref
- 0 Scopus
- 7,957 View
- 42 Download
- Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


